Difluoroacetic Acid: Tracing the Path from Chemistry Bench to Industry Mainstay
A Chemical with a Shifting Story
Difluoroacetic acid might sound like one of those esoteric compounds that only pop up in research labs or niche industries, but its journey goes much farther back than most people realize. Chemists in the early twentieth century began playing with simple fluorinated compounds before much was understood about their unique behaviors. Making difluoroacetic acid was originally a shot in the dark, accomplished only after careful studies with hydrofluoric acid and the controlled introduction of fluorine atoms. These were dangerous times for early chemists who rarely shied away from the health risks. Over time, as synthetic routes matured and analytical chemistry sharpened, difluoroacetic acid found a place among the handful of small, highly reactive acids that drew intense interest from organic and medicinal chemists. That the story is still evolving speaks more to the unpredictability of the science than the predictability of industrial demand.
What Sets Difluoroacetic Acid Apart
If you’ve ever handled strong acids, you probably know what to expect—sharp smells, corrosive bottles, and stern safety cautions. Difluoroacetic acid doesn’t buck the trend. This compound, built around a two-carbon skeleton swapped with two fluorines, stands out because those fluorine atoms do most of the chemical talking. They give difluoroacetic acid a much higher acidity than most carboxylic acids but not as punchy as trifluoroacetic acid. That balance matters in several lab processes where overkill or underperformance leads to either damage or inefficiency. In its pure form, this colorless liquid has the signature bite of a strong acid—fumes that should keep you alert and a corrosiveness that runs through glassware in no time if you’re not paying close attention. Chemists who have spilled even a drop or caught a whiff in the air know to leave the bravado at the door because the compound seldom offers a second warning.
Stepping into the Lab: Technical Details and Labeling Realities
On the labeling front, difluoroacetic acid bottles get the full treatment—hazard diamonds, corrosive icons, and regulatory hazard codes—reflecting the seriousness with which safety officers approach its use. The technical specs often detail concentration, purity (with levels often above 98%), and water content. Measuring these isn’t a simple task; precision counts because even minor impurities shift the behavior of the acid during reactions. The origins of any batch—whether it’s made in a high-throughput commercial reactor or a university lab—carry weight, as tracking impurities helps investigators pinpoint causes of failed syntheses and unexpected side products. Every experienced chemist develops a healthy respect for keeping bottles tightly sealed and clearly labeled, knowing the consequences of misidentification spill far beyond wasted chemicals.
Old-School Chemistry: Ways to Make Difluoroacetic Acid
Producing difluoroacetic acid isn’t like brewing up vinegar or fermenting ethanol. Applying fluorine atoms onto carboxylic acid frameworks demands conditions that most processing plants shun. Early routes used silver difluoroacetate, a precursor that cost far more than anyone wanted to pay. Later, direct halogen exchange reactions using bromodifluoroacetic acid improved matters, but scaling up these processes required attention to detail—fluorination by its nature can turn hazardous quickly. Phones ring regularly in chemical supply companies with panicked questions whenever new graduates find themselves responsible for these reactions. The trick is minimizing byproducts such as monofluoro- or trifluoroacetic acid, both unwelcome in downstream syntheses. Clean workups and reliable purification, often by distillation under reduced pressure, make more difference than the choice of starting material.
Under the Microscope: Chemical Behavior and Modification Opportunities
If you drop difluoroacetic acid into a flask of basic solution, the reaction zips along as expected, with the acid proton eagerly snapped up to form a difluoroacetate salt. That same acidity tempts a host of nucleophiles, though the real interest comes from how altering those two fluorines changes the downstream chemistry. Medicinal chemists, for example, are always in pursuit of small tweaks—swapping fluorines in and out, attaching fragments to the carboxyl group. These modifications unlock whole new classes of drugs or agricultural chemicals, mostly because fluorine substitution can transform the fate of a molecule inside living cells. A seasoned synthetic chemist uses difluoroacetic acid as a tool for building more advanced molecules or probing the limits of existing reaction sequences. In practice, pushing the boundaries means navigating not just acidity but also the electron-withdrawing nature of fluorine, which sometimes requires risk-taking with unknown byproducts.
