Looking at Difluoromethanesulfonyl Chloride: The Journey, The Science, The Challenges
The Historical Path of Difluoromethanesulfonyl Chloride
The backstory behind difluoromethanesulfonyl chloride stretches across several decades of chemical exploration. It’s not the sort of compound that pops up in popular science, but those working on synthetic routes in organic chemistry have paid close attention to it for good reason. Early research in fluorinated compounds, kicked off by the mid-20th century chase for new refrigerants, agrochemicals, and pharmaceuticals, led chemists to poke at new ways of sticking tricky functional groups onto hydrocarbon backbones. This pursuit meant new tools, and difluoromethanesulfonyl chloride found a place in hard-to-reach corners of sulfonylation chemistry. Instead of broad sweeps of public attention, this compound became a focus in writings about improving selectivity, controlling reactivity, and, ultimately, pushing the boundaries on how fluorinated compounds could shape new molecules. I remember reading accounts from the late 1980s, where researchers grumbled about synthesis yields and practical handling headaches, yet still pressed on because the molecule filled a gap nothing else could.
Product Overview and Physical & Chemical Nature
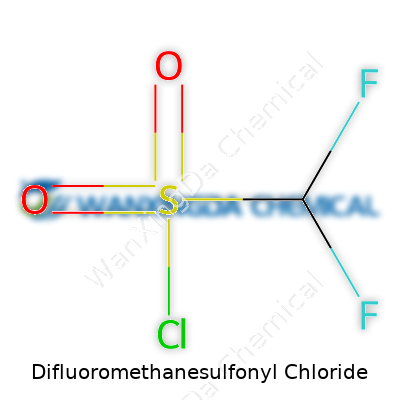
Difluoromethanesulfonyl chloride brings a sharp profile: colorless, volatile, and ready to catch the attention thanks to its pungency. This chemical is a bit like the quietly assertive person at a party—modest at a glance but unmistakable up close. The contained power in the molecule lies in how the difluoromethyl group can boost electron withdrawal. Anyone who’s spent time with sulfonyl chlorides knows they're dealing with a reactive electrophile, and tacking two fluorines onto the methyl kicks up that effect. This structure translates into distinct handling needs; it emits fumes, it reacts fast, and it doesn't tolerate moisture or open containers. Density, boiling point, smell—these numbers and sensory cues spell out a compound that pushes back if treated casually. It’s one thing reading about this reactivity, but until you crack open a fresh bottle, you don’t truly understand why chemical companies underline the importance of efficiency and care.
Technical Details and Labeling Realities
Bottles from reputable suppliers come stamped with hazard codes and precaution statements in language anyone working in a laboratory can recite by heart. Safety data highlight the irritant qualities, the environmental risks, and, most importantly, specific details about gloves, goggles, and ventilation. It’s not paranoia but experience that breeds these practices, as plenty of seasoned chemists can point to minor spills resulting in hours of lab remediation and paperwork. The blend of technical detail and plainspoken warnings in the documentation isn’t bureaucratic noise—it’s hard-earned advice. Some labels will mention the unique CAS number and standardized nomenclature, but for all their order, none of those codes capture the memory of a fume hood smelling sharply of acid gases.
Preparation, Reactions, and Tweaks
Difluoromethanesulfonyl chloride doesn’t just pop up in nature. Its synthesis starts from difluoromethane precursors, joining with sulfur trioxide or chlorosulfonic acid, kicking out water and building the sulfonyl group in the process. The preparation needs fine-tuned conditions; temperature and time matter, and the process churns out a stream of byproducts needing careful handling. Every seasoned chemist keeps a close eye on reaction kinetics, not just yield. Once in hand, the chloride group is wide open for nucleophilic attack—amines, alcohols, and various organometallics have all been tested, and the resulting sulfonamides or sulfonates carry the unique signature of the difluoromethyl group throughout subsequent reactions. Each new modification opens opportunities for small tweaks, shifting the way these molecules interact in biological testing or materials science.
