Cesium Triflate: The Hidden Hand of Modern Synthesis
Historical Roots Meet Modern Science
Tracing the story of cesium triflate is a bit like charting the development of synthetic chemistry itself. Back in the twentieth century as organic and organometallic chemistry surged forward, scientists constantly pushed the boundaries searching for stable, non-nucleophilic anions. Non-coordinating anions promised to make tricky reactions both practical and reliable. Out of that race to innovate, cesium triflate came up as a dark horse. The big appeal: this salt could toss its weight around in reactions without sticking its nose in where it wasn’t wanted. Ask any seasoned chemist about their 'clean up' nightmares, and most will say that reliable, easy-to-handle salts become heroes very quickly.
Inside the Bottle: What Does Cesium Triflate Bring to the Table?
Open a jar of cesium triflate, and you’re looking at a white, often crystalline compound. It packs a punch with its unique structure: cesium cation paired up with the trifluoromethanesulfonate anion. That mouthful stands for stability under harsh conditions and the ability to behave quietly in the background. The salt doesn’t meddle with other reactants, meaning researchers can run reactions that would fall apart with more reactive partners. The polar nature and large size of the cesium ion helps it dissolve in both polar and some non-polar solvents, a powerful trait for anyone juggling solvent compatibility headaches.
Physical and Chemical Character: Useful and Unassuming
What sets cesium triflate apart is its melting point and stability under moisture and some heat, making storage less of a circus. Unlike hygroscopic alkali salts that drain resources, cesium triflate’s low affinity for water means fewer error-prone steps. Its crystalline structure and relative inertness cut down on side-reactions, especially in organometallic and catalysis settings. In my experience, using reagents that don’t demand round-the-clock coddling goes a long way in real-world labs. The salt breaks down only at temperatures high enough that few synthetic routes need to worry about decomposition. Solubility in acetonitrile, dichloromethane, and alcohols further expands options when designing multi-step sequences.
Production: Not for the Impatient
Making cesium triflate tends to follow a metathesis approach, where cesium carbonate or cesium hydroxide reacts with triflic acid. It’s a straightforward reaction, but anyone who has handled triflic acid knows the hazards; careful neutralization and removal of acid residues are non-negotiable. Improper technique risks dangerous exotherms or unwanted byproducts. The subsequent filtration and drying take patience, especially if high purity is needed. Customer feedback from research teams remains consistent: in-house preparation rarely matches commercial material for large-scale jobs, mostly due to the need for controlled moisture and finely tuned purification steps.
Chemistry in Action: Reactions and More
Cesium triflate often appears in the background of transition-metal mediated reactions. In Suzuki and Stille couplings, it stabilizes labile intermediates, boosting yields where sodium or potassium bases fail. I’ve seen entire project timelines shrink because a switch to cesium triflate slashed side-reactions and poor conversions. Cross-coupling isn’t the only trick; in heterocycle formation and C–H activation, the salt behaves like a referee—guiding competing reactions without stealing the spotlight. There’s a quiet revolution in ionic liquid research, too, with cesium triflate forming highly conductive mixtures that power research in energy storage and microelectronics.
Other Names, Same Impact
In journals or catalogues, cesium triflate also turns up as cesium trifluoromethanesulfonate or cesium trifluoromethanesulphonate. Don’t let the variation trip you up; the chemistry at the core hasn’t changed. It’s not about a single supplier’s label—what matters is the high-purity, anhydrous material that finds its way into cutting-edge R&D worldwide.
Staying Safe: Beyond the Data Sheet
Safety with cesium triflate often comes down to respect for standard protocols rather than dramatic hazards. The absence of significant volatility or strong reactivity means accidental spills rarely lead to life-threatening events, but no researcher should gamble with unknowns. Dust management matters, especially for labs with high volumes or poorly maintained fume hoods: inhalation, though rarely fatal, should always be guarded against. Direct skin contact brings mild irritation at worst, but it’s the culture of routine glove and goggle use that has kept my teams incident-free for years.
