Chlorodifluoroacetic Acid: A Closer Look at Its Role and Responsibility in Chemistry
Historical Development
Chlorodifluoroacetic acid emerged from the broader exploration of halogenated organics, tracing back to the chemical curiosity of the late 19th and early 20th centuries. Chemists back then began swapping hydrogen atoms with halogens, which led to the discovery of an array of acids, solvents, and intermediates. As refrigeration and polymer industries exploded, the world paid more attention to fluorinated compounds. Chlorodifluoroacetic acid, a byproduct of many fluorination processes, caught the eyes of researchers not only as a chemical curiosity but also as a valuable intermediate for industries hungry for novel building blocks. Once fluorine chemistry gained momentum, so did the awareness and investigation of compounds like this one, making it a staple in technical laboratories and chemical manufacturing lines.
Product Overview
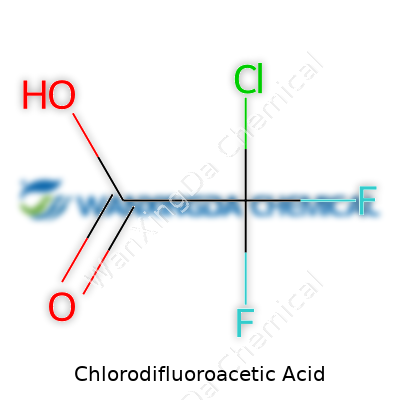
Chlorodifluoroacetic acid isn’t an everyday household item, but it holds serious weight in specialty labs and industries. With a chemical formula of C2HClF2O2, it sits among haloacetic acids, meaning it’s got one chlorine and two fluorine atoms hanging on its acetic acid backbone. Its role isn’t so much about everyday consumption as it’s about making things happen at the molecular level — be that in pharmaceuticals, agrochemicals, or certain advanced polymers. Researchers often see it as a rung on the ladder to synthesizing more complex chemicals, each with its own promise or peril for health, industry, and the environment.
Physical and Chemical Properties
This acid shows up as a clear, colorless liquid or sometimes a crystalline solid, depending on storage conditions and purity. It has a sharp, pungent smell — characteristic of smaller halogenated acids. Its boiling point rests higher than standard acetic acid but lower than most industrial solvents. That comes from the heavy halogens pulling the molecules together through strong intermolecular forces. It readily dissolves in water and organic solvents, showing the versatility that chemists love when they’re making target molecules. When handled in open air, it can fume due to its reactivity and volatility, which keeps lab techs wary. As for reactivity, chlorodifluoroacetic acid does not sit quietly — it participates in a wide range of substitution and elimination reactions, opening new paths in both laboratory and industrial chemistry.
Technical Specifications & Labeling
Lab stocks and commercial shipments typically mark this acid with standard hazard pictograms and precautionary statements. Concentration and purity levels vary by supplier, but the best lots boast purity above 98 percent. Such labeling is more than regulatory — it keeps professionals safe and processes reliable. Every container requires chemical-resistant labeling that won’t fade with splash-back or time, because it’s easy to mix up small bottles on crowded shelves. Suppliers include crucial data about concentration, presence of stabilizers, and guidelines for handling, since improper storage or mislabeling could mean dangerous exposure or spoiled material.
Preparation Method
Industrial synthesis draws on powerful halogenation techniques, often starting with monochloroacetic acid. Manufacturers bombard the starter acid with fluorinating agents such as hydrogen fluoride under precise conditions, hoping to swap hydrogen for fluorine without shredding the carbon skeleton. This dance with corrosive reagents and strict temperature controls yields not just chlorodifluoroacetic acid, but sometimes its siblings — difluoroacetic or trichloroacetic acids — so purification takes patience and skill. Laboratory syntheses usually avoid large-scale excesses but still demand good ventilation and full protective gear, given the volatility and corrosivity of reactants and products alike.
Chemical Reactions & Modifications
Chemists rely on chlorodifluoroacetic acid for its aggressive halogenation properties. It’s reactive with alcohols, amines, and alkali metals, creating a playground for organic synthesis. The acid can undergo decarboxylation under certain conditions or serve as a starting point for nucleophilic substitution reactions. In drug development, swapping out the acid group with various organic fragments lets teams probe different biological activities. This makes it invaluable when optimizing molecules for activity, potency, or metabolic stability. In advanced materials, modifications of this acid help tailor properties like water resistance, dielectric constant, and thermal stability. Such versatility keeps it at the front lines of both applied and fundamental chemical research.