Synonyms and Nomenclature: More Than Just a Name
Walk into different labs or poke through chemical catalogs and you’ll find a handful of aliases for the same compound. Whether it’s called difluoromethylsulfonyl chloride or carries a systematically numbered designation, the ring of familiarity only comes after seeing it in context. For those navigating literature or product orders, getting these names straight is more than technical accuracy—it’s about avoiding mistakes in procurement, confusion on paperwork, and wasted experimental hours. It’s a quiet reminder of the quirks built into organic chemistry, where two syllables can change the destination of a sample shipment or the outcome of a reaction.
Safety, Handling, and Standards in Practice
Lab safety isn’t just about rules; it’s about keeping scars off your hands and eyes, and about not turning a building’s vent system into a liability during a clumsy spill. Difluoromethanesulfonyl chloride sits high on the list of substances you don’t want to meet outside a well-functioning fume hood. Gloves, splash goggles, face shields—they all get used for a reason. Local exhaust systems and chemical waste bins stand ready for any drop or vapor that escapes. Personal experience teaches blame-free discipline. The memories of quickly neutralizing an unexpected splash with sodium bicarbonate, or waiting out the lingering fumes after a minor incident, never fade. It’s not about following a checklist, but about a lived respect for materials that can turn disastrous with one mistake. Standard practices documented by regulatory bodies exist because people learned the hard way what happens without them.
Applications: Where It Actually Matters
Few outside specialist circles realize where difluoromethanesulfonyl chloride ends up. In pharmaceuticals, it gets slotted in for building blocks or introducing the difluoromethanesulfonyl motif, which often changes biological properties in ways both subtle and profound. Agrochemicals pick it up to yield compounds that resist breakdown in harsh environments. For battery research, the fluorine atoms play a critical role, especially in electrolyte components for advanced battery cell technology. These applications might seem narrow, but in each niche, the compound fills a role nothing else quite matches. Each time a process engineer manages to make a cleaner reaction or a medicinal chemist finds an unexpected result, the molecule earns its keep.
Progress in Research and New Directions
The appeal of difluoromethanesulfonyl chloride has never rested idle—today’s research teams keep hunting for greener synthesis methods, ways to cut down on hazardous waste, and modifications that reduce byproduct formation. Academic and industrial labs alike keep investigating new applications, from medicinal chemistry projects aimed at fine-tuning drug candidates to innovative polymers durable enough for challenging conditions. Some of the most interesting experiments come from efforts to expand its role as a fluorinating or sulfonylating agent, hunting for selectivity that doesn't demand expensive or dangerous reagents. Investment follows success; government grants and private investments back up projects that promise safer, more sustainable chemical processes. This is how the rough-and-ready synthesis routines of the past gradually shift toward tomorrow's clean chemistry.
Toxicity Research and What It Means
The handling of difluoromethanesulfonyl chloride highlights the ongoing clash between practical chemistry and the health impacts of working with potent reagents. Lab animals exposed to sulfonyl chlorides commonly exhibit signs of respiratory distress, tissue irritation, and organ toxicity at higher doses. Technical papers have reported cellular damage and strong inflammatory responses; inhalation risks for humans can’t be dismissed or waved away by assuring oneself of proper technique. Protective measures do more than keep the workplace running—they safeguard those who spend their days around these molecules. Studies point to the importance of both personal protection and automated dosing or remote handling, especially as processes scale up. Better ventilation systems and the constant improvement of personal protective gear come out of these toxicological insights.
The Road Ahead: Future Prospects for Industry and Research
Looking forward, difluoromethanesulfonyl chloride won’t disappear from research benches or production plants. Its value in precision synthesis, especially when adding strong, electron-withdrawing groups to organic frameworks, gives it a secure position in medicinal chemistry, materials innovation, and specialty agrochemicals. The growing focus on sustainability turns up fresh challenges: cleaner synthesis protocols, recyclable waste management plans, and alternatives to the harshest processes involving this chemical. Advances in automation and reaction control promise safer environments, and every incremental improvement carries the potential for a broader impact—not just on the molecule’s niche applications, but on the shape of advanced chemical manufacturing as a whole. Experience and insight, not just theoretical knowledge, set the course for how it will shape the coming years of applied and academic research.