Real-World Uses: More than Academic Interest
Industry demand for cleaner, faster, and more selective syntheses keeps cesium triflate a staple in pharmaceuticals, agrochemicals, and electronics. Medicinal chemists favor its gentle hand in forming sensitive carbon-carbon and carbon-heteroatom bonds. Its workhorse role in small-molecule screens and even active pharmaceutical ingredient production demonstrates that practicality matters as much as novelty. In electronics, the material’s ionic conductivity gets tested in prototyping next-generation capacitors and batteries—a sign that sodium and potassium salts no longer have the field to themselves.
Pushing Boundaries: R&D and Emerging Insights
Every year, the research community cracks open new use cases. In catalysis, teams probe how cesium’s heft tunes selectivity unattainable with lighter alkali metals. Ionic liquid research keeps drawing on cesium triflate’s unique properties to design tunable environments for reactions that resist scale-up in traditional settings. Investigators are looking at greener synthesis pathways, too: opportunities to recover and recycle the salt are starting to align with industrial sustainability goals. Researchers remain quick to share both breakthroughs and setbacks, fostering genuine progress.
Digging Deeper: What We Know About Toxicity
Toxicology for cesium triflate remains a work in progress. Most safety data point to low acute toxicity, but inhalation and chronic exposure risks haven’t been fully mapped. Based on the broader understanding of cesium compounds, the risk of bioaccumulation seems low, but that’s no reason for complacency. More work is needed to map long-term effects, especially as application scales up. The professional community often stresses that real safety comes down to workplace culture—a culture sensitive to chemical hygiene across every shift and skill level.
Where Cesium Triflate Might Take Us Next
The future holds promise for cesium triflate, thanks to its flexibility and reliability. Green chemistry movements and process intensification trends push researchers to look for reagents that enable cleaner, simpler, and more sustainable transformations. The compound’s role in ionic liquids, high-throughput reaction screening, and electrochemical applications keeps it near the front of innovation dialogue. Years of daily lab work make it clear: real progress depends on tools that deliver performance with manageable risk. Cesium triflate doesn’t draw headlines, but it delivers real value where it counts—in saving time, driving selectivity, and supporting the next jump forward in synthesis science.
The Story Behind the Salt
Cesium triflate sounds like the kind of substance tucked away on a high shelf in a chemistry lab most people never see, let alone understand. This white, crystalline salt shows up in chemical patents and research papers, but it rarely finds the spotlight. Having spent years in chemical research, I see a lot of materials come and go. Few stick around for as many problem-solving roles as cesium triflate.
Why Chemists Reach for Cesium Triflate
Cesium triflate acts as a super-reliable partner in the world of organic synthesis. For anyone working on new pharmaceuticals or advanced materials, this salt offers a reliable push that speeds up key chemical reactions. You’ll find it in reactions that need a strong, non-nucleophilic base or a trusty catalyst when nothing else quite fits. Research shows chemists lean on it for Friedel-Crafts reactions, where making bonds between carbon atoms quickly and cleanly can save days of work.
Other salts—like sodium or potassium triflate—try to play the same game, but cesium’s heavyweight atomic size helps dissolve reaction barriers. Its large ions push reactants together, driving transformations that simply refuse to work with lighter elements. This detail sets it apart in a very crowded chemical toolbox.
Big Gains for New Drug Design
Pharmaceutical chemistry depends on careful choices at every turn. Small changes lead to safer drugs, cleaner production, and fewer toxic leftovers. Cesium triflate’s clean reaction profile checks all those boxes. It cuts down on byproducts and waste, a real concern now as chemical companies face tighter environmental rules. Publications from the last decade show blockbuster drugs, especially antiviral and anticancer candidates, owe a part of their modern routes to this salt.
Its use stretches beyond building drug molecules, too. Polymer chemists reach for cesium triflate to put together new plastics or smart materials with unique electronic or mechanical tricks. Imagine the kind of flexible screens or medical sensors seen in recent tech headlines—these innovations got there faster because someone had cesium triflate in their drawer.
Environmental Headaches and Responsible Choices
Nothing in chemistry comes easy. Cesium is a rare element, and digging it out of the earth comes with environmental baggage. The mining and processing can scar ecosystems, and there’s always a temptation to overlook that when chasing laboratory success. This is where research needs to slow down and ask: Do we really need cesium triflate for every project, or are there greener, cheaper swaps available?