What’s in a Name: Synonyms and Recognizable Labels
Over the years, difluoroacetic acid earned its fair share of alternative names, many of which confuse rather than clarify: 2,2-difluoroethanoic acid, DFA, or even “CHF2COOH” in shorthand. In research papers, the chemical acronym appears so often that newcomers sometimes miss the deeper connection to a family of halogenated acids. These different names matter when ordering from global suppliers or digging through literature, as mistakes can slip in easily—one misplaced fluorine atom on a label can doom an experiment or introduce an unexpected safety risk. Reading the fine print and learning the subtle differences between names remains a rite of passage, reminding everyone that chemical literacy counts every bit as much as technical skill.
Staying Safe, Staying Sharp: Operational Norms in Industry
Anyone who spends real time with reactive fluorine compounds brings tales—scorched gloves, etched glassware, unexpected pressure buildup. Difluoroacetic acid doesn’t offer much forgiveness for lapses in safety: proper ventilation, nitrile gloves, and eye shields are mandatory, with fume hoods turned on and spill kits within arm’s reach. Regulatory agencies such as OSHA and the European Chemicals Agency outline best practices—never store near incompatible reagents, avoid open flames, and ensure secondary containment. Veterans in the field develop habits: double-checking seals, never rushing by the bench, and keeping antidotes or eyewash stations close. Training young chemists means passing on both the unwritten rules and the written safety data, knowing that no checklist can guarantee safety against complacency. Experience counts more with difluoroacetic acid than with many traditional lab chemicals, as mistakes tend to have outsize consequences.
Where Difluoroacetic Acid Leaves a Mark
Industry finds use for difluoroacetic acid primarily in the synthesis of pharmaceuticals, where adding fluorine atoms can radically alter how a drug behaves in the human body. Many blockbuster medicines and crop-protection agents owe their effectiveness to this kind of molecular tinkering. The food industry rarely touches the compound due to toxicity and flavor concerns, but labs focused on creating diagnostic agents, enzyme inhibitors, or advanced polymers often turn to difluoroacetic acid for help. In my time consulting with agricultural chemical producers, the value showed up in the clever way difluoroacetic acid transforms inert baseline structures into hardier, longer-lasting field agents. What comes as a surprise to some is just how these small molecular changes can build up—leading to new materials, better therapies, or fresh problems, depending on who’s steering the research.
Pushing Boundaries: Research, Development, and the Hunt for Better Chemistry
Research teams worldwide keep searching for safer, cheaper, and more sustainable ways to make and use difluoroacetic acid. Techniques such as flow chemistry, in which reagents mix continuously under close monitoring, open opportunities to produce difluoroacetic acid on-demand, cutting down on storage and transport risks. Catalysts tuned to encourage selective fluorination make a big difference, trimming costs and reducing unwanted waste. These projects rarely make headlines, but inside the innovation labs, competition is fierce. Academia and industry collaborate to find tweaks that deliver high yields while minimizing environmental and workplace hazards. If you’ve spent time near leading-edge chemical R&D, you know the satisfaction of finally solving a production puzzle that once ate weeks or months of work.
Toxicity: Grappling with Risks
Every new compound raises questions about safety, and difluoroacetic acid ranks high on lists of chemicals where attention needs to stay sharp. Animal toxicity tests show it can cause organ damage, derail enzyme action, and disrupt metabolic pathways typical of many fluorinated acids. Even experienced techs respect the threat of inhalation or skin exposure, knowing that the symptoms often build gradually—tissue irritation, respiratory trouble, or worse. Environmental toxicity research underscores the need to keep difluoroacetic acid far away from soil and groundwater, as breakdown can linger and accumulate. Hard evidence from published case studies keeps regulatory authorities interested, and waste management protocols reflect lessons learned from past spills. In practical lab work, this means treating even dilute solutions with care, doubling up on containment, and verifying disposal through specialized waste contractors. Old lessons about respect for hazardous chemicals feel especially relevant.
