The Changing Landscape of Ethyl Bromodifluoroacetate: Past Lessons and Future Hopes
Tracing the Path: How Ethyl Bromodifluoroacetate Entered the Laboratory
Ethyl bromodifluoroacetate hasn’t always been a staple of organic chemistry pursuits. Its origins thread back to the evolution of organofluorine chemistry during the mid-20th century, an era marked by researchers eager to push the boundaries of fluorinated compounds for medicinal, agrochemical, and material purposes. I recall digging through old library stacks and seeing how synthetic tricks developed out of necessity rather than convenience. Chemists searching for new routes to CF2-group transfer landed on this reagent because they needed something handleable, liquid at room temperature, and reactive toward nucleophilic partners. The rise of perfluorinated building blocks—especially once the pharmaceutical industry caught on to how fluorine tweaks drug metabolism—put simple, robust reagents like ethyl bromodifluoroacetate in the spotlight.
Getting to Know the Molecule: Product Overview and Familiar Names
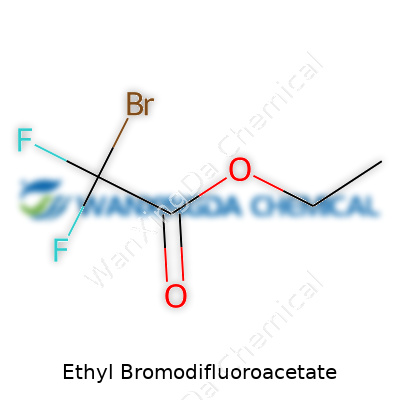
Pulling this compound from a bottle is nearly as familiar as coffee and potassium carbonate to a synthetic chemist. I’ve seen it labeled under a few aliases, like “Ethyl 2-bromo-2,2-difluoroacetate” or “Bromodifluoroacetic acid ethyl ester,” which can make inventory searches frustrating if you’re not careful. It’s a small molecule, highly reactive thanks to its electrophilic carbon center, and those tightly bound fluorine atoms. The colorless, sometimes faintly odoriferous liquid hides a punchy chemical personality. Its structure—an ethyl ester tipped by a gem-difluorinated bromoacetyl group—serves as a solid starting point for introducing difluoromethylene groups into more complex molecules.
Tangible Qualities: Physical and Chemical Properties You Notice in the Lab
Anyone who’s handled bromodifluoroacetate esters will pick up on their volatility and tendency to irritate. During my grad school days, I kept all my manipulations under fume hoods because there’s a sharp, somewhat acrid vapor. Boiling point hovers low enough to make open-flask heating dangerous, and even just sitting exposed invites significant evaporation losses. It dissolves well in common organic solvents like dichloromethane or ether. The bromine atom offers a handy site for further substitution, but it increases the molecule’s sensitivity toward light and heat. Fluorine’s presence boosts the stability of the adjacent bond, making this compound a rare blend of reactivity and chemical ruggedness. In short, you learn respect fast when pipetting or distilling this ester.
Preparation and Bench-Scale Realities
Creating ethyl bromodifluoroacetate often starts at the intersection of halogenation and esterification chemistry. In many setups, difluoroacetic acid reacts with ethanol under acidic conditions, then bromine is introduced to substitute a hydrogen and provide the crucial bromodifluoroacetyl center. The process is not trivial. There’s plenty of exotherm, toxic off-gassing, and a fight to prevent over-halogenation, which can send yields plummeting. Watching experienced chemists pull off this transformation brings home the importance of thermoregulation and thoughtful addition rates. On large scales, containment and ventilation take on even greater importance, especially as brominated volatiles are environmental and workplace hazards. Expect to see purification handled through careful distillation and drying, where a little water spells disaster for both yield and worker comfort.
How Chemists Bend and Shape: Key Reactions and Modifications in Practice
In real-world organic synthesis, ethyl bromodifluoroacetate earns its keep by acting as a versatile source of the difluoromethylene group. It reacts well with nucleophiles—think amines, alcohols, or carbanions—producing a suite of difluorinated intermediates. Historically, these transformations fueled the development of improved agrochemicals and drug scaffolds, owing to the unique effect of gem-difluoro groups on biological activity. My own research once focused on tweaking peptide backbones for drug candidates, and this compound allowed for stepwise incorporation of difluorinated motifs with high selectivity. Its reactivity with Grignard reagents or organolithiums provides routes to fluorinated alcohols or acids, steps that chemists prize when mapping out synthetic plans. Whenever the goal includes boosting lipophilicity or metabolic stability, ethyl bromodifluoroacetate is often the reagent of choice. Its flexibility becomes apparent after you’ve run a few dozen reactions; despite its niche appearance, it slots into many classic and modern synthetic strategies.
Safety, Lab Culture, and Professional Standards
Handling ethyl bromodifluoroacetate involves a blend of vigilance and routine. Over the years, I’ve watched even mature researchers make rookie mistakes—like skipping goggles or handling this chemical with bare hands. Reports peg it as an irritant to the skin, eyes, and respiratory tract, and inhalation of vapors can trigger headaches or worse. It reacts aggressively with strong bases and acids, sometimes generating hazardous byproducts. Agencies like OSHA and similar oversight bodies in other regions lay out strict rules for proper storage—cool, well-ventilated, away from sources of ignition or incompatible reagents. Spills need instant cleanup with specialized absorbents. From experience, the biggest surprise is how quickly this compound breaks through poor-quality gloves or sits as a lingering vapor if hoods aren’t running strong. Responsible chemists keep detailed SOPs for storage and disposal, since over time bottles degrade and seals weaken, upping the chances for leaks and contamination. Training staff properly ranks as just as important as labeling bottles or logging acquisition dates—because the most advanced safety system collapses without worker buy-in.
What It’s Good For: Application Areas That Stir Progress
Investigating the impact of difluoromethylene groups on biological activity, I quickly saw why ethyl bromodifluoroacetate shows up in pharma R&D. Its ability to insert a CF2 group between carbons imparts new metabolic stability to peptides, boosts binding affinity in enzyme inhibitors, and can transform an inactive molecule into a blockbuster. The agrochemical industry also finds value—modifying herbicides and fungicides to resist breakdown in the environment is a huge driver, and this reagent delivers. The electronics and polymer sectors make use of its building block status for specialty surface coatings and high-performance materials. I’ve