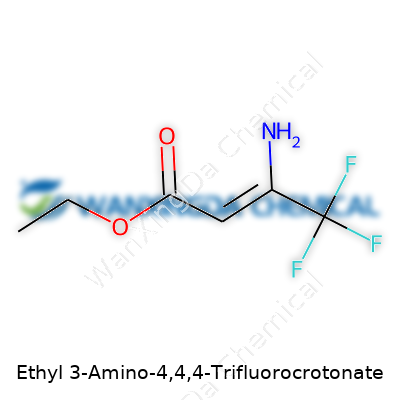
Ethyl 3-Amino-4,4,4-Trifluorocrotonate: Its Place in Modern Chemistry
Historical Development
Chemists have long chased after compounds that combine the flexibility of organic molecules with the punch of fluorine atoms. Ethyl 3-amino-4,4,4-trifluorocrotonate stepped into the spotlight as research moved from simple hydrocarbons toward functional fluorinated molecules. Looking back, early organofluorine chemistry aimed to harness fluorine’s unique properties, which go way beyond resisting rust or making non-stick pans. The quest for more stable, potent analogs of natural compounds led researchers down the pathway to building blocks like trifluorocrotonates. This push didn't just serve niche interests – it mirrored a bigger shift in chemical synthesis focused on boosting drug efficacy, creating agrochemicals with better performance, and building molecules with sharper electronic traits.
Product Overview
Ethyl 3-amino-4,4,4-trifluorocrotonate brings together several features synthetic chemists crave. At its core, you get a crotonate backbone, a simple structure, yet efficient for introduction into larger, more complex molecules. Tagging on an amino group amps up its value, opening doors to numerous follow-up reactions. This compound doesn’t just sit around in a catalog; it goes into the lab and finds its way into advanced pharmaceutical intermediates and modern agrochemical development programs. Fluorine’s presence on the tail end, tripling up to make the 4,4,4-trifluoro motif, grants it both chemical robustness and a certain unpredictability. That’s exactly the kind of character you like to have when you push scientific boundaries.
Physical & Chemical Properties
In the real world of flasks and fume hoods, Ethyl 3-amino-4,4,4-trifluorocrotonate stands out as a colorless to pale yellow liquid, matching what you expect from a mid-sized ester. Those fluorines aren’t just for show—they wield heavy influence on reactivity and stability. Boiling and melting ranges lean toward values typical of small ester molecules with a bit more volatility from the trifluoro group. This compound doesn’t play well with water; moisture can start to mess with its ester bond over time. With three fluorines hung at the end, you see a decent bump in molecular weight and a real impact on how it takes part in subsequent reactions, making it a strong electrophile in many transformations.
Technical Specifications & Labeling
For a working chemist, details matter. Purity usually runs above 95%, sometimes higher, depending on the synthesis and purification route. Labels in the lab show the CAS number and IUPAC name, but most bench chemists just call it “ethyl trifluoro crotonate amino.” These practical details keep safety data sheets nearby and help avoid confusion with other, similar fluorinated crotonates. Experienced researchers keep an eye out for storage instructions—the ester likes a cool, dry spot in an amber bottle, away from strong acids and bases that could trigger unwanted side reactions.
Preparation Method
Making this compound isn’t a one-step affair. Most common routes employ trifluoromethyl precursors, letting synthetic teams take advantage of nucleophilic addition onto an activated alkene. After building the crotonate core, the amino group finds its way onto the structure through amination reactions, which must be handled with care to dodge over-reaction or side-product formation. Organic solvents come into play, with meticulous work-up and purification through column chromatography or careful distillation. Yields track pretty well if reaction details get dialed in, but reproducibility demands both hands-on skill and a sharp eye for detail. For scale-up, robust reaction control reduces the risk of hazardous byproducts and helps limit environmental impact—a serious concern in any fluorine chemistry endeavor.