Chemistry That Gets Things Done
Chemistry shapes the world, though most folks never see the lab work behind everyday products. Difluoromethanesulfonyl chloride sits in the mix of those quiet but powerful reagents that don’t become household names. Instead, it does the dirty work at the molecule level, clearing a path for more advanced materials and medicines. Anyone who’s worked in a synthesis lab knows the value of a reagent that actually does what the paper says—and doesn’t blow up in your face. Difluoromethanesulfonyl chloride usually fits that bill.
Its main role lies in making organic fluorine compounds. In my own experience, nothing boosts a molecule’s stability, or helps it cross that tricky hurdle of metabolic resistance in drug discovery, like dropping a few fluorine atoms into the structure. Medicinal chemists constantly chase longer shelf life, potency, and precise behavior in the body. Adding a difluoromethylsulfonyl group, thanks to this reagent, opens avenues for designing those advanced pharmaceuticals. Not a week goes by in big pharma or a university lab where someone isn’t trying to push a drug candidate farther, to last longer or fend off liver enzymes a bit better.
Pushing Electronics Further
Electronics rely on incredibly stable materials, and a lot of that stability comes from well-placed fluorine atoms in polymers or specialty chemicals. Difluoromethanesulfonyl chloride gives researchers a tool to insert functional groups that help craft these important materials. For instance, folks designing next-gen battery electrolytes—those seeking greater capacity, longer life, and better safety—often turn to organofluorine chemistry. Strong, stable molecules built with parts from difluoromethanesulfonyl chloride enter these battery fluids, raising the bar for how far a single charge goes and how resilient a cell turns out after hundreds of cycles.
Working on a side project once, I remember trying to solve a problem with a stubbornly unstable electrolyte. Swapping out a structural group with a difluoromethanesulfonyl version finally gave us much better resistance to both heat and humidity. That subtle chemistry played a real-world role in moving the whole device from the bench to something engineers could actually use.
Protecting Functional Groups in Synthesis
Organic chemists face the recurring headache of keeping certain parts of a molecule safe during reactions. Difluoromethanesulfonyl chloride steps in as a protecting group agent in some cases, shielding specific sites from unwanted reactions while the rest of the chemistry runs its course. This can turn a frustrating, low-yielding process into something efficient and repeatable, saving both time and expensive starting materials.
I think back to a tough project where we spent months chasing a cleaner route to a target compound. Every shortcut involved selective protection, and reagents like this one often made the difference between an ugly mixture and a crisp, single product.
Weighing Potential and Risk
Difluoromethanesulfonyl chloride, like many good lab tools, comes with a need for careful handling. It reacts aggressively, and nobody wants a chemical burn or cloud of toxic by-products. Inexperienced hands can turn a standard reaction into a safety event, so proper training and modern ventilation aren’t just bureaucratic hurdles—they’re lifelines.
Green chemistry principles nudge us all to look for alternatives that do the same work with less danger. That effort’s ongoing, but for now, this compound sticks around in labs because it gets results in pharma and material science. As industry standards tighten, and as more sustainable options show up, the way we use—and replace—these strong reagents will keep evolving, the same way the chemistry itself always adjusts to fit new needs.
Most folks outside the lab haven’t heard of difluoromethanesulfonyl chloride. Chemically active and unforgiving if mishandled, this compound demands respect—not just from chemists but everyone around. Through the years, I’ve watched novice researchers get too comfortable with new chemicals. This attitude almost always runs into trouble. With reagents like difluoromethanesulfonyl chloride, it’s not just smart to be careful—it’s vital.
