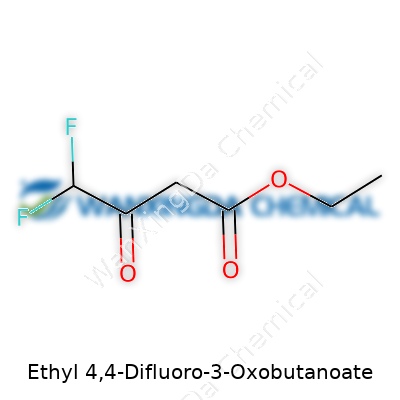
Ethyl 4,4-Difluoro-3-Oxobutanoate: Illuminating Its Place in Modern Chemistry
Historical Development
Going back, the roots of ethyl 4,4-difluoro-3-oxobutanoate trace into the ever-advancing era of organofluorine chemistry. The late 20th century brought a surge in interest around fluorinated compounds, largely because of the growing pharmaceutical industry's demand for metabolic stability and bioisosteric replacements. The difluoromethylene group, introduced into various skeletons, was seen as a ticket to novel biological activity and patents. Researchers learned that modifying ketones with fluorine could dramatically alter reactivity, stability, and utility. That tinkering led to a wave of specialized building blocks, among which ethyl 4,4-difluoro-3-oxobutanoate stood out, blending the benefits of a diketone with the double-whammy of fluorine atoms. Over time, this molecule shifted from a curiosity to a valued cornerstone in research toolkits.
Product Overview
Ethyl 4,4-difluoro-3-oxobutanoate doesn't attract much attention outside synthetic labs, but within those walls, it holds a quiet importance. It sits in the sweet spot between accessible and reactive—a balance that appeals to researchers. Whether building out a new medicinal scaffold or hunting for an efficient route to a tricky intermediate, this ester brings options. Its formula, simple at a glance, hides a layer of reactivity beneath the surface. Chemists looking to create fluorinated analogs of established drugs or to explore uncharted territory for agrochemicals find themselves returning to this molecule because it offers reliability in transformations without excessive fuss or unforeseen side reactions.
Physical & Chemical Properties
Examining ethyl 4,4-difluoro-3-oxobutanoate up close, its pale color and modest odor leave little hint of the power it packs. As a liquid at room temperature, it pours easily and blends into standard laboratory workflows. The real intrigue lies in its structure: the electron-withdrawing difluoromethyl group tugs at the electron cloud, shifting tautomeric balance and amplifying the acidity of nearby hydrogens. The carbonyl stretches, unencumbered, make for easy reading by IR spectroscopy, and NMR signals set apart the CF2 moiety for clear identification. Its volatility demands care, but that same trait helps make purification by distillation straightforward, which matters when turnaround speed counts in lab work.
Technical Specifications & Labeling
Chemists eye the CAS registry number and structure, but those details matter less to day-to-day use than reliability and clear handling advice. The labeling on bottles tends to point out core hazards: high flammability, moderate toxicity, and strong reactivity with bases and nucleophiles. Flash point, boiling point, and storage temperature—practically relevant figures—often guide storage decisions more than a laundry list of purity grades. What's essential is that grad students or postdocs reaching for the bottle trust what's inside and know precisely what to expect—one less unknown in a process often riddled with surprises.
Preparation Method
Preparation of ethyl 4,4-difluoro-3-oxobutanoate pulls together key skills from organic synthesis. Starting with commonly available esters, careful introduction of difluorocarbene or a difluorinated acyl source under controlled conditions gets the skeleton in place. The trick lies in managing the exotherms and avoiding over-fluorination—a misstep can produce more heavily substituted byproducts and add headaches to purification. Experienced chemists quickly learn the importance of incremental additions and consistent temperature control. For labs with strong safety cultures, standard glassware and fume hoods suffice, keeping risk manageable and scale adjustable.
Chemical Reactions & Modifications
The beauty of ethyl 4,4-difluoro-3-oxobutanoate emerges in its reactions. Tapping into its active methylene position, chemists run alkylation, condensation, and reduction reactions, sometimes stringing together one-pot transformations that speed up synthetic campaigns. The difluoro group both blocks unwanted side reactions and steers selectivity, helping to carve chemical space unavailable with non-fluorinated analogs. In the rush to build small-molecule drug candidates, this backbone feeds into Hantzsch syntheses, Michael acceptor chemistry, and β-ketoester condensations. The fluorines make it possible to reach products with increased metabolic stability, which pharmaceutical teams crave for new lead compounds.