Green chemistry points the way forward. Alternative bases and catalysts, made from earth-abundant elements, stand ready to cover some of the same ground. If a reaction still demands cesium triflate, better waste handling and recycling programs should sit at the center of every lab’s routine. It takes effort to balance discovery and responsibility, but real progress means facing that head on.
The Takeaway: A Most Useful Tool—Used with Care
Anyone serious about making new molecules knows cesium triflate’s value. Its role in speeding up stubborn reactions puts it on the shortlist of chemical problem-solvers. Still, its benefits come with trade-offs—reliability, cost, and impact all matter. Skillful chemists keep learning, stay honest about resources, and experiment with alternatives. That’s how science moves forward and keeps faith with the world outside the lab.
Understanding Cesium Triflate in Everyday Terms
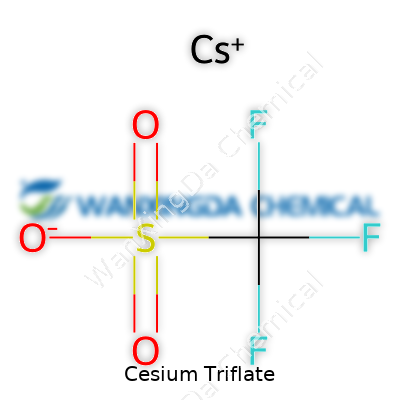
Mention cesium triflate to a group of chemists and you’ll get quick nods because it’s earned its spot as a reliable reagent in advanced labs. The chemical formula, CsO3SCF3, seems like a riddle at first. What matters for most of us is what this mix means and how it finds its way from specialized bottles into processes that affect daily life, technology, and research.
Cesium hasn’t always been in the chemical limelight. It might come up as a footnote in chemistry class, but its involvement with the triflate group creates a compound that stirs interest across the globe. Chemists look to cesium triflate for its salt-like stability and reactivity, qualities built into the formula. The large cesium ion matches well with the triflate’s electronegative trifluoromethanesulfonate group, making this salt special.
Beyond the Formula: Why Cesium Triflate Counts
Living through a few decades of chemical advancements, I’ve seen how reagents like cesium triflate punch above their weight. Organic synthesis, a backbone for pharmaceuticals and new materials, often relies on chemistry that runs more smoothly thanks to this compound. Reactions like Suzuki and Heck coupling, which build up complex molecules, often benefit from the unique kick that cesium triflate delivers. If you ever swallowed a pill for a persistent cough or had a smartphone battery last a bit longer, there’s a good chance innovations from labs using this compound helped along the way.
Facts speak loudly here. Increased yields and cleaner reaction pathways save both money and time. Waste drops, which matters for large-scale production. The cesium triflate formula ensures that the product dissolves easily in both water and organic solvents — kind of like being bilingual in the chemistry world. It’s no exotic rarity, either. This chemical pops up in research papers and global supply chains, quietly smoothing out some of the toughest reactions.
Navigating Concerns and Looking Ahead
Nothing with cesium comes totally risk-free. That’s not a knock against the triflate itself. Cesium mining requires thoughtful stewardship of land and resources. As labs scale up, safe storage and disposal still challenge researchers and manufacturers, since triflate anions resist breakdown in the environment. Open discussions and stricter protocols have already improved conditions in many settings, and sharing best practices keeps everyone safer.
Talking solutions, I see two fronts. First, strengthening supplier transparency and training helps users spot reliable products and handle the chemical safely from start to finish. Second, investing in greener alternatives — or better recycling — supports sustainability. Some university groups and companies already share their findings freely, helping others skip rookie mistakes. Responsible chemistry starts with a solid understanding, and getting the formula right is just the beginning.
So that’s cesium triflate, boiling down not just to six elements on a page, but to how chemistry makes our world tick a bit faster, safer, and smarter. It pays to look beyond formulas and weigh the real-world impact, both for science at large and the people who use its results.