Hazards Lurking in Everyday Mistakes
Experience shows that minimizing accidents often comes down to habit. Chemicals known for aggressive reactivity, like difluoromethanesulfonyl chloride, react sharply with moisture and basic compounds. You splash even a drop on wet glassware, and that signature hiss and smoke mean toxic fumes fill the air. Once, a spill in an unventilated storage closet meant everyone in the building learned the meaning of “trace vapor exposure.” Lesson learned: drier, cleaner, and cooler pays off.
The risk of gas leaks is very real. Inhalation accidents turn up in academic publications and workplace incident logs. A small bottle break or a missing seal doesn't just trigger a cleanup—it can send an entire lab scrambling. The acute exposure symptoms reported—coughing, lung irritation, headache—line up with what we see in other strong sulfonyl chlorides. No one wants another case added to the record.
Temperature and Air: Not Just Details
I’ve met plenty of scientists intent on bending these storage rules. Room temperature may feel comfortable, but for volatile chemicals, even a few degrees can shift stability. A slight rise above normal room temp, and contents may slowly break down or the vapor pressure climbs. That makes a bigger difference in a storeroom than most people realize. Cold, dry storage—not freezing, not humid—truly makes both health and shelf life manageable.
No one wants corrosion on container lids, loose seals, or weird residues forming over time. That’s exactly what creeps in if humidity sneaks into storage or if the container sits out uncapped for even an hour during stock checks. Keep acids and bases clear; reactions will release choking fumes and heat, raising the hazard level inside the lab. Ask around, and you’ll hear more than enough stories about overheated vials bulging or popping when workers ignore these warnings.
Practical Steps: What Actually Helps
In some teams, each researcher personally checks expiration dates and does their own safety inspections. In my experience, group accountability serves better. Apps and stock lists haven’t replaced simple visual checks and a clear logbook. Good gloves, goggles, and splash-resistant coats end up being less for show and more for the inevitable bumps and spills. Fume hoods need to be running, not just present.
Transporting between buildings should mean keeping bottles upright, snug, and away from the bottom of a rickety bag. One careless jolt can crack glass and set off trouble. Upgrading to shatter-resistant containers cuts down on accidents; more than once I’ve seen a cheap plastic carrier prevent what could have become a hospital trip.
Room for Improvement
Institutions have improved by sending their new hires through hands-on chemical safety. By focusing on worst-case scenarios, labs adopt a mindset that puts safety before convenience. Sharing accident case reports, without hiding details, turns mistakes into learning opportunities. Some universities encourage quick reporting of near misses—no blame, just lessons shared. This culture saves careers, money, and lives.
Difluoromethanesulfonyl chloride is no different from any hazardous chemical: its dangers go down as care goes up. Storage and handling requirements aren’t just fussy rules or bureaucratic speedbumps—they keep labs safe, experiments reproducible, and researchers healthy, year after year.
No Room for Carelessness in the Lab
Step into any chemistry lab, and it’s easy to spot who takes safety seriously and who cuts corners until something goes wrong. Difluoromethanesulfonyl Chloride lands in the category of chemicals where you don’t want to roll the dice. This isn’t just another reagent you pour and forget about. It’s strong, reactive, and unforgiving to those who shrug off safety routines.
The Danger Is Not Overhyped
A compound with both a reactive sulfonyl chloride group and difluoromethyl, it doesn’t ask politely before causing trouble. Eye protection, gloves, and a fastidious lab coat become as necessary as oxygen. If someone has ever gotten even a drop of an acid chloride on their hand or near their face, they’ll tell you the pain isn’t subtle. Chemical burns from sulfonyl chlorides feel worse than simple acids. Splashes in the eyes can mean permanent injury.
Fumes drift off this reagent, especially when moisture creeps in. Inhalation burns the nose and airway. It’s the kind of chemical that makes a mask more than a formality. Respirators rated for acid gases, or at the very least a well-ventilated hood, stand between you and a trip to the emergency room. No one walks away from inhaling these fumes and talks about mild discomfort.