Chemical Reactions & Modifications
Chemists appreciate versatility, and this ester brings a useful toolkit to the bench. The double bond offers spots for further functionalization; you can push in nucleophiles or go for Michael-type additions. The amino group, if it’s not tied up with something else, opens up condensation reactions or provides a point to introduce various protecting groups, making it a flexible intermediate. Its ester group can be hydrolyzed or swapped out under the right conditions, allowing creation of acids or amides for a wide range of downstream products. The trifluoromethyl group, for all its electron-withdrawing power, transforms how the rest of the molecule behaves, letting scientists tune physical and biological activity in fine steps not possible with non-fluorinated analogs.
Synonyms & Product Names
Ethyl 3-amino-4,4,4-trifluorocrotonate isn’t short on aliases. It sometimes pops up as Ethyl (E)-3-amino-4,4,4-trifluorobut-2-enoate, reflecting the same E-stereochemistry. Some researchers shorthand it as “trifluorocrotonic acid ethyl ester, amino derivative,” or just refer to it by its systematic name when ordering for the lab. The proliferation of names doesn’t always help, but veteran chemists know the importance of checking structural details to avoid swapping in the wrong molecule.
Safety & Operational Standards
A careful approach pays off when dealing with any organofluorine. Ethyl 3-amino-4,4,4-trifluorocrotonate isn’t wildly toxic in small quantities, but exposure risks come into play during synthesis and handling. The ester’s vapors can irritate eyes and respiratory passages—standard fume hood practices apply, along with nitrile gloves to avoid skin contact. Disposal routes deserve just as much attention, since strong acids or bases can break it down to more hazardous fragments. Modern labs encourage close reading of safety data and enforce standards that cover not just storage and handling but also emergency spill management and accidental exposure.
Application Area
This compound earns its place in the toolbox of medicinal and agricultural chemists. The electron-withdrawing fluorines help tweak molecular recognition in drug design, raising metabolic stability or changing biological targeting profiles. Synthetic campaigns that need new analogs of active molecules often turn to fluorinated crotonates to break out of dead ends with common building blocks. In agricultural chemistry, the trifluoro group’s stubbornness against metabolic degradation means derivatives linger longer where they’re needed, cutting down on repeat applications and lowering total chemical load in the environment. Materials scientists working with fluorinated monomers have also played with this molecule, searching for new polymers or specialty plastics with tailored properties.
Research & Development
Academic labs and corporate R&D shops won’t stop searching for smarter, safer, more efficient fluorine chemistry. In my own experience, research groups continually tinker with reaction pathways, looking for greener solvents and catalytic systems to build this molecule more efficiently. Plenty of grant money now flows toward reactions that reduce by-products or minimize energy use, as the chemical enterprise faces pressure to clean up its act. Ongoing collaborations between universities and industry partners demonstrate the appetite for new derivatives—once a research group publishes a selective method for functionalizing the crotonate backbone, you can bet synthetic chemists everywhere leap to test its utility.
Toxicity Research
Despite its utility, the medical world doesn’t take the introduction of new fluorinated compounds lightly. Toxicity studies—ranging from cell cultures to animal models—play a big role in defining the boundaries of use. Ethyl 3-amino-4,4,4-trifluorocrotonate shares the cautious approach of all new building blocks: before moving up the development chain, researchers screen for acute and chronic toxicity, metabolic byproduct profiles, and bioaccumulation. Fluorinated organics, in particular, inspire extra scrutiny after issues with legacy compounds—think PFAS—taught everyone to watch for unintended environmental effects.
Future Prospects
As demands rise for pharmaceuticals that last longer and agricultural chemicals that hang on in the environment just long enough, fluorinated scaffolds make up the vanguard of molecular innovation. Ethyl 3-amino-4,4,4-trifluorocrotonate will see new transformations and applications, especially as synthetic tools keep evolving. There’s momentum behind more selective, sustainable methods, driven both by regulation and public pressure. In the lab, researchers seek to pair reactivity with better assessments of long-term impact. The balance between human ingenuity and chemical responsibility continues to shape how products like this enter and leave the marketplace. For those of us with a hand in research or teaching, the challenge stays real—build better, safer molecules while keeping one eye on their future in the world outside the lab.