A Salt That Defies Expectation
Pulling a vial labeled “cesium triflate” from a lab shelf always sets my mind working. On paper, it looks like a straightforward salt: cesium, a heavy alkali metal, paired with triflate, the perfluorinated cousin of the more familiar sulfate or nitrate. You’d think tossing it in water would dissolve it as easily as table salt. Reality, though, doesn’t line up with that assumption, and this speaks volumes about chemistry’s ability to upend day-to-day expectations.
What Actually Happens in the Lab
Ask any synthetic chemist who works with transition metal catalysts or organic transformations. Cesium triflate—a.k.a. cesium trifluoromethanesulfonate—does not dissolve well in water, at least not at room temperature or in any practical amount you’d need for aqueous chemistry. A little will disappear, far less than sodium triflate or potassium triflate. If you try forcing more into solution, you just end up with a cloudy mess.
This matters in real work. I’ve sat through more than one research meeting where someone suggests an aqueous route “just using cesium triflate”—as if swapping in a heavier cation would sidestep solubility rules. Soon enough, we’re staring at a beaker with undissolved crystals at the bottom, wishing we'd checked a solubility chart first.
Chemical Roots of Poor Solubility
Science has an explanation. Cesium ions are larger than sodium or potassium, and the triflate anion packs a heavy punch of fluorine atoms. Their combination leads to lower hydration energy, so water just can’t tear the lattice apart like it does with sodium or potassium pairs. Instead, most cesium triflate, especially in powdered form, sits stubbornly on the bottom of the flask.
For comparison, sodium and potassium triflate salts both fall apart easily in water, making them favorites for ionic conductivity and phase-transfer catalysis. Their cesium counterpart stays behind, which is bad news for anyone craving a simple water-based cleanup.
Wider Impact: Solvent Choices and Chemistry Problem-Solving
Struggling with cesium triflate’s solubility forces chemists to think harder about solvent selection. The industry leans into polar organic solvents—acetonitrile, dimethylformamide, methanol—because these can drag the cesium and triflate apart. Switching from water slows some reactions but increases chances of getting a clean result, a lesson written into many organic synthesis protocols. Going down this path, though, nudges research farther from green chemistry’s preferred water-based systems.
Every synthetic project, whether in an academic lab or in pharmaceutical development, runs up against chemical facts like this. Choosing reagents isn’t just about price or availability. It’s a matter of understanding how molecules interact so the process doesn’t grind to a literal halt. Engineers and chemists can’t shortcut fundamental limitations, though some keep searching for workarounds—new solvents, sonication, or thermally-assisted dissolution all make the list of “maybe this will work.”
Finding Solutions and Moving Forward
What helps here isn’t just an eye for detail but respect for the facts. Checking published measurements, like the ones tucked away in handbooks or patents, keeps waste down and budgets intact. Improving solubility often means adjusting the workflow—using more soluble salts where possible, or leveraging cesium triflate’s unique properties only where its use makes a crucial difference.
Trusting data and sharing hard-won experience in the lab means fewer false starts and a clearer path to efficient, effective reactions. Cesium triflate’s story serves as a reminder: the most basic-seeming chemicals sometimes behave in ways that demand a closer look before dropping them into the reaction flask.
Understanding Why Proper Storage Matters
Chemistry labs never feel like just a collection of glassware and bottles—each compound brings its own quirks and needs. Cesium triflate stands out from the usual set of salts. This pale solid, often used as a catalyst or reagent in organic synthesis, packs a punch in reactivity and deserves a seat at the grown-ups’ table when it comes to storage.
I’ve seen colleagues treat shelf chemicals with a shrug, expecting powders to behave no matter where they land. That attitude rarely ends well with materials like cesium triflate. Ignoring the conditions under which it sits means running the risk of clumped, degraded, or even hazardous material down the line. This isn’t scaremongering. Reports and peer discussions tell clear stories: strong electrophiles like cesium triflate absorb moisture from air. If humidity makes its way into the bottle, what once acted as a reliable triflate salt turns sluggish or unpredictable in the flask, wasting both time and money.
Learning from Past Experience
I'll admit, in my early days, a carelessly stored bottle of cesium triflate wound up with a crust of sludge at the rim—a reminder that it doesn’t forgive shortcuts. Not only did it take longer to coax the material back into usable condition, it also led me to dig through literature for best practices. Those lessons stick. Cesium triflate’s role in fine organic reactions, especially when high yields or cleanliness matter, means any contamination from air or moisture will haunt results.