Solid Prep Isn’t Just for Show
Routine starts before cracking open the bottle. This means clear labeling, access to spill kits, and a game plan in case things get messy. Anyone handling Difluoromethanesulfonyl Chloride needs to scan the Safety Data Sheet, not just file it away. I once watched a well-trained chemist prep a reaction and stack all the right absorbent pads, wipes, and a neutralizing solution within reach. No one laughed—everyone understood the consequences of a spill.
Water mixes with this chemical to unleash a storm of hydrogen chloride gas. That stuff can blister skin and wreak havoc on eyes and lungs. No one wants to watch a fume hood fog up and realize too late they forgot to double-check the sash height or the airflow. I’ve seen lab mates run from an unexpected puff of gas—seconds lost dodging fumes mean more risk, not less.
The Right Way: Training and Tools
Having all the fancy gear in the world doesn’t replace hands-on training. I’ve seen safety videos by the dozen but nothing replaces the memory of your supervisor barking instructions as you measure out milliliters while gloved hands start to sweat. It matters that new chemists walk through spill drills. Labs practicing emergency stops and showers make a difference.
Storage isn’t just an afterthought. Heat and light ramp up pressure inside bottles and can compromise seals. Acid chlorides demand use of sealed containers kept out of reach of moisture and incompatible chemicals like bases and amines. There’s no shortcut to careful inventory and regular checks to keep things contained.
Culture Over Compliance
True safety grows out of more than a checklist. It comes from respecting what even small mistakes with strong chemicals can do. Labs where people look out for each other and call out lapses without fear of embarrassment walk away with fewer accidents and better stories. Handling Difluoromethanesulfonyl Chloride the right way means more than reading a label. It’s the difference between a smooth day at work and an ambulance ride you never forget.
Digging into the Details
People working in organic synthesis, battery research, or pharmaceutical labs spend a lot of time picking apart small details most folks overlook. Difluoromethanesulfonyl chloride stands out as one of those chemicals that keeps popping up in new studies. The tiniest impurity or a wrong bottle size can throw off weeks of work. Purity and packaging may sound like small stuff, but in this field, they're pivotal. Folks who spend their days in labs checking reaction yields know that.
Why Purity Comes First
Most research-grade difluoromethanesulfonyl chloride arrives at 97% purity or higher, meeting expectations for demanding syntheses. Highly pure versions cut down the risk of unwanted side reactions, which is important since nobody enjoys purifying a multistep synthesis gone awry. Experience across universities and industry tells anyone that skipping even a tiny impurity often costs more time than sourcing pure material in the first place. Some specialty suppliers bump purity to 98% or more, though high-end batches come at a premium. Anyone with experience running NMRs after a reaction knows the headache caused by impurities creeping into an isolation step.
Packaging Sizes: Practicality Over Flash
Not every research project uses chemicals by the kilo. Small bottles—ranging from 5 grams to 100 grams—see constant use in labs running exploratory reactions. Researchers always look for the sweet spot: enough to cover test runs, not so much that stock expires or becomes a safety headache. Large-scale synthesis groups and pilot plants lean toward 250 gram or 500 gram bottles, sometimes up to kilogram jugs for renewable energy and material science projects.
Safe and effective storage isn’t just a detail either. This stuff reacts if not sealed right. Most suppliers choose amber glass or PTFE-lined bottles to keep light and moisture away. The packaging isn't a mere afterthought; it keeps the chemical from decomposing or posing an unnecessary hazard for folks in the stockroom. Scientists make decisions every day that hinge on the simplest things: a tight cap, a correct label, an up-to-date batch. Anyone who’s had to track down the source of a mystery impurity knows small packaging choices make a big difference.
Why All This Matters
If scientists and engineers had endless budgets, purity and packaging would be less of a worry. But budgets aren’t like that. Access to high-purity chemicals, and a reasonable choice of packing sizes, keeps everyone nimble—especially smaller research outfits. Wasted material or compromised purity doesn't just cost money; it stalls science. That carries weight whether you’re working toward cleaner batteries, smarter drug design, or just a publishable synthesis.
