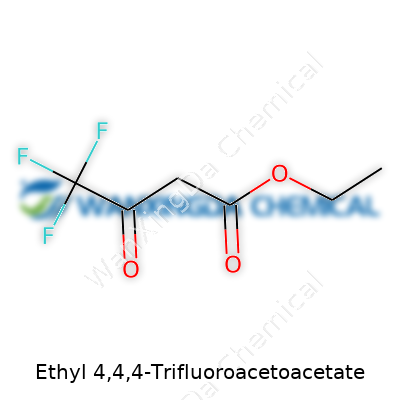
Ethyl 4,4,4-Trifluoroacetoacetate: A Closer Look at Its Role in Modern Chemistry
Tracing the Path from Discovery to Laboratory Mainstay
Ask any chemist with a few years under their belt, and they’ll likely recognize Ethyl 4,4,4-Trifluoroacetoacetate by its telltale structure or even a handful of its many trade synonyms. This compound traces its roots back to the growth of fluorinated chemistry in the twentieth century, when researchers realized the striking impact that trifluoromethyl groups could deliver in both stability and reactivity. Decades ago, as the appetite for organofluorine compounds in pharmaceuticals and agrochemicals picked up, lab benches started to see more bottles of this versatile building block. Its emergence didn’t follow a linear story, but rather a scatter of research efforts converging as the need grew for compounds that could deliver performance—and sometimes unpredictability—in both academic and industrial research.
The Traits That Define It
This compound feels almost emblematic of how a simple change—adding a trifluoromethyl group—can transform an acetoacetate. The pale liquid serves as proof. With its faint but unmistakable fruity smell, Ethyl 4,4,4-Trifluoroacetoacetate walks a fine line: Solid at cooler storage, fluid as temperatures inch up. It sports a melting point just below room conditions, and as for boiling, it doesn’t need much encouragement from a heating mantle before turning to vapor. Its fluorinated tail brings a distinct density and less common solubility behavior. Unlike less interesting esters, it loves mixing with organic solvents, but it largely shrugs off water. The trifluoromethyl group also makes it more resistant during reaction conditions that tend to chew up or modify other acetoacetates. Such quirks aren’t just trivia—they dictate how chemists think about storing, handling, and deploying this tool.
A Closer Look at the Bottle Label
Many might not know it by its full formal name but instead as ethyl trifluoroacetoacetate, trifluoroethyl acetoacetate, or simply as its abbreviation—ETFAA—depending on the region or application. Chemistry catalogs frequently group it under acetoacetate esters, with CAS numbers and chemical codes playing supporting roles for procurement and research. These details pop up in academic writing, patents, and batch records, making even the small details like labeling or batch IDs surprisingly important to traceability.
Painting a Picture of How It's Made
Lab veterans know this compound rarely arrives without some fuss in production. The trifluoroacetoacetate skeleton generally emerges from reaction sequences involving ethyl acetoacetate and trifluoroacetic anhydride, or sometimes through controlled fluorination of other acetoacetate precursors. The process demands care. Control of moisture, reaction timing, and work-up conditions plays a starring role. Even a minor misstep in purification can flood the product with byproducts that alter its behavior in downstream reactions. As someone who has chased purity through multiple distillations, it’s easy to appreciate where the art and science blend together in these preparations.
Where the Chemistry Gets Interesting
Let’s talk reactivity. Unlike basic esters, Ethyl 4,4,4-Trifluoroacetoacetate draws its power from both its active methylene group and the electron-hungry trifluoromethyl tail. These features enable classic transformations: alkylation, condensation, and cyclization all come to mind. For example, it often acts as a substrate for Knoevenagel condensations or serves in the synthesis of heterocyclic rings that form the backbone of many drug molecules. The electron-withdrawing trifluoromethyl plays tricks with acidity, making the methylene hydrogens easier to pluck off with mild bases. That adjustment transforms possibilities in carbon-carbon bond-forming reactions, offering routes that simply aren’t accessible with non-fluorinated cousins.
No One-Size-Fits-All in Safety & Operations
Anyone who’s ever cracked open a bottle of this compound learns quickly to respect its volatility and the hazards of inhalation. The trifluoromethyl ester isn’t the most menacing chemical on the bench, but it can irritate eyes and airways. Good ventilation, gloves, and eye protection become habits rather than afterthoughts. Storage away from heat or open flames makes a difference, especially given the risk of slow decomposition or the production of acidic fumes under the wrong conditions. Waste disposal also deserves more than a cursory glance—fluorinated organics linger in the environment, and so adherence to local waste protocols goes beyond just “following the rules” and moves into responsibility for environmental health.