Looking Forward: Opportunities and Unanswered Questions
The future for difluoroacetic acid sits at a crossroads of necessity and caution. Pharmaceutical and agrochemical development will keep calling for sophisticated building blocks, and difluoroacetic acid remains a key tool. Expectations around green chemistry and sustainability pressure industry to refine synthesis and cut down on emissions, so breakthroughs in catalysis or recycling will likely separate leaders from laggards. Tightened regulatory scrutiny could push demand toward safer alternatives, while new applications in polymer science or drug design could boost market importance. Advanced analytical techniques promise cleaner production, and collaborations between universities and manufacturers might shorten the timeline for safer, smarter difluoroacetic acid usage. In my experience, real innovation usually comes from the willingness to question entrenched methods and to face the discomfort of changing protocols that seemed to work well enough. The balancing act between progress and precaution defines the next era for difluoroacetic acid, making chemical judgment and hands-on experience more valuable than ever.
Most people don’t run across difluoroacetic acid in daily life, but in research circles and certain industries, it turns up as a tool with plenty of bite. A few years ago, I spent some late nights with a research group working on organic synthesis, where the name difluoroacetic acid bounced around the benches more than most folks would expect. Scientists use it because it’s a small, punchy acid, and the two fluorine atoms give it a chemistry that stands out from the usual kitchen acids like vinegar or citric.
Difluoroacetic acid isn’t something you buy for a home project. In labs, it’s a reagent that helps build other compounds. Think of it as a wrench that fits certain bolts, particularly in drug discovery and synthesis. The presence of fluorine in drug molecules changes their behavior—helps them last longer in the body, makes them more effective at their job, and sometimes tones down harmful side effects. Big-name medicines often owe their character to these subtle tweaks, and chemists have leaned on difluoroacetic acid for introducing fluorine in just the right way.
Some analysts use difluoroacetic acid in high-performance liquid chromatography, which separates stuff in a mixture so you can see what’s really there. Tweaking the acid content during analysis sharpens the results, pushing accuracy higher. This matters not just for pure science, but for any field keeping tabs on drug quality, food safety, or environmental samples.
Its usefulness comes at a price. Difluoroacetic acid doesn’t belong in your sink or trash. It’s quite a bit tougher than household acids: the fluorine atoms make it corrosive, and its volatility can turn a careless moment into a safety incident. In the lab, we kept gloves and safety eyewear close and cut off its use at the smallest practical scale. Companies that make or ship this stuff rely on tight rules for containment and disposal. Those rules exist for a good reason: hydrofluoric acids, even ones with only a couple fluorines per molecule, don’t play nice with living tissue or ecosystems.
It’s tempting to focus on what difluoroacetic acid helps create, but we can’t forget the costs tucked into the process. Waste handling matters. Workers need proper training, and institutions have to respect chemical safety at every turn. Rather than seeing hazardous reagents as unavoidable evils, it makes more sense to invest in smarter chemistry—processes that use less toxic materials, make cleaner by-products, and still get the job done. Small steps in green chemistry pay out over time. Some labs have switched to milder acids or better recycling, not for show, but because the risks eventually catch up.
There’s real value in understanding where our chemicals come from, what jobs they do, and how we manage their shadows. Difluoroacetic acid plays an outsized role in making molecules that help people live healthier lives, but the balance always matters. Progress depends as much on respect for the tools as it does on the workmanship behind them.
Understanding What You’re Working With
Anyone who has spent time around chemical labs quickly learns that some reagents demand more respect than others. Difluoroacetic Acid ranks high on that list. It looks like any clear liquid, but just because it doesn’t call attention to itself doesn’t mean it’s harmless. Breathing it in or splashing it on skin isn’t a minor inconvenience. From firsthand lab experience, even brief contact brings out its no-nonsense side—think instant skin and respiratory irritation, not to mention long-term health issues if you don’t take it seriously.
Personal Experience Shows Why Gloves and Goggles Are Non-Negotiable
Plenty of folks get tempted to cut corners with personal protective gear, especially during busy lab days. It’s a mistake. Nitrile or neoprene gloves, a lab coat, and snug-fitting goggles aren’t just “suggestions.” I’ve seen what happens when someone tries to pour Difluoroacetic Acid over a beaker with bare hands. Burning and redness can show up almost immediately, and washing the area right away is your only hope to keep it from getting worse. Eyes are even trickier. A splash, even a minuscule drop, calls for a solid fifteen-minute rinse at the eyewash station. Because irreversible damage stays with you long after the experiment ends.