Synonyms & Product Names
Chlorodifluoroacetic acid appears in literature and trade catalogs under several names. One might see it as CDFAA, or simply as 2-chloro-2,2-difluoroacetic acid. Some specialized catalogs also call it chloro-difluoroacetic acid, keeping the arrangement of halogens clear for those scanning for specific substitution patterns. Understanding the alternate naming system matters, especially when collaborating across borders or disciplines, since a missed synonym could stall work or waste resources.
Safety & Operational Standards
This acid is not something to splash around without care. Direct skin contact leads to burns, and inhalation of its vapors can irritate or damage respiratory tissue. Lab protocols require acid-resistant gloves and splash goggles, and fume hoods aren’t optional. Storage involves tightly sealed, labeled containers, usually glass or certain plastics resistant to corrosives, far from incompatible chemicals like bases, oxidizers, or organic solvents prone to violent reactions. Disposal also needs careful planning — neutralization steps and secure collection for hazardous waste channels. Compliance with these rules means fewer accidents and greater peace of mind in both academic and industrial settings.
Application Area
Chlorodifluoroacetic acid draws attention mainly as a synthetic intermediate in pharmaceuticals, agrochemicals, and performance materials. Drug chemists use it to introduce fluoroalkyl groups, often boosting bioactivity or metabolic stability in ways that hydrogen or chlorine substitutions can’t achieve. The agrochemical sector tests it as a bridge to herbicides and pesticides with tailored activity and longer environmental persistence. Materials scientists turn to it for new polymers or surface treatments, aiming to add chemical resistance or tweak electronic properties in circuit boards and specialty films. Although it’s not the endpoint in most applications, its use as a foundation means that safety, traceability, and integrity in handling matter all the way up the supply chain.
Research & Development
Academic labs and corporate R&D teams keep looking for greener, more selective fluorination routes, hoping to harness the value of this acid without the health and waste problems that usually tag along with halogenation. Some efforts target catalysts that work at lower temperatures or in more benign media. Others explore ways to recycle byproducts and minimize toxic effluents. In drug and material discovery, the acid’s halogen content attracts interest for crafting new carbon–fluorine bonds, which often unlock uncharted performance frontiers. Not every experiment leads to a blockbuster result, but every new insight helps shape safer and more efficient production pathways.
Toxicity Research
Like most organofluorines, chlorodifluoroacetic acid comes with environmental and biological concerns. Its sharp acidity and persistent halogen content make it rough on aquatic life if released unchecked. Toxicological tests in lab animals point to corrosive effects on skin and mucous membranes, with potential for longer-term organ damage if exposures run high or chronic. Environmental chemists track its breakdown and movement in soil and water, knowing that some halogenated acids don’t break down easily. Monitoring, safe containment, and best practices in waste treatment stand central to responsible use. Industries and regulators keep pushing for data-driven standards, knowing that prevention costs less than cleanup.
Future Prospects
What lies ahead for chlorodifluoroacetic acid largely depends on advances in green chemistry and regulatory shifts. As demand for fluorinated pharmaceuticals and advanced materials rises, so will the search for cleaner synthesis and better risk management. Young chemists see room to redesign both reactions and products using milder reagents or better catalysts. There’s also momentum building toward tighter life-cycle tracking — from cradle to grave — so every ounce of the acid produced finds its highest-value use and avoids accidental release. Adoption of digital process controls and AI-guided manufacturing could make production both safer and more transparent, offering stronger assurances to workers, consumers, and environmental stakeholders alike.
Looking Past the Technical Jargon
Chlorodifluoroacetic acid belongs to the quirky corner of the chemical world, yet its footprint stretches farther than most people realize. Many people wouldn’t spot it on a standard list of familiar chemicals, though it weaves through a surprising range of manufacturing and research sectors. I’ve seen how these lesser-known compounds can fuel improvements in products many take for granted, and I’d like to unpack what makes this one stand out.
Hidden Benefits for Industry
At its core, chlorodifluoroacetic acid acts as a building block for other molecules. Chemists value its role as a key ingredient in making advanced fluorinated compounds. These aren’t just esoteric lab products—they’re the foundation of things like certain crop protection chemicals and cutting-edge pharmaceuticals. I’m convinced that this quiet influence matters because these sectors drive huge progress in food security and disease treatment.