Understanding the Backbone: Structure Speaks Volumes
Think of a molecule as a neighborhood where each atom brings its own story. With Ethyl 3-Amino-4,4,4-Trifluorocrotonate, chemists see more than just a string of letters. At its heart sits a backbone built off the crotonate family — a four-carbon chain with a double bond. This one stands out thanks to a trifluoromethyl group perched on the fourth carbon. In plain terms, that’s three fluorine atoms replacing hydrogens at one end, turning an otherwise ordinary stretch of carbon into something durable and quite reactive.
This molecule carries an ethyl group attached to the oxygen of its carboxylate end. That simple tail brings in a flexibility common to many pharmaceutical intermediates. What really spices things up: the amino group linked to the third carbon, adding a touch of basicity and promising easy participation in further reactions. Chemists often sketch it like this: EtO2C-CH=CH-CF3 with an NH2 attached at the third position.
Why Does Structure Matter?
Years of work in lab settings teach the same lesson — fluorine is no ordinary atom. Replacing hydrogen with fluorine doesn’t just “tweak” a molecule, it transforms its behavior. Drugs and agrochemicals built around trifluoromethyl groups can resist breakdown, pass through membranes more easily, and bond tighter to targets. This makes a molecule like Ethyl 3-Amino-4,4,4-Trifluorocrotonate more than chemistry for chemistry’s sake; it’s part of a wider toolkit driving new treatments, crop protectants, and materials.
The amino group adds versatility. While working with amino-functionalized molecules, I’ve found they open doors in peptide synthesis, dye manufacturing, and catalyst development. Some projects have demonstrated just how an NH2 at the right spot turns an inert carbon skeleton into a platform for almost endless possibilities. Chemists who keep an amino group close find themselves with more options for attaching new fragments or building complexity into a molecule.
Challenges and Responsible Handling
Any trifluoroalkyl compound demands respect. Fluorine’s famous for its strength and stability—traits that don’t always make disposal or degradation easy. I’ve seen firsthand how persistent some fluorinated compounds can be, so careful handling and smart disposal featuring neutralization or incineration stays crucial. In the push for innovation, these details matter just as much as the big breakthroughs.
Ethyl esters, thanks to their volatility, remind me to look out for laboratory air quality. Even small spills evaporate quickly and can irritate or cause headaches, so adequate ventilation and sealed storage helps keep my workbench comfortable. Education about risks saves more trouble than trying to fix a mess later.
Building Toward Safer and Smarter Chemistry
What draws many chemists to a molecule like this isn’t just utility — it’s the challenge of harnessing its potential while minimizing its environmental impact. New research keeps offering more sustainable fluorination techniques, milder reaction conditions, and greener solvents. By looking for ways to make and use fluorinated amino esters responsibly, the chemical community can push boundaries without leaving trouble behind.
Ethyl 3-Amino-4,4,4-Trifluorocrotonate captures some of the biggest conversations in applied chemistry: balancing power and responsibility, dreaming big but paying close attention to every detail. The molecule’s story says a lot about where the field has gone and where it still hopes to travel.
Folks who’ve worked around pharmaceutical labs or organic synthesis will know that Ethyl 3-Amino-4,4,4-Trifluorocrotonate isn’t one of those chemicals you run into at the grocery store. Still, it plays a surprising role in making things we rely on, often without even realizing it. The real value of this compound starts to shine the minute you step into the world of modern drug development and synthetic chemistry.