Ventilation Always Pays Off
Few things feel as careless as mixing up a volatile acid in a tight, stuffy room. Difluoroacetic Acid gives off fumes that bite at the nose and throat. Anytime a procedure calls for it, I head straight for a fume hood. Fan systems keep it out of your lungs. As someone who’s spent long stretches in labs, I can confirm: it’s not just about the discomfort. Over time, exposure increases risks for asthma-like symptoms and aggravated respiratory problems.
Storage and Labeling Matter—Not Just For You, But For Colleagues
One oversight that invites trouble is sloppy storage. Difluoroacetic Acid doesn’t take kindly to metal containers and can corrode certain plastics over time. I keep it in tightly sealed glass bottles with clearly marked hazard signs. Once, an unmarked bottle in a shared fridge nearly led to a dangerous mix-up. Luckily, a double-check saved the day. Proper labeling helps the whole team stay safe—especially in busy workspaces where new hands rotate in.
Spill Protocols Aren’t Optional
Spills in the lab happen, sometimes when you least expect it. In the moment, panic makes it easy to reach for the nearest towel. Mistake. Absorbent pads and neutralizing agents keep us from turning a minor incident into a full-blown emergency. From my own experience, practicing drills and reading up on spill protocols before starting the work makes everything easier when the real thing happens.
Building a Culture of Safety
Handling Difluoroacetic Acid safely shouldn’t rely on printed sheets tucked into a drawer. It thrives on routine conversations and a strong sense of responsibility. Trust between team members keeps accidents to a minimum. When someone points out a shortcut or calls attention to missing PPE, that’s not nitpicking—that’s keeping people out of the emergency room. Regular refresher sessions and peer checks raise awareness. Over time, the group expectation becomes: safety isn’t negotiable.
Looking Forward
Some chemicals let you get away with bad habits, at least for a while. Difluoroacetic Acid doesn’t. From proper gear and smart ventilation to crisp labeling and spill readiness, every step counts. Inviting newcomers into that mindset and keeping old hands vigilant turns shared space into a much safer place. In my experience, nobody regrets putting safety first—only skipping it.
Stepping Into the World of Difluoroacetic Acid
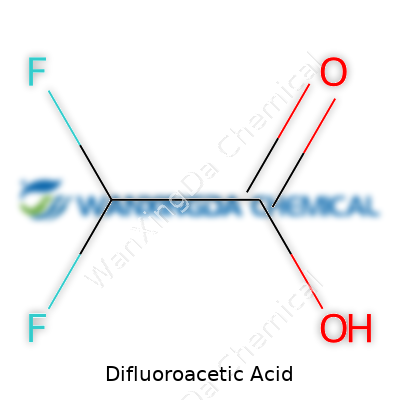
Old chemistry textbooks contain pages full of cryptic letters, but every formula tells a story. Difluoroacetic acid’s formula—C2H2F2O2—packs plenty of story. The formula shows two carbon atoms, two hydrogens, two fluorines, and two oxygens. That may look dry on paper, but this pairing of fluorines to a simple acetic acid backbone sparked more than a new entry on a chart. With just those couple of chemical swaps, routine molecules can head down a very different path in the lab, the environment, and possibly even human health.
What Changes with Two Swapped Atoms?
It’s easy to imagine acetic acid, that classic sour liquid in vinegar. Throw in two fluorine atoms, and chemistry class gets much more interesting. Fluorine isn’t subtle—add it, and molecules get tougher, sometimes sharper, often better at resisting breakdown. As someone who spent hours breathing in faint vinegar fumes during college organic labs, I couldn’t ignore the way fluorinated compounds behaved. Adding fluorine doesn’t just tweak a molecule—it often turns the whole playbook upside down.