Synonyms & Product Names
Search for this molecule, and the chemical literature throws up a handful of aliases—ethyl 4,4-difluoroacetoacetate pops up most regularly. That tongue-twister might confuse anyone outside the field, but it simplifies communication among chemists. Synonyms can reflect the various routes to the molecule or slight quirks in regional naming conventions, though the structure remains the same. A few catalog codes come and go depending on the supplier, but anyone scanning a bottle wants to match up the key functional groups and fluorine count right away. In practice, the conversation around it stays direct and focused: clear names, clear intent, no room for error.
Safety & Operational Standards
Lab stories often begin with a cavalier attitude and end with a lesson in caution—ethyl 4,4-difluoro-3-oxobutanoate enforces this lesson every time. Those fluorine atoms raise volatility and bring more severe health risks than more familiar, non-halogenated analogs. Skin and eye contact cause irritation, and inhalation risk looms thanks to that volatility. Standard PPE—gloves, goggles, and lab coats—are minimum requirements, but real diligence comes from disciplined fume hood use and careful waste handling. Spills turn into headaches fast, both for safety officers and colleagues sharing tight bench space. The culture around handling reflects long-term improvements in organizational safety: clear training, regular refreshers, and visible reminders about risks born from real-life incidents, not just safety sheets.
Application Area
Though no household name, ethyl 4,4-difluoro-3-oxobutanoate pops up in fields that touch almost every life. Medicinal chemistry labs reach for it to build analogs of drugs for everything from cancer to infection. Agrochemical research leans on it for making pesticides and herbicide leads with better resistance to soil bacteria. Its structure opens pathways to fluorinated amino acids that land in specialty food science and peptide chemistry as well. Every reaction with it offers a shot at a molecule that sticks around longer in microbes or humans, dodges metabolic breakdown, and brings new biological properties. In my time in multi-disciplinary research, seeing how a simple building block could ripple into something with real-world impact—sometimes showing up years later as a part of a new medication—never lost its thrill. This ripple effect makes even the most routine compound worth close attention.
Research & Development
Research with ethyl 4,4-difluoro-3-oxobutanoate fuels ongoing quests to understand both chemical mechanisms and downstream biological impact. Collaborative projects thrive on building blocks like this, where cross-functional teams can try new routes to active pharmaceutical ingredients, run screens on fresh analogs, and revisit optimization campaigns with fresh eyes. Analytical chemists work to refine detection and quantification methods, given the ubiquity of fluorinated fragments in mass spectrometry workflows. Development trends point toward greener routes—fewer harsh reagents, more efficient yields—and better scalability, since future applications may demand kilo-scale runs. Academic drive, paired with industry adoption, brings out better catalysts and cleaner transformations, keeping this molecule a step ahead of advancing sustainability and safety standards.
Toxicity Research
Discussion around any fluorinated reagent has to tackle its biological impact head-on. Short-term exposure data points to local irritation but not overwhelming acute toxicity compared to wilder organics. The longer-term story, still in progress, circles around biodegradation, environmental fate, and possible accumulation. Transparent reporting of incidents and near-misses means the collective understanding keeps growing. Toxicologists pair structure-activity studies with cell-based assays, watching for both cytotoxicity and genotoxicity. The practical lesson here: risk never goes away, but informed protocols, data sharing, and continuous monitoring tamp down dangers and allow progress to move forward, not back.
Future Prospects
Looking at the path ahead, ethyl 4,4-difluoro-3-oxobutanoate holds more promise than many might guess. Decades of research hint at even more effective uses, from catalysis refinement to new reaction pathways for constructing complex scaffolds. The broader push toward biocompatible synthons—compounds that slot smoothly into bioactive molecules—offers an opening for this molecule to expand beyond current uses. Improved waste reduction, cleverer recovery strategies, and real-time reaction monitoring chip away at barriers that once made wider use impractical. Every time a grad student or seasoned chemist reaches for this bottle, they're standing on the shoulders of those who learned, sometimes the hard way, how a small change in structure unlocks surprising utility. In an age where every molecule matters, both on the bench and in the field, compounds like this show how incremental progress and collaboration drive new waves of discovery.
Getting Real with Molecular Science
Ethyl 4,4-difluoro-3-oxobutanoate rarely makes headlines. Its chemical formula, C6H8F2O3, looks like a secret code, but behind it lies a practical story in synthetic chemistry. In research labs, chemists care about formulas like C6H8F2O3 not for their mystery, but for what they can unlock. This isn’t a household name alongside bleach or acetone, but once you start working in medicinal chemistry or advanced materials, you bump into such molecules surprisingly often.