The impact ripples out. In the past, researchers in chemical manufacturing have used chlorodifluoroacetic acid to create specialty refrigerants, which are crucial for efficiency in cooling systems. With regulations clamping down on ozone-depleting substances, there’s an active hunt for replacements that can balance function and safety. This acid has shown promise in providing a stepping stone for cleaner, more responsible options.
Health and Environmental Stakes
Few of us ever see chlorodifluoroacetic acid in its pure form. Its value isn’t in household goods, but in what it allows companies to make. Still, where there’s manufacturing, there’s risk. Mismanagement can create lasting effects on soil and water. I’ve watched regulatory agencies step in to keep tabs on emissions and waste, but oversight can only go so far. It takes real investment in monitoring and education to keep such chemicals working for us, not against us.
Over the past few years, scientists have raised questions about how these intermediate compounds behave outside the factory—whether they break down or end up lingering. Chlorodifluoroacetic acid falls under the broader PFAS family, known for their tough, long-lasting chemical bonds. That means leaks or dumping can stick around much longer than anyone wants, pulling this substance into worldwide discussions about ‘forever chemicals’.
How Do We Move Forward?
Progress rarely follows a straight line. We need companies, labs, and regulators to keep asking tough questions about safety and sustainability. Open data sharing would help hold manufacturers accountable, and more independent testing would give communities around production sites peace of mind. The lessons from other persistent chemicals underline the need for proper disposal methods and investing in safer alternatives.
The story of chlorodifluoroacetic acid is one of balance—balancing usefulness with responsibility. The promise of better pharmaceuticals and efficient refrigeration gets weighed against the shadow of environmental harm. My own view: steering chemical innovation toward environmental stewardship matters as much as any miracle material, and it’s time more people caught up to that idea.
Reality in the Chemistry World
Working in labs for years, I’ve seen chemicals get underestimated until someone ends up with a nasty burn or a lab’s air thick with an eye-watering fog. Chlorodifluoroacetic acid, with its intimidating name and sharp, acidic bite, is nothing to shrug off. Folks who deal with it daily will tell you that taking shortcuts invites trouble, and mistakes have a way of coming back hard.
Goggles and Gloves: Not Just for Show
Splash a strong acid and you’ll remember what you wore, or wished you did. Before ever cracking that bottle, I reach for nitrile gloves (never latex), splash-resistant goggles, and a full lab coat—acid-resistant, ideally. The acid loves to damage skin and eyes, so leaving wrists or neck exposed courts disaster. Chemical burns don’t give second chances.
The Importance of Good Ventilation
Crack a container of chlorodifluoroacetic acid, and the fumes punch you fast. A chemical fume hood belongs in every workspace where this stuff gets used. I don’t lean over the bench—always keep the container well inside the sash. Breathing in acid vapors doesn’t just tickle the nose; it can scar lungs. If your lab’s ventilation rattles or underperforms, raise hell to get it fixed. Short-term exposure knocks you back, and that damage sticks around.
Storage: Not an Afterthought
Shoving chlorodifluoroacetic acid in with general supplies risks more than clutter—this belongs away from bases, oxidizers, and anything water-reactive. I use tightly sealed containers, labeled with permanent marker large enough for anyone to see. Heat or sunlight breaks down containers and compounds, so keep storage cool and dark. If a bottle gets old or starts looking warped or discolored, it goes to hazardous waste collection—not the trash.
Clean-Up and Spills: Acting Fast
Spills create panic for rookies and headaches for veterans. The fix: practice makes speed. I keep spill kits close, stocked with absorbents rated for acids and neutralizers that don’t trigger a vigorous reaction. Baking soda might spring to mind, but specialized neutralizers stay safer and control the release of gases. After cleaning, ventilate the area and double-bag the wipes or residue for disposal. Leaving a mess for the night shift risks someone else’s hide, which isn’t fair.
No Eating or Drinking in the Lab
I’ve watched coworkers eat lunch without washing up after handling acids, then wonder why their mouth tingles. Traces of chemicals cling to fingertips and sleeves. It only takes absentmindedly rubbing your eye to create a medical emergency. Wash hands twice and swap to fresh clothes before touching food.
Training Saves Lives
Reading a label doesn’t substitute for practice. Training isn’t a box-check activity. See how the acid reacts in a controlled environment, and you learn to respect it. In one job, we ran spill drills twice a year. That routine kept nerves steady during real emergencies.