Looking at Where It Ends Up
A strong suit of Ethyl 4,4,4-Trifluoroacetoacetate lies in research—particularly at the intersection of fine chemicals and life sciences. Medicinal chemists value its contribution as a building block for molecules with metabolic stability and pharmacokinetic promise. Agrochemical developers reach for it to modify the properties and persistence of active ingredients used on crops. Beyond these main pillars, it finds minor but significant appearances in the development of imaging agents and specialty polymers. I’ve personally watched colleagues wrestle with synthesis campaigns where this reagent made or broke success—either by granting just the right structure at the right moment or by stubbornly resisting transformation until conditions were just right. Its versatility keeps it in the synthetic toolbox, even as new fluorination technologies evolve.
Curiosity Fuels New Discoveries
With so many routes for modification, it’s no surprise that research articles featuring this compound keep coming. Scientists keep pushing for greener, more efficient synthetic pathways, both for the parent molecule and the diverse suite of derivatives made from it. Many focus on catalytic systems that lower the activation energy or improve selectivity, reducing both cost and environmental footprint. Researchers in pharmaceutical labs explore new ways to link or transform the trifluoroacetoacetate scaffold, chasing properties that might tune metabolism, absorption, or biological activity. These aren’t theoretical explorations either—new drugs and advanced materials often draw a straight line back to experiments with this compound many steps earlier.
Understanding Toxicity: Both Old Problems and New Insights
No discussion feels complete without a look at toxicity. Fluorinated organics always spark questions about bioaccumulation and breakdown. Past animal studies on structurally related esters show low acute toxicity but leave open concerns about long-term exposure or the behavior of breakdown products. Many researchers continue to scrutinize downstream metabolites and trace residues, especially since regulators grow more insistent on understanding environmental persistence. Safe handling and informed risk assessment ought to go hand in hand with the ingenuity that drives research forward.
What Comes Next?
The field keeps evolving. Sustainability and safety pressures push chemists to discover new routes to make and use fluorinated esters with less waste and lower energy input. Analytical advances allow for better tracking of residues in both lab and field work, opening doors for safer applications and tighter compliance with ever-shifting regulations. As new industries pick up on the value of fluorinated scaffolds—from medical diagnostics to novel polymers—the range of uses for Ethyl 4,4,4-Trifluoroacetoacetate won’t shrink. Every new application draws on decades of hard-earned lessons, underlining both the promise and responsibility that comes with handling such specialized chemistry.
Understanding the Role of Ethyl 4,4,4-Trifluoroacetoacetate
Ethyl 4,4,4-Trifluoroacetoacetate is one of those compounds you don’t hear about unless you spend a lot of your day thinking about molecules, but it quietly holds a pretty important spot in research labs and pharmaceutical companies. If you’ve ever sat down with a chemist at the bench, they’ll tell you: this compound comes up a lot during the hunt for new medicines. Its value springs from a pretty unique structure—those three fluorine atoms tacked onto one end change the game for how other chemicals can react with it.
The Heart of Drug Development
If you talk with folks working on new drug candidates, you’ll hear about the importance of “building blocks.” Ethyl 4,4,4-Trifluoroacetoacetate is one of these, giving chemists a head start toward constructing more complex molecules. The trifluoromethyl group isn’t just a chemical quirk — it delivers real impact. Drugs containing fluorine often stick around longer in the body and sometimes even work more effectively, all thanks to a few well-placed fluorine atoms.
New painkillers, antibiotics, and heart medications frequently trace their roots back to reactions involving molecules like this one. What stands out to me is the way trifluoromethylated structures have allowed scientists to fine-tune how drugs behave, leading to fewer side effects or more targeted treatment. Once you dive into the academic research, there’s a clear trend: more than a third of all pharmaceuticals approved in the last decade include at least one fluorinated group, according to multiple pharmaceutical industry reviews.
Synthetic chemists use Ethyl 4,4,4-Trifluoroacetoacetate in reactions designed to build up more complicated frameworks, adding important pieces to the puzzle. It’s versatile, offering a clean way to introduce fluorine atoms into new compounds, which isn’t a trivial feat. Ask anyone who’s spent too long with a reaction that just wouldn’t cooperate—having a reliable reagent makes all the difference.