Behind the Name: Why the Structure Matters
Dissecting the structure helps. Ethyl groups, those snug two-carbon chains, soften a molecule’s bite and tweak how it behaves in a reaction. The “4,4-difluoro” part turns heads because fluorine atoms, by swapping places with hydrogens, make molecules more robust. That’s huge for drug design. Medications last longer in the body, withstand breakdown, and hit their targets more precisely because chemists learned to sprinkle in a fluorine or two.
Add a ketone (the “3-oxo” piece), and now you’ve got a flexible scaffold. Builders use scaffolds to hold up whatever they want, and chemists do much the same. These building blocks transform easily, offering scientists a shortcut to more complicated or specialized molecules. Bottom line: C6H8F2O3 isn’t abstract chemistry, it’s foundation material for the next wave of pharmaceuticals and agrochemicals.
Why Fluorinated Molecules Draw Crowds
I remember the first time I saw a fluorinated intermediate take a plain old biological assay and send it off the charts. Out in the wild, fluorine is rare in life. Slip a fluorine atom into the mix, and living systems just don’t know what to do with it. That confusion helps scientists steer reactions, dodge some breakdown pathways, and keep compounds lasting longer. It’s no wonder that more than 20 percent of the top-selling drugs pack a fluorine punch these days.
Fluorination isn’t risk-free—manufacturing fluorinated chemicals chews up energy and can give off persistent byproducts. Chemists have gotten smarter, using milder reactions that waste less and avoid harsh reagents. It’s not perfect, but the field recognizes the ethical demand to work cleaner and cut environmental fallout. Scrutiny keeps the pressure on, and the move to greener chemistry stands as more than a talking point. Workers in labs need safe handling rules, and frontline communities near chemical plants seek higher standards for emissions. This pressure means more effort to develop gentler reactions and better containment tech.
Moving Forward
Scientists learn more by building on what works. Molecules like ethyl 4,4-difluoro-3-oxobutanoate don’t get billboards, but they help make medicines last longer, work better, and reach more people. The chemical formula, C6H8F2O3, represents more than a trivia question. For students, it’s a lesson in molecular logic. For workers in pharmaceuticals, it’s a reliable stepping stone. Keeping these molecules—and the way we make them—transparent, safe, and responsible pays off for the whole supply chain, from lab bench to health clinic. Chemistry isn’t just about symbols—it’s about giving materials, and people, a better shot at something lasting.
What This Chemical Means for Modern Chemistry
Ethyl 4,4-difluoro-3-oxobutanoate sounds unfamiliar to most people, but for scientists chasing better ways to build new chemicals, it attracts serious interest. That’s because it serves as a building block, especially where chemists want to add the strong, electronegative touch that fluorine brings. You find fluorine in plenty of the world’s pharmaceuticals and crop-protection agents, not by accident, but because this element can change how a molecule behaves in people, plants, and pests.
Pharmaceuticals: The Heart of Its Applications
Walk into any modern drug lab and you see how researchers value substances like this ester. Statins used against cholesterol, and antiviral treatments, both owe much of their effectiveness to the presence of clever fluorinated starting materials. Ethyl 4,4-difluoro-3-oxobutanoate delivers two fluorine atoms in just the right place, creating paths to compounds that the body might absorb differently, or break down slower. Drug makers use it to craft structures that keep medicines active longer, or make them less toxic for the liver.
Real experience counts here — projects aiming for more targeted cancer treatments often seek out new fluorine patterns. Colleagues in labs have told me how swapping in this ester has shifted drug candidates from mediocre to promising, simply due to changes in solubility and metabolic stability.
Pesticides and Agrochemicals: Feeding the Planet Safely
Farmers need tools to grow enough food, avoid spoilage, and cut down on crop losses. Many high-end pesticides work thanks to fluorinated chemicals, since altering just a few atoms on a molecule can swing it from ineffective to a potent protector against insects and disease. Companies interested in the next generation of safe pesticides lean toward building these products from strong, reliable starting points — and this ester checks that box.
With pests adapting faster than ever, agrochemical researchers keep pushing for compounds that break down at just the right rate in soil or water, without lingering risk to human health. This ester’s difluoro group helps strike that balance. It can slow down degradation just enough to finish the job, then fade away.
Crossroads With Specialty Chemistry
In the world of fluorinated materials, everything from Teflon coatings to polymer additives starts with smaller, more manageable pieces. Chemical makers draw on reactions with ethyl 4,4-difluoro-3-oxobutanoate to anchor fluorine securely in the backbone of custom-made plastics. My own stints in chemical plants made it clear that putting a tricky molecule like this to work actually guides future innovations in electronics and coatings, not just drugs or sprays.