Unlocking the Building Blocks
Pharmaceutical chemistry thrives on invention. Scientists spend months or years piecing together molecules one fragment at a time, hunting for something that could treat a stubborn infection, ease chronic pain, or address a rare disease. Ethyl 3-Amino-4,4,4-Trifluorocrotonate fits right into this puzzle as a versatile building block. Its amino group at the third position and the strong electron-withdrawing trifluoromethyl group liven things up, opening doors to a variety of medicinal chemistries. You’d spot this compound in the synthesis of intermediates for active pharmaceutical ingredients. Whenever a new drug candidate rolls off a bench, there’s a real chance something like this compound played a part in its origin.
Packing the Punch with Trifluoromethyl Groups
Looking at the trifluoromethyl group in this molecule, chemists almost instinctively picture improved metabolic stability and better bioavailability. This kind of fluorine-rich structure resists being chewed up by enzymes in the liver, which leads to longer-lasting medicines. In drug design meetings, many researchers scout for ways to tuck in a trifluoromethyl group, hoping the drug will stick around in the body long enough to do its job. That’s where Ethyl 3-Amino-4,4,4-Trifluorocrotonate steps in. Its structure allows known transformations, such as nucleophilic addition or condensation, offering up tailored fragments for other molecules—especially when people want drugs to handle water solubility better or cross cell membranes more easily.
Polymer and Agrochemical Applications
Step outside pharmaceuticals, and the compound shows up in the design of specialty polymers and agrochemicals. In the polymer world, introducing fluorinated units can make the resulting materials chemically resistant and more durable. Surfaces that shed water or coatings that resist grime owe a lot to fluorinated chemistry. On the agrochemical side, modifying pesticide or herbicide molecules with molecular pieces from this compound can result in products that hold up better in sunlight and rain, sticking around on crops long enough to do their work. Real-life field tests, rather than just theory, prove these benefits out in practice.
Problems and Paths Forward
Of course, every brilliant tool comes with a catch. The global chemical industry faces sharper scrutiny about how persistent chemicals move through our environment and ecosystems. Even useful molecules like this one spark debate over safe manufacturing and responsible waste handling. Stricter regulations around fluorinated compounds urge labs to adopt greener chemistry routes and keep an eye on byproducts. Now, many researchers focus on improving efficiency and reducing the number of chemical steps so less waste and fewer resources get used up along the way.
Direct Experience in the Lab
Anyone who has handled reactive intermediates like this one will recognize the excitement and the pressure of getting transformations just right, especially as you scale up. One missed calculation, or a misjudged reagent, and your whole batch can fall apart. The care and training poured into handling such chemicals pay off in safer labs and more robust supply chains.
Few compounds pop up in modern chemistry labs with a name as bulky as Ethyl 3-Amino-4,4,4-Trifluorocrotonate. Behind the letters and numbers sits a clear, almost innocent-looking liquid—though folks working with it know there’s much more going on beneath the surface. Handling a chemical like this is one of those unsung tasks that keeps everything running safely.
Why Handling Matters
It’s tempting to treat every bottle on a shelf the same, but I’ve seen sloppy habits turn a simple experiment into an afternoon of cleanup. Ethyl 3-Amino-4,4,4-Trifluorocrotonate—let’s just call it “the trifluorovinyl ester” for short—has its quirks like any specialty chemical. You can’t let your guard down since exposure or degradation risks get real, real fast.
Knowing Your Storage
A lot of graduate students walk in the first day, toss reagents into a shared cabinet, and move on. They don’t always realize that some of these liquids demand more attention. This ester likes it cool, dark, and dry. Direct sunlight usually alters chemical stability, not just the trifluorovinyl ester but almost any compound sensitive to UV. I’ve seen containers yellow and cloud when left too near a bright window. Humidity is another silent enemy; even sealed bottles turn sticky when moisture sneaks in.
From long days in busy academic and pharma labs, I’ve learned to keep these containers in tightly sealed amber glass bottles, often in a designated fridge or chemical cabinet. It’s not about luxury—it’s about preventing slow reactions inside the bottle and stopping breakdown before you can use the compound. Even a sniff of air through a loose cap shortens shelf life. At larger scales, some folks purge their storage bottles with nitrogen or argon before sealing tight. Small steps, but they add years to your chemicals and reliability to your data.