Beyond Pharmaceuticals
While it’s got a big reputation in drug discovery, Ethyl 4,4,4-Trifluoroacetoacetate has found work in other arenas too. Agrochemical researchers value it for the same reason—those fluorine atoms improve stability and performance in pesticides and herbicides. This means lower dose requirements and, in some cases, less environmental persistence, which is nothing to scoff at if you’ve spent any time worrying about run-off and ecosystem health.
A lot of specialty materials depend on molecules like this to deliver their performance. If a researcher wants to develop a new plastic or coating that holds up in harsh environments, a trifluoromethyl group often brings the durability needed.
Responsible Use and Future Directions
There’s no denying these fluorinated chemicals come with baggage. Last year, several publications flagged concerns over fluorinated pollutants in manufacturing waste, and regulators continue to review safety profiles. Responsible sourcing and new purification techniques have helped, but chemists need to keep one eye on the safety sheets and another on developing greener alternatives.
Institutes and companies now pour resources into both improving synthesis and managing environmental impact. This dual track means future generations of molecules may offer the benefits without the baggage. Green chemistry teaches that innovation doesn’t stop at discovery; it keeps going until everyone benefits.
Ethyl 4,4,4-Trifluoroacetoacetate holds its place at the intersection of scientific promise and responsible practice. Its story isn’t just about better drugs or crops, but about pushing chemistry — and the people who practice it — into a more thoughtful future.
Understanding the Chemical Formula
Ethyl 4,4,4-trifluoroacetoacetate draws the attention of anyone who’s spent time hunched over lab benches deciphering reagent lists or scouring patent databases for synthetic shortcuts. The chemical formula, C6H7F3O3, is a string of letters and numbers that actually tells a story—a molecule packed with character. The three fluorine atoms, lined up at the fourth position, give it away as something special in the world of organic chemistry. The presence of both ester and keto functional groups makes this compound a handy building block. You see it pop up often in the synthesis of fluorinated pharmaceuticals and agrochemicals. There’s an art to unraveling formulas, and this one shows off how substituting just a few atoms can open doors to a whole new catalog of reactions.
Why Details Like Chemical Formulas Still Matter
Some might brush past the formulas, but anyone who’s tried to replicate a published synthesis knows it’s the details that trip you up or let you move forward. I’ve run into the headaches caused by vague ingredients or ambiguous names. It’s not just about formality—it’s about trust. The fact remains: if a synthesis calls for C6H7F3O3 and you grab the wrong flask, your months of work might end with a useless slurry at the bottom of a round-bottom flask. This risk only grows when companies scale up for production, where a single letter difference has real financial and safety consequences.
Everyday Impacts of Precision in Chemistry
The molecules we put together in the lab don't stay behind closed doors. They wind up in fields, clinics, and sometimes kitchens. Fluorinated compounds like this one are prized because they deliver unique activity; the trifluoromethyl group brings stability and shifts boiling points or reactivity, which the pharmaceutical world loves for making drugs that hang around in the body and break down slowly. Knowing exactly what you’re working with means those benefits actually make it to patients or crops without unexpected side effects or recalls.
Improving Accuracy and Knowledge Sharing
Mistakes happen when communication falters. During my grad school days, I saw how an error in a single digit within a chemical formula could stall research for weeks. This drove home the point: education in chemistry can’t just skim through nomenclature or stop at the first pass. Students should regularly practice breaking down names like “ethyl 4,4,4-trifluoroacetoacetate” into formulas and structures. Working chemists benefit from using reliable digital tools that cross-check structures and names, like ChemDraw or PubChem databases, which help prevent costly errors.
Chemistry’s Broader Responsibility
Chemists play an ongoing role in keeping information correct and accessible. With more people seeking answers online—students, researchers, professionals—verified sources matter more than ever. Sharing chemical formulas isn’t dry recitation; it’s about building a foundation others can trust. Strong technical accuracy and transparency drive everything forward, whether it's the next crop protection molecule, a cancer therapy, or just saving someone from a day ruined by a botched experiment.