Difluoroacetic acid stands out. The fluorines can steer the compound toward unique reactions. Chemists know this means new possibilities for making pharmaceuticals or specialty plastics. End-users want products that last. The flip side: long-lasting molecules can pile up. Our modern world is packed with these fluorine-containing chemicals, and this draws questions from environmental researchers. Can water systems easily wash them away? Or do they linger years after their intended use?
Why Should Anyone Care About That Formula?
People ask what difluoroacetic acid does. Right now, it doesn’t show up on grocery shelves or in household sprays. Still, chemists see it as a key building block. The presence of difluoroacetic acid in a synthesis lab opens access to medications and industrial compounds. These chemicals give modern products strength, stability, or water resistance that old-school chemistry can’t deliver. Medicine, electronics, and fabric coatings owe a debt to fluorine-bearing compounds just like this one.
There’s a clear flipside. Longer-lived molecules often land in unexpected places—rainwater, soils, and streams around the world. Traces of related compounds sometimes surface far from where anyone handled them. This isn’t a minor science footnote. Regulators and scientists worry about “forever chemicals.” Fluorine atoms bind tightly enough that breakdown fails to keep pace with production and release. Every new chemical built off difluoroacetic acid could add to a legacy that future generations inherit.
Better Chemistry, Better Approach
Making stronger and more enduring products has value. Nobody wants electronics or medicine that breaks down too soon. Yet, relying on highly persistent molecules—especially ones built with fluorine—brings pressure to invent smarter. Researchers developing new fluorinated acids push for careful studies of breakdown and potential health impacts right from the start, not after the fact. Claiming ignorance won’t satisfy anyone watching compounds spread across the environment. Responsible scientists advocate for transparency and independent review before new uses expand.
Efforts to design less persistent alternatives would help. Some teams want to build clever chemical “escape hatches” so that molecules like difluoroacetic acid break apart more easily after their job is done. This requires funding, creativity, and real-world testing, but the ideas are already moving forward. Strong partnerships among chemists, toxicologists, and environmental scientists will make or break the next chapter of this story. For a formula as simple as C2H2F2O2, the impact stretches far beyond a laboratory bench.
Why Proper Storage Matters for Difluoroacetic Acid
Difluoroacetic acid sounds like a chemical only seen in specialized labs, but stories about mishandling this kind of substance show that dangers aren’t reserved for big industry. My own time working with chemicals in a university setting taught me that even a minor oversight can make anyone painfully aware of the risks. With difluoroacetic acid, we’re talking about a corrosive liquid that packs a punch on contact, whether it’s through fumes or skin contact. Shortcuts never end well, so putting in the effort upfront keeps people out of emergency rooms.
What Science Tells Us About Safe Storage
Chemicals like difluoroacetic acid don’t respond well to carelessness. The acid releases fumes, breaks down some plastics, and reacts strongly with moisture and incompatible materials. A good rule: store it in containers that resist its aggressive nature, usually high-quality glass or resistant polymers, never regular metal or generic plastic. I once saw an old bottle eat through a cheap plastic cap—that memory sticks.
Keeping it tightly sealed is a must because the fumes aren’t just a hassle, they’re seriously harmful. In most labs, people keep it away from direct sunlight and heat sources to prevent pressure from building up inside the container. Even minor temperature changes can spell trouble. An acid-resistant tray beneath storage bottles adds an extra layer of protection in case the worst happens.
Respecting the Rules Helps Everyone
No one gets praise for following the rules until something goes wrong, then everyone looks at the storage log. The legal standards—those government safety regulations—help take the guesswork out of handling chemicals. Agencies make clear that corrosive substances need separate storage, far from anything flammable or incompatible. Real-world lab audits almost always focus on chemical segregation, for good reason. You don’t mix acids with bases or flammable solvents unless you’re asking for a disaster.
Ventilation saves lives. A fume hood seems like overkill for a few milliliters, but inhaling harsh vapors leaves lasting damage. There are plenty of stories of seasoned chemists getting sloppy and paying with trips to occupational health. Storage cabinets designed for acids often come lined with protective coatings and offer enough airflow to keep vapor levels low.