Direct Handling Risks
Anyone who’s handled chemicals with reactive amine or ester groups knows the smell and the sudden bite on exposed skin. Nitrile gloves and eye protection come out long before unscrewing anything. I’ve seen colleagues with red, irritated skin from not wearing proper gloves, or headaches after neglecting the chemical fume hood. A few drops spilled on a bench or into a glove can linger for hours, making the rest of your day uncomfortable at best.
Beyond personal protection, clean working spaces matter just as much. Lab benches with last week’s spills or clutter make it far more likely to cross-contaminate, especially when dealing with small volumes. Every time I skipped a quick wipe-down before opening a bottle, I regretted it—sticky messes, mystery reactions, wasted product. Simple routine: inspect, clean, measure, and keep records. It’s not bureaucracy; it’s just the kind of discipline that pays off every time you avoid a lab mishap.
Room for Improvement
There’s often a gap between written safety rules and what happens in a real-world lab. Too many written procedures don’t survive busy days or tight deadlines. I’d push for more open discussions in the lab, regular walk-throughs, and routine checks on storage conditions. No one remembers every safety guideline by rote, but reminders and mentorship from the old hands in the lab keep everyone sharper and safer. Even a quick group review at the start of each semester saves headaches.
Ultimately, handling tricky chemicals never becomes so routine that you can forget about it. With the right storage, airtight seals, protective gear, and constant attention to your workspace, the risks drop. That keeps good science— and healthy researchers—moving forward.
Making Sense of the Chemical Maze
Ethyl 3-amino-4,4,4-trifluorocrotonate isn't a household name. Few people outside specialty labs ever hear about it, let alone handle it. As someone who's spent years poring over Material Safety Data Sheets and cleaning up spills in graduate school labs, I’ve learned that even the strangest-sounding compounds deserve a closer look for their risk profile. The name might sound niche, but it doesn't mean the hazards take a vacation. In a world where so many new chemicals pop up every year, knowing what you’re dealing with makes all the difference.
Why This Chemical Stands Out
This molecule carries a fluorine punch—three fluorine atoms on the same carbon tend to grab attention from toxicologists. While plenty of fluorinated compounds do end up in stable materials like Teflon, others pack real volatility or pose toxicity issues. From experience, anything with multiple fluorines and an amino group belongs on the “handle with respect” list.
Looking through publicly available data, researchers working with this chemical use gloves, goggles, and good ventilation. That’s not just out of habit—these practices help reduce the everyday risks. For reference, trifluorinated compounds sometimes act as respiratory or skin irritants and could trigger headaches or nausea if fumes seep out. Amino esters have their quirks, sometimes irritating eyes or causing allergic responses on the skin. Combining these traits, I can see why people don't take chances with casual handling.
Calling Out Real-World Risks
If you’ve spent time in labs, you know nobody likes guessing games when it comes to chemical hazards. The challenge with chemicals like ethyl 3-amino-4,4,4-trifluorocrotonate? There's not a huge pile of publicly cited studies laying out its toxicity in rats, mice, or fish. Still, good lab safety draws from chemical structure and experience. People often assume that if something is rare, it must be safe, or at least harmless in small quantities. In my own work, I saw that even low-profile reagents could cause real headaches if left out or spilled without care. Volatile organic compounds, for example, don’t always smell before they make you dizzy or give you a rash.
Many research protocols keep this chemical under fume hoods. That choice sends a message—there’s an expectation that fumes present an inhalation hazard. Gloves matter, too, because skin absorption could happen even if splashing seems unlikely at first. For disposal, nobody dumps leftovers down the sink. Instead, waste drums collect every drop, following hazardous organic waste protocols. The goal: no surprises, no unplanned exposure, no regrets the morning after.