Chemical Formula of Ethyl 4,4,4-Trifluoroacetoacetate: C6H7F3O3The Realities Behind a Fancy Name
Ethyl 4,4,4-Trifluoroacetoacetate sounds like a chemical straight out of a high-level textbook, but in the lab, it’s just another tool—a tool with bite. I’ve worked in university research labs where people learn not to underestimate chemicals like this. You see the safety goggles, the white coats, and might think you’re already doing enough. Yet, one slip with a compound like this can land someone in the emergency room or damage sensitive instruments. So what’s actually at stake?
Direct Routes of Exposure Matter
Every chemist learns that no matter how careful you feel, skin contact remains the most common mistake. Ethyl 4,4,4-Trifluoroacetoacetate burns on contact, and even a splash on uncovered wrists leaves a reminder for weeks. Splash-proof goggles don’t make you invincible, but regular glasses don’t cut it either. Full face shields offer that extra peace of mind after witnessing a seasoned researcher forget to close a vial and get a faceful of fumes. It’s never just about minimizing discomfort—the compound can irritate the lungs, eyes, and skin, which means solid gloves, lab coats with long sleeves, and closed shoes belong in the conversation every time this bottle leaves the shelf.
Ventilation: More Than Just a Suggestion
A lot of people ignore the power of a proper fume hood until they start coughing after an experiment goes sideways. Ethyl 4,4,4-Trifluoroacetoacetate doesn’t look menacing, but its vapors can hit hard. Standard practice in our lab has always been to move all transfer and measurement tasks inside a working fume hood. That’s not just a habit—it’s based on years of watching near-misses turn into cautionary tales. Airflow matters, and a working fume hood reduces the risk of breathing in anything dangerous. Keeping the sash low and the workspace uncluttered feels tedious, but that routine saves headaches down the line.
Spill Response: Fast Hands, Smart Mind
Staring at a spreading puddle after a dropped flask, adrenaline spikes. It’s natural to panic, but quick, organized response wins the day. We learned to keep absorbent pads and proper neutralizers nearby—no one wants to scramble for materials while a hazardous spill seeps under the benchtop. Small spills prompt glove changes and careful disposal, never shortcutting the waste process. Larger spills might mean evacuating everyone; it doesn’t take much to create hazardous fumes in a tight space. I once saw a graduate student wipe down a spill with standard tissues, only to find their hands breaking out in red welts an hour later.
Labeling and Storage: Preventing Accidental Mishaps
It’s easy to underestimate organization’s value until fatigue tempts you to grab the nearest clear bottle. Ethyl 4,4,4-Trifluoroacetoacetate usually gets stored in tightly sealed containers, away from heat or open flames. Materials like this don’t take kindly to sunlight or damp, so chemical storage cabinets provide that critical buffer. Labeling saves lives; I can’t count how many times I’ve watched colleagues avoid near-disasters by double-checking those big, simple hazard labels on every bottle.
Training and Checks: The Foundation of Safety
People make mistakes, but good training turns those mistakes into stories instead of disasters. At my university, no one touched a new chemical until they ran through the safety sheets and performed a mock dry run. Keeping up with safety updates turns out to be just as important as reading the latest research paper. Regular refreshers keep bad habits from creeping in, and they’ve helped our lab avoid serious accidents.
Ethyl 4,4,4-Trifluoroacetoacetate demands respect. Reminding yourself of risks and staying sharp with protocols builds an environment where the focus stays on discovery, not damage control. Keeping safety grounded in real routine—gloves, goggles, hoods, labels, and backup plans—lets us get the work done and head home safe every day.
The Role of CAS Numbers in Chemistry
In labs and warehouses, chemicals with long, complex names stack up side by side. Trying to keep track of them gets confusing fast. Ethyl 4,4,4-Trifluoroacetoacetate is a mouthful on its own, but it’s easy to see how mistakes happen when chemicals are traded, transported, or stored. That’s where CAS numbers cut through the confusion. These unique strings of digits come from the Chemical Abstracts Service, and they give every substance a sort of fingerprint. For Ethyl 4,4,4-Trifluoroacetoacetate, the CAS number is 372-30-5.
Why Numbers Cut Down on Risk
Accurate identification saves lives and money. I’ve seen mix-ups in research and industry when only chemical names were marked on containers. A similar-sounding name, a smudge on a label—mistakes get dangerous when you’re dealing with potent compounds. With CAS numbers, there’s no debate. If a barrel holds something labeled 372-30-5, people across continents can quickly look it up and confirm exactly what’s inside. This matters in hazardous material response, shipping, and regulatory reporting.