Smart Storage Habits for This Sensitive Salt
Worrying about safety and quality in the lab never ends with exotic chemicals. For compounds like cesium triflate, a few habits make all the difference:
- Keep it cool, but not freezing. Standard recommendation puts it in a tightly sealed container, in a dry area with controlled room temperature. Don’t lock it away in a freezer or fridge unless manufacturer guidance insists; cycling cold containers in and out of ambient air often invites condensation and trouble.
- Minimize light and air exposure. I’ve watched careless exposure turn fine white powders into sticky masses. Screw caps tightly, and work quickly. Shove silica gel packs or other drying agents into the storage container if possible. This dries out stray moisture before it sees the salt.
- Label well and date every use. Even if you remember, others may not. Most lab mistakes trace back to missing or fading labels.
Why the Details Count
Every time researchers cut corners, labs pay for it. Contaminated cesium triflate doesn’t just slow the workflow—it compromises experimental data, leads to failed reactions, and inflates costs with unnecessary repurchasing. Search major journals or lab notes, and you’ll see repeat cautionary tales.
Getting everyone in the lab on the same page boosts safety and science at once. Regular reminders—whether through meetings, signs, or friendly nudges—lock in habits. Make storage best practice as normal as wearing goggles or logging chemical use. It might feel tedious, but nothing beats a spotless reaction and easy reproducibility come publication time.
Coming across cesium triflate in a lab takes me back to days hunched over flasks, gloves on, eyeing every move. Nobody forgets the first sharp scent of an organofluorine compound or the stories about what goes wrong if a lid isn’t tight enough. Cesium triflate isn’t a showboat—quiet, white, even kind of boring in the jar. The catch is, it brings together the hazards of cesium and triflate, each with its own punch.
Protecting Yourself and the Lab
The first thing I learned: you don’t handle this salt in street clothes or with bare hands. Nitrile gloves are a must, not the cheap stuff since splits and tears feel like invitations for accidents. The powder flows smoothly and sneaks onto surfaces or up your nose in a drafty room. Goggles with side shields, a fitted lab coat, and a breathing mask if there’s a risk of dust round out the essentials. The compound can irritate skin and mucous membranes, and no one wants a trip to the nurse explaining why their hands tingled for hours.
Engineering controls stack up quickly. Fume hoods serve as insurance. I’ve seen plenty of researchers try to save time by working open-air, only to spend an afternoon scrubbing floors after one clumsy bump. A fume hood pulls volatile releases away, keeping cesium triflate where it belongs. Benchtop work just isn’t worth the risk.
Safe Storage and Housekeeping
Shelf space in a chemical stockroom is always tight, but cesium triflate earns a spot in a sealed, labeled bottle, tucked away from water, acids, and bases. Any condensation or accidental splash sets off chain reactions you’d rather read about than witness. Desiccators help keep things dry since any absorbed moisture makes handling tricky.
Solid waste and leftover reagent build up on benches if no one’s diligent. I treated every paper towel and glove that touched cesium triflate like hazardous material—not because anyone loves paperwork, but because forgetting leads to bigger messes. Specialized chemical waste bins make cleanup less stressful for those who may not know what’s in the trash.
Training and Vigilance
Lab safety flows from culture, not from posters on the wall. Once, a summer intern asked how bad triflate exposure could get. Rather than lecture, I handed over the safety data sheet and walked her through the emergency shower and eyewash station. That walk-through worked better than a dozen warnings.
Emergency procedures deserve a spot in team meetings. Even practiced chemists fumble when powder gets on a sleeve or someone sneezes near a weigh boat. Immediate decontamination works—plenty of soap, running water—but only if everyone knows the drill. Local health authorities and university guidelines put it plainly: prevention beats response every time.
Better Practices for the Future
The core issue comes down to respect and preparation. Personal safety gear, secure storage, and shared training keep small accidents from snowballing. If work demands routine handling of cesium triflate, investing in single-use tools and closed weighing stations pays off. The goal is simple: finished experiments, healthy chemists, and stories that end with a laugh, not a warning.