The Checklist That Makes a Difference
Storing chemicals safely isn’t just about fancy gear, either. Labels matter, and so do routine checks. I’ve seen accidents just because a bottle got put back in the wrong spot and someone else picked it up, not knowing what they were holding. Keeping an up-to-date inventory and making sure containers stay labeled with purchase dates and hazard symbols cuts down on confusion.
Emergency response materials—neutralizing agents, spill kits, and fresh eye wash stations—belong close at hand, not buried in a storeroom. Practicing spill response helps, too. In my own experience, even a dry run gets people thinking more clearly when an accident actually happens.
Bringing It All Together
The bottom line is simple: chemicals like difluoroacetic acid won’t forgive sloppy habits. Using the right storage containers, keeping things organized, respecting health and safety rules, and treating storage seriously keeps people safe and the workspace in one piece. Anyone who’s dealt with a chemical scare knows those few extra steps aren’t a burden—they’re peace of mind.
More Than Just a Chemical Name
Difluoroacetic acid tends to show up in conversations among chemists and researchers who care about the details. Anyone who’s spent enough time weighing bottles in a lab has noticed how those tiny printouts and colored labels communicate numbers that matter: purity, grade, and the kind of confidence you can have in every drop. I’ve spent hours racking shelves and double-checking inventories, so the numbers and grades on bottles stopped being background noise a long time ago. They’re the signal you rely on, especially when stakes run high for synthesis, analytical work, or regulatory compliance.
Packing a Punch: Purity Levels Seen in Market
Most difluoroacetic acid on the market claims purity that falls between 98% and 99%. Those numbers don't come from thin air; labs and manufacturers work hard to keep contaminants at bay during production. A stray trace of water or an unexpected byproduct spells headaches later. Analytical chemists often check paperwork, look for gas chromatography data, and pay attention to the latest batch certificates. If 0.5% of something else sneaks in, some experiments land in the trash. Over time, the stories circulate—those times someone tried to save a buck only to lose weeks of data.
In practice, the actual purity you see lining the shelves owes a lot to the methods used in manufacture, whether that’s older fluorination techniques or modern continuous-flow reactors. If you skim through the catalogs of major chemical suppliers, 98% is the baseline for research grade, while 99% or higher sits in the premium bracket for tasks like pharmaceutical work or LC-MS reference standards.
Grade Labels: Decoding the Real-World Impact
Grade doesn’t just mean purity; it carries extra weight about how the chemical has been handled, tested, and packaged. I’ve seen the scramble that happens when someone grabs a reagent labeled ultra-pure or high-purity only to spot the warning sign: “For industrial use only.” Research grade reagents often go through extra rounds of testing, especially for trace metals, non-volatile residue, and water content. In offices and labs focused on synthesis or drug development, “analytical grade” means you can run sensitive tests without worrying that the acid holds hidden gremlins.
Difluoroacetic acid used in electronics or for cleaning can get away with lower purity, and the grade flags that: technical or industrial-grade acids might work for etching or cleaning lines but won’t fly for NMR prep or peptide synthesis. In my own time with contract research, analysts always asked for certificates, gravitating toward anything labeled “analysis grade” or “for synthesis.” The difference comes down to trust—the trust that what’s inside matches the label.
Impurities Still Lurk: Testing Out of the Shadows
Even with high-grade labels, impurities never completely disappear. You have to trust but verify. Modern tools like NMR, HPLC, and mass spectrometry catch phosphates, chlorides, or trace metals hiding out below the percent level. In drug and material science, the drive for consistency puts pressure on suppliers to provide batch-to-batch data. Years of lab work have taught me to request recent analysis instead of relying on catalog claims.
Fixing the Gaps: What’s Next for Sourcing
Market pressure for cleaner, better-documented difluoroacetic acid keeps growing. Labs hit bottlenecks with inconsistent reagent quality. One promising approach involves tighter supplier relationships, open sharing of lot-by-lot analysis, and closer scrutiny of delivery logistics to plug contamination risks. Advocates push for stronger regulations on labeling—so buyers don’t play guessing games with integrity or safety. As a scientist and someone who’s made those midnight emergency orders, I know the value of having straightforward, trustworthy information about reagents. It saves time, money, and sometimes your sanity in the lab.