The science linking these uses boils down to fine control. Chemists steer reactions with tremendous precision these days, and reagents like this one make the difference between ordinary results and breakthrough discoveries. While regulatory hurdles and environmental impact drive caution, this ester gives scientists options they would otherwise miss. That’s a big deal for anyone watching new medicines, smarter pesticides, or advanced materials evolve with each decade.
Getting Real About Chemical Precautions
Working in labs or chemical storerooms, one question tends to surface often: “How risky is this stuff, and what’s the right way to store it?” Ethyl 4,4-difluoro-3-oxobutanoate falls into that bucket of specialty chemicals that can trip up even seasoned users. Having spent years inside university research labs and small chemical plants, I’ve learned that assumptions about safety can be costly. Chemicals that seem mild still demand respect.
The Real Risks We Face
Essential details sit in the ingredient’s identity. Ethyl 4,4-difluoro-3-oxobutanoate carries a couple of potent fluorine atoms—these often increase reactivity. I remember a grad student leaving a loosely capped bottle of a related compound on an upper shelf; the next day, a strong odor told everyone something reacted. Here, similar carelessness could invite respiratory issues or accidental skin contact. Most esters serve as solvents or intermediates, and many release hazardous vapors. Neglecting a secure and well-ventilated spot for storage can lead to unexpected headaches, both figuratively and literally.
Storage Tips That Actually Work
Put the container somewhere cool, away from sunlight or hot pipes. Temperature swings make chemicals break down. From my own experience, the fridge in our old lab wasn’t meant for food—it was stocked with glass and Teflon-sealed jars, nothing else. We labeled every shelf with reminders, and nobody ever stashed lunch by accident.
Glass remains a smart choice for this liquid, though high-density polyethylene also gets used. Sulfuric acid once corroded a metal shelf in another lab, requiring a call to facilities and an afternoon of clean-up. With organofluorine chemistry, leaks or spills can get serious if material touches metal. Separate incompatible substances. Don’t position esters near bases, acids, or oxidizers. The fewer surprises, the better.
Wear the Gear—Every Time
Goggles, nitrile gloves, and a decent lab coat sound old-school until the day a splash lands near your eyes or wrists. People sometimes pretend that routine use means routine safety. In forensic science, I watched a technician develop a rash from a drop of reagent ignored during a busy morning. A fresh glove would have prevented that mess.
Keep spill kits nearby—don’t push them into a corner. Having dry sand or vermiculite to soak up drips keeps the workspace safe. If someone breathes in too much vapor, fume hoods and real airflow—not just a cracked window—make all the difference.
Training Makes a Difference
Nobody starts out knowing the quirks of every chemical, but experienced trainers shape safe habits. At one workplace, our old supervisor gave us quick demos for every new substance. He let us ask “dumb” questions and explained why not all gloves were equal. Those five-minute pauses eliminated so many near-misses.
Up-to-date fact sheets, clear hazard signs, and annual refreshers keep safety culture alive. Regulatory rules set the standard. The real commitment comes from actually walking through practices and listening to folks who know the risks from years of up-close work.
Building a Safer Workbench
Dealing with compounds like ethyl 4,4-difluoro-3-oxobutanoate isn't just about avoiding fines or filing paperwork. It’s about keeping coworkers healthy and making sure no project gets derailed by sloppy handling. The right routines, tools, and real-world stories go further than a binder full of protocols ever will. Remember, no shortcut replaces the lessons people learn by doing things right the first time.
Looking at the Risks: A Realistic Perspective
Every time I hear about new chemicals popping up in labs or manufacturing, I get that familiar prick of concern. Science always pushes for new boundaries, but every substance introduced in research and industry has a story that deserves a close look. Ethyl 4,4-difluoro-3-oxobutanoate—long name, strange formula—doesn’t ring a bell for most people, but for anyone who handles chemicals or cares about environmental safety, asking questions about danger and toxicity makes good sense.
From my time in university chemistry labs, the rule to treat every compound with respect never failed my classmates or me. Ethyl 4,4-difluoro-3-oxobutanoate stands out for a couple reasons. First, its structure signals a degree of caution. Fluorinated compounds often break down slowly when they get into the environment, and some turn out far more persistent or tricky to dispose of than researchers used to admit. This one, with its pair of fluorines stuck to the carbon backbone, draws alarm bells for folks tracking chemicals that hang around in soil or water.