Looking Ahead: Safer Habits, Smarter Choices
Solving the safety puzzle means starting with clear, honest communication. For any lab or production space considering this compound, robust safety briefings become the difference between things running smoothly and disaster. Companies should supply up-to-date training—people shouldn’t treat safety goggles as optional. Strong ventilation standards, enforced glove policies, ready access to eyewash stations, and proper storage all make this kind of chemistry manageable. If accident response isn’t practiced, people freeze and make mistakes when it counts most.
Streamlining access to toxicity data—where it exists—brings another layer of protection. When key studies or case reports come up, sharing those findings widely educates not just chemists, but also environmental health workers. In my own circles, swapping stories about near-misses convinces newcomers that a few minutes of prep outpaces a hospital visit every time. That mix of practical habits and open information gives the best shot at working with ethyl 3-amino-4,4,4-trifluorocrotonate without the drama.
Why Purity Actually Matters
The chemistry world doesn’t throw terms like “purity” around for fun—purity makes or breaks a reaction. Most folks in drug development and advanced synthesis rely on reagents with purity above 97%, which keeps side reactions from muddying the waters. I’ve seen what happens with lower-purity materials: batches crash, data doesn’t add up, and labs lose days figuring out where things went sideways. Every percent below high purity makes troubleshooting a guessing game. For Ethyl 3-Amino-4,4,4-Trifluorocrotonate, high purity matters even more, because a sneaky impurity can cut yield, introduce safety risks, or even compromise the function you’re after. Scientists in medicinal chemistry and materials work watch out for water, residual solvents, or leftover starting materials—these trip up sensitive reactions.
Customers expect the label to match the lab report. Analytical techniques—NMR, HPLC, and GC, for the most part—back up those numbers. It’s not about gold-plating for its own sake; it’s about running reactions that don’t bite back. Suppliers offering lower-purity batches might attract bargain hunters, but experienced chemists know what that shortcut costs down the line.
Different Purity Grades, Different Goals
Some suppliers list Ethyl 3-Amino-4,4,4-Trifluorocrotonate at a technical grade—usually 95–97% pure. This works for some applications, but most research-grade work leans toward 98% or higher. I remember trying to cut corners with a lower grade on a fluorinated analogue, only to spend a week cleaning up after a stubborn impurity. For regulated industries like pharma, anything less than high-purity risks regulatory headaches, product recalls, or plain-old failed batches.
Packaging: More Than Just a Container
You can judge a supplier by how they package their chemicals, especially the oddball ones. In my own lab days, I learned packaging counts as much as purity. Exposure to air, moisture, or light can wreck a whole shipment of sensitive ester compounds. Glass bottles are the staple for small to medium quantities—usually between 5 grams and 500 grams. Brown or amber glass protects from UV, especially for light-sensitive stuff. For larger orders, you’d see chemical-resistant plastics, like fluorinated HDPE, but never for products known to leach or react with containers.
Real-world chemists rarely need commercial drums of niche intermediates, so flexible packing—in 10-gram vials or 100-gram bottles—matters for cost and safety. Custom pack sizes show suppliers get what researchers need, rather than sending everyone bulk without a choice. Heavy-duty seals and secondary containment prevent leaks during transit. It’s basic lab safety: nobody wants to sign for a box soaked in unknown residue. Cold packs or dry ice keep the temperature under control if the compound won’t survive a hot warehouse.
Fixing the Gaps
Transparency stands out as the biggest thing missing from how manufacturers talk about purity and packaging. Detailed certificates and real shipping photos could cut the guesswork. In some cases, asking for a lot-specific analysis before buying solves headaches before they start. Lab results should travel with the bottle, not just sit in a database no customer sees. Communication with suppliers—sometimes a forgotten art—helps sharpen the match between what’s needed and what arrives.
If chemists speak up about pain points in purity or flawed packaging, suppliers can raise their game. The simple truth: clear info up front saves money, time, and reduces everything from lab waste to frustration. Ethyl 3-Amino-4,4,4-Trifluorocrotonate may be a specialized molecule, but these basics never go out of style.