Transparency Supports Science
Ethyl 4,4,4-Trifluoroacetoacetate plays a role in research, synthesis, and even medicine. Scientists rely on certainty. When I worked in a lab, experiments often hinged on molecules like this—alter one small group on the structure, and the results change dramatically. Documentation had to be clear. CAS numbers let journals, producers, and researchers keep records everyone can check, compare, and trust.
The Cost of Error
Every time a chemist or logistics worker catches a slip thanks to CAS numbers, they dodge wasted hours and potential harm. Misidentified compounds can ruin product batches, lead to environmental spills, or trigger cross-contamination that’s sometimes invisible until much later. Cleaning up these mistakes proves both expensive and dangerous. Sticking to unambiguous codes helps prevent such problems before they start.
Pushing for Consistency
Despite the value of universal identifiers, not everyone remembers to use them all the time. Smaller labs and companies without strict checks sometimes rely on habit or shorthand. Larger players usually bake CAS numbers into their systems, but gaps remain. Consistent use, even in short email exchanges or quick notes, could weed out many mistakes. Pushing industry and researchers toward always using these numbers increases transparency and safety. Education goes a long way—students who learn with CAS numbers grow into professionals who share that discipline.
Building a Culture of Precision
Routine makes all the difference. It’s not just about ticking boxes; it’s about supporting a culture where everyone puts safety and clarity first. For something as widely used as Ethyl 4,4,4-Trifluoroacetoacetate, relying on a clear system like CAS numbering doesn’t only serve compliance requirements. It makes sure everyone is on the same page, reducing the odds of costly, avoidable errors. Bringing precision to everyday routines shapes better science, safer workplaces, and more reliable supply chains.
Getting Real About Chemical Storage
I’ve spent a lot of time around labs, beakers, big drums with warning stickers—places where everyone knows that safe storage is more than a suggestion. It’s how you keep work rolling and people out of trouble. Storing ethyl 4,4,4-trifluoroacetoacetate proves this point. One small mistake can create difficulties for the crew and risks for the surrounding space.
Why This Chemical Deserves Respect
This compound has found its way into research labs and production runs for its usefulness in making other chemicals, especially those with a complex structure. But its trifluoro group means it attracts attention for its volatility and reactivity. Failing to consider what it interacts with or how easily the fumes might escape spells trouble.
Keep it away from heat, direct sunlight, sparks, and open flame. Even simple tasks—like placing a bottle too close to a heat source—can turn routine work into a hazard. Stories circulate about bottles swelling or springing leaks after being left in the wrong spot. Using dedicated, ventilated cabinets and never improvising with old kitchen fridges limits those risks. Chemical-resistant shelving and spark-proof refrigeration offer peace of mind in a way plywood shelving never will.
Moisture and Contamination: Traps to Sidestep
Moisture sneaks into containers left open. You might not notice right away, but months later, you’ll find yellowed liquid or odd smells pouring out when you need it most. These changes chip away at reliability and add steps to your work. Flushing out humidity begins with solid closures and silica gel packets tucked inside storage cabinets—not in the drum itself. Use original containers or bottles made for hazardous liquids, clearly labeled with hazard warnings in bold.
Knowledge and Good Habits Save the Day
Old-timers will tell you—don’t rely on luck. I remember hearing stories in the break room about spills and ruined research because someone stored a solvent on the same shelf as a powerful oxidizer. If everyone checks compatibility charts and keeps acids, bases, and oxidizers in separate areas, you avoid mixing chemicals that trigger dangerous reactions.
The air matters, too. Work in spaces with ventilation that vents outdoors. This means local exhaust fans and fume hoods for anything that can evaporate into the workspace. It keeps fumes out of noses and lungs, which keeps people healthy and lets you focus on the task, not the smell.
Safe Storage Is a Practice, Not a Checklist
What always stands out: labeling matters. Every bottle should scream its contents and the hazards they bring. Faded labels invite mistakes, especially when staff change or memories blur. Keep a log of how much sits in storage and watch for expiration. Run through regular checks for corrosion, drips, or pressure building up in sealed bottles.
What works for me: treat every chemical like it’s the one that could go wrong. Enforce good habits, ask questions if any storage situation looks off, and create a culture where people aren’t afraid to double-check. This attitude gets results—a safer lab, a happier team, less stress, and uninterrupted research. For something as reactive as ethyl 4,4,4-trifluoroacetoacetate, these steps don’t just help. They make all the difference.