What Science Says About Health and Environment
Nobody hands out safety awards to compounds just for existing. Safety depends on direct testing and transparent data. Ethyl 4,4-difluoro-3-oxobutanoate isn’t as widely studied compared to some other industrial chemicals, but certain trends merit attention. Similar fluorinated esters bring risks, especially when inhaled or if they come in contact with skin. Lab reports indicate irritation to eyes and the respiratory tract, raising the possibility of more serious effects if people don’t use gloves, ventilation, and basic protective gear.
Long-term effects take time to surface. Studies of related compounds show some build up in the body, sticking around long after exposure ends. That part always makes me uneasy. As a kid, I used to wonder why old factories needed big warning signs about leftover chemicals; as an adult, I recognize echoes of that same problem in today’s labs using newer, little-studied ingredients. Persistent chemicals, especially those with fluorine chains, have shown up in groundwater supplies and food webs, sometimes turning into bigger issues for communities than anyone ever expected.
Safer Practices and Smarter Choices
Reading about incidents in research and industry circles, most problems come not from intentional misuse, but from routine exposure and accidental spills. Choosing protective tools—the right gloves, goggles, and hoods—protects against the usual burns or rashes. Careful labeling, sharing up-to-date safety info, and training everyone in emergency procedures makes a huge difference; I’ve seen both the chaos of a poorly handled lab leak and the calm, almost boring process of a well-prepared response.
Green chemistry should be more than a buzzword. If a company or research group has a safer or more biodegradable option, picking that path avoids a spiral of future cleanup and health headaches. It’s less about fear-mongering, more about respecting the unknown and throwing energy behind responsible innovation. As consumers and professionals, pressing for better toxicity testing and environmental monitoring protects both individual health and the world we live in. Ethyl 4,4-difluoro-3-oxobutanoate probably won’t make the evening news, but it does push us to stay vigilant and value safety alongside progress.
Chemistry Outside the Textbook
Chemical names tend to sound intimidating, but inside every complicated title sits a practical compound with very real uses. Ethyl 4,4-Difluoro-3-Oxobutanoate falls into that category. Under the microscope—or really just on the benchtop in most labs—this molecule shows a story of transformation and application that runs deeper than its mouthful of a name.
What You Actually See and Weigh
In the lab, getting your hands on Ethyl 4,4-Difluoro-3-Oxobutanoate offers a direct look at applied organic synthesis. Chemists don’t just care about abstract concepts: the work starts by pouring, measuring, and reacting honest-to-goodness substances. This compound, for many, appears as a clear to slightly yellowish liquid. Having worked with plenty of esters over the years, I've learned to look for that faint but distinct ester scent—slightly sweet, slightly sharp, a reminder of basic organic chemistry labs in college that always left your hands smelling like an odd fruit salad.
Unpacking the Numbers
Run the math behind its molecular formula (C6H8F2O3) and the molecular weight clocks in at about 166.12 grams per mole. It’s not the largest compound around, but its size puts it in a useful range for most lab-scale syntheses. These numbers aren’t abstract for folks like me working in research or industry. Calculating a reaction yield, preparing a solution, or just keeping an eye on your chemical inventory all pivot on knowing the weight and having a mental image of the liquid in your glassware.
More Than a Number in a Catalog
Fluorinated compounds earn special attention in both pharmaceutical work and materials science. Ethyl 4,4-Difluoro-3-Oxobutanoate brings those difluoro groups right into the core of the molecule, letting chemists experiment with fluorine’s impact on reactivity and biological activity. The World Health Organization doesn’t just draft guidelines for fun: fluorine can sometimes boost drug stability and change how a therapeutic compound behaves in the body. This compound gives organic chemists a straightforward building block to introduce those tweaks early in a synthetic pathway.
Keeping Lab Safety Front and Center
Working with esters usually feels routine until someone in the lab learns the hard way about ventilation. Clear liquid doesn’t always mean harmless. Most esters—especially ones carrying heavier halogens like fluorine—need careful handling and the right protective gear. Avoiding splashes and inhalation gets taught early, but it bears repeating. Over my years in the lab, I’ve seen how a bit of complacency turns a small spill or a sniff of vapors into an unnecessary scramble for the eyewash bottle.
Connecting Knowledge to Problem-Solving
Molecular weight and physical appearance aren’t just trivia. To synthesize a fluorinated pharmaceutical candidate, to predict storage conditions, or to trace down a reaction’s byproducts, scientists lean heavily on these facts. A well-documented compound saves labs from guesswork, uncertainty, and risk. By focusing on clear, practical information about substances like Ethyl 4,4-Difluoro-3-Oxobutanoate, chemists, students, and anyone dealing with new chemicals can sharpen both safety and performance. Strong, shared knowledge builds smarter solutions, better experiments, and ultimately safer labs.