Better Solutions Start with Better Standards
Smart procurement teams don’t cut corners on reporting purity specs or checking storage details. Shared knowledge goes a long way—open communication between supplier, stockroom, and researcher stops problems before they start. There's room to improve transparency. Public batch testing results, easy-access certificates of analysis, and real-time stock tracking could help everyone from postdocs to industry chemists.
Details sometimes sound boring, but nobody trying to make genuine progress takes them lightly. Chemistry keeps demanding the best from everyone—down to the bottle on the shelf.
Chemistry Isn’t for the Faint-Hearted
Working in chemical research and manufacturing has taught me there’s always more to a story than a product’s name or CAS number. Difluoromethanesulfonyl chloride is one of those tongue-twisters that shows up on lists for synthetic chemists and specialty chemical producers. It isn’t a chemical the average person finds in a local supply shop. Its applications tilt toward the dirty work of organic synthesis, pharmaceuticals, and maybe a few niche electronics or agrochemical processes. Every batch, every liter, matters, because consistency can mean the difference between a breakthrough and a failed synthesis.
Supply Chains and the Specialty Chemicals Dance
Industries working with such specialty compounds often wage a quiet battle against unpredictable supply chains. Bulk orders for difluoromethanesulfonyl chloride don’t move like flour or crude oil. The market depends on a small group of advanced chemical manufacturers, and sometimes even those suppliers source key precursors from overseas. If a client asks for several hundred kilos, it can kick off a negotiation not just about price or lead times, but about technical specs, regulatory paperwork, and shipping logistics. I’ve seen supply dry up overnight because of geopolitical shifts, or a regulatory change in environmental handling standards.
Production processes for fluorinated sulfonyl chlorides can get touchy. The workers must balance specialized equipment, strict purity requirements, and evolving safety standards. Some plants pivot between custom and bulk production based on changes in demand or seasonal fluctuations in other product lines. Nobody cranks out these chemicals like widgets. Quality control can get intense with reactive gases and aggressive reagents. If you’ve ever been part of a plant safety drill for halogen chemistry, you remember the sting in your nose and the way those chemicals demand respect.
Why Access Matters for Progress
Laboratories, both large and small, depend on reliable access to intermediates like difluoromethanesulfonyl chloride. If a supplier can’t confirm bulk availability, a project might stall for months. Scale-up teams chase grams, then kilograms, then tons, always knowing the finish line shifts depending on market and regulatory pressure. Missed deliveries push back not just launch dates, but livelihoods—scientists and production crews waiting for shipments that got caught at a customs checkpoint or a port backlog.
Custom orders sometimes offer a way out, but they add uncertainty and extra paperwork. Labs must certify usage, storage protocols, and end applications. Producers dig through client specs and confirm that what worked for an order last year still applies today. Delivering custom chemicals isn’t about showing off flexibility—it just means more layers in regulatory compliance and more staff hours.
Building a Responsible Path Forward
Finding solutions takes effort across the entire value chain. Strong supplier relationships go a long way. I’ve watched clients and manufacturers set up long-term contracts or collaborate on alternate sourcing, sometimes even backing joint research to tweak the production process or find replacement reagents. Digitized supply management tools help, giving a clearer sense of future needs so makers can plan and avoid surprises.
On the compliance side, open communication with local regulators and transparent paperwork smooths out bottlenecks. Companies that invest in air-handling and waste mitigation often keep their doors open longer, weathering storms that shutter less responsible outfits. There’s a real cost to chemicals that travel halfway around the world—and it isn’t just financial. Maintaining tight oversight, sustainable sourcing, and fair labor is the only way this industry moves forward. Difluoromethanesulfonyl chloride may not be a household name, but its story mirrors the whole sector: technical, sometimes risky, and always a collaboration.