Moving Forward
Chemicals like chlorodifluoroacetic acid don’t care about intention, only caution. The best protection lies in gear, careful routines, and respect for the hazard. It’s easy to get comfortable around familiar risks, but each shortcut shaves off a layer of safety until something goes wrong. Prioritize upgrades for old equipment. Speak up if the right gloves or goggles aren’t around. And always remember: safety gets built from habits carried out every single time.
Why Getting the Formula Right Matters
Chemistry isn’t always about flashy reactions or wild colors in test tubes. Sometimes, the small details—like the correct formula for a compound—carry the most weight, not just for the folks in lab coats but for anyone connected to environmental safety and public health. Chlorodifluoroacetic acid, with its tongue-twister name, stands as a solid reminder that you need to get the fundamentals right. Its formula is C2ClF2O2. That’s one chlorine, two fluorines, two carbons, and two oxygens. On the page, it may look simple. In reality, it connects to much bigger issues.
The Chemical World Isn’t Just for Chemists
I remember years ago, digging through dusty reference books during college, hunting for the tiniest error because one wrong atom could trip up a whole experiment. Chlorodifluoroacetic acid might not sound familiar at your neighbor’s barbecue, but its presence links back to industries you count on daily. It pops up in the chemical manufacturing sector as an intermediate in the production of more complex fluorinated compounds. Think pharmaceuticals, agrochemicals, and refrigerants—the kind of products that end up in households, fields, and hospitals.
Tiny Changes, Big Impact
The two fluorine atoms in the molecule are no small detail. Fluorinated chemicals can be incredibly useful, like the difference between a nonstick pan and a burned breakfast. At the same time, some of these compounds—if not handled with care—can lead to pollution problems. That’s where environmental stewardship, solid regulation, and transparent data matter. Mislabeling a compound’s formula or misjudging its structure can ripple out into mistakes during manufacturing, slip-ups in disposal, and even health hazards if accidental exposure occurs.
What’s at Stake
Getting chemical formulas right isn’t just about earning an A in class. Take perfluoroalkyl substances (PFAS) as a lesson. These man-made chemicals have made international headlines, thanks to their persistence in the environment and possible health risks. Chlorodifluoroacetic acid doesn’t fall directly into the PFAS group, but it sits near enough on the spectrum that its fate—where it goes, how it acts, what it turns into—deserves attention. If an industrial process releases this compound unchecked, the chlorine and fluorine inside won’t just vanish. They stick around, traveling through soil, water, and sometimes into living things.
Solutions That Start With Honesty
To tackle these problems, simple honesty in reporting chemical structures is the first line of defense. Labs, industries, and regulators alike count on accurate chemical data to make good decisions—whether they’re designing a safer pesticide or trying to keep communities out of harm’s way. Solid research and transparency build trust. On a larger scale, stricter rules about chemical emissions, waste tracking, and routine monitoring of water and soil keep surprises to a minimum.
Science Demands Respect for Details
Every time we cut corners on chemical data, somebody picks up the tab—sometimes the planet, sometimes future generations. It doesn’t take a leap of imagination to picture the fallout from one wrong label. By sticking to facts, being precise about chemical formulas like C2ClF2O2, and not letting small errors slide, we keep the foundation strong for public health, industry progress, and environmental safety.
Hard Lessons from Handling Tough Chemicals
Chlorodifluoroacetic acid pops up in a handful of conversations among folks working with specialized synthetic chemicals. The acid’s formula—C2HClF2O2—carries a bite, not only for what it does in a lab but for the risks it brings outside the flask. A memory comes to mind of a routine day at a chemical storeroom, standing in front of a cabinet labeled with faded warning stickers. Every label told a story about accidents that happened not from lack of knowledge, but from ignoring obvious risks.
Experience Is a Steady Teacher
Fifteen years working in research brought me face-to-face with compounds that act up when dumped into the wrong bottle or left in the wrong room. Chlorodifluoroacetic acid doesn't announce itself like sulfuric or hydrofluoric does—no steam, few visual cues. Yet, you treat it with the same care because leaks and spills make a mess that’s tough to clean and dangerous for skin, lungs, and whoever cleans the floors next.
Straightforward Steps Make the Difference
I never liked overcomplicated safety routines that no one follows. A practical approach goes further than laminated instruction posters. Workspaces thrive when everyone understands the chemical’s quirks. For this acid, glass bottles with tight, chemical-resistant caps beat plastic every time. Fluorinated solvents often gnaw at common plastics, leaving a sticky mess over months. Leave this acid anywhere near heat or sunlight and the risks only get worse—pressurized gases can form, bottles swell, and basic labels peel off.
A good storeroom sets acid containers far from anything reactive. Mixing chlorodifluoroacetic acid and metals, bases, or even organic substances spells trouble. Climate control seems unnecessary until you see how high summer heat stirs up vapors despite sealed bottles. I’ve sited bottles in low cabinets, behind closed doors, where spills can be contained to trays lined with absorbent mats.
Labeling and Responsibility Go Hand in Hand
Labeling turns out to be more than compliance. A clear, printed tag tells everyone who enters the space what to expect, what gear to grab, and what to avoid. I keep emergency procedures taped inside the storeroom, not buried in management’s basement binder. A proper eyewash station nearby is standard, not a luxury. Even seasoned workers hit a patch of bad luck—one slip, one splash, one hasty reach.
Regulations and Real-World Adjustments
People in labs often talk about OSHA and EPA like distant authorities, but their rules make sense when you’ve seen fresh acid burns or an evacuation over fumes. Keeping up with federal and local storage rules earned me less hassle from inspectors and easier nights sleeping. If I notice an unlabeled or aging bottle, I make it a point to re-label or properly dispose of it. This common sense approach saves time, worry, and health in the end.
Practical Solutions Come from Shared Knowledge
Not every storage challenge gets solved by spending money or locking doors. Sharing experiences between shifts, raising safety concerns in staff meetings, and having the guts to slow down when something feels off—these do more than any official sign. I’ve seen young techs quietly fix labeling mistakes after watching one bad day unfold. Peer pressure shapes better habits than memos alone.
Storing chlorodifluoroacetic acid shouldn’t feel mysterious or out of reach for those willing to respect its hazards. By focusing on clear labeling, chemical compatibility, secondary containment, and a living culture of awareness, storerooms stay safer and the acid does its work without becoming a headline for the wrong reasons.
Unpacking the Risk Behind an Unfamiliar Name
Most folks might breeze past the mention of chlorodifluoroacetic acid unless they’re involved in chemistry or environmental science. This chemical has found its way into the conversation because of its connection to refrigerants and industrial processes. While some call it niche, its legacy as a byproduct of larger chemical reactions makes it a concern worth noticing.
Potential Health Hazards
Breathing in chemicals at work or from pollution always comes with risk. Chlorodifluoroacetic acid is no exception. Even without mass reporting of poisonings in daily life, toxicity data from animal studies points to its ability to irritate mucous membranes, impact lung health, and cause headaches, nausea, or worse with higher exposures. Most people don’t encounter it in their kitchens or living rooms, but the workers who handle it or live near industrial sites take on a bigger risk. Chemicals don’t need to cause immediate, obvious harm to be a problem. Prolonged or accidental exposure—even at low levels—can increase the burden on the body, and sometimes, symptoms don’t show up until much later.
Impact on Water, Soil, and Air
From a practical standpoint, the environment catches what industries throw away. Chlorodifluoroacetic acid holds up in water and soil, not breaking down as quickly as some might hope. It’s showing up in wastewater, streams near factories, and even in some studies looking at groundwater. Persistent pollutants add up over time. Once they get into water supplies, the filtering systems used by towns and cities sometimes aren’t enough to fully remove these chemicals.
Direct impact on plants and wildlife matters too. Studies raise red flags about long-term chronic effects, even with small doses, putting aquatic life especially at risk. These animals can't simply move to cleaner water when pollution drifts downstream. With new technologies pushing for cleaner refrigerants, some replacements produce chlorodifluoroacetic acid as a byproduct, compounding the problem instead of solving it.
Experience at the Intersection of Science and Community
Living near industrial corridors or working in manufacturing brings a different perspective on chemical risk. Over the years, I’ve seen communities struggle with unexplained sickness, only to discover pollution as the culprit after long fights and investigations. Public trust falters when industry and regulators aren’t transparent. The lesson hangs over every chemical story: out of sight doesn’t mean out of risk.
Pushing for Sensible Solutions
Better transparency and regular testing can prevent communities from becoming afterthoughts. Industrial users need clear rules and robust safeguards—sealed systems, real-time leak detection, and adequate worker protection prove far more effective than relying on cleanup after the fact. Water utilities could upgrade treatment processes, using advanced filtration to keep persistent chemicals out of taps.
Community watchdog groups often bridge the gap between people and policy. Long-term monitoring and health tracking give scientists the data they need to connect pollution with real-world outcomes. When companies and local governments share information openly, communities can take practical steps to safeguard their health and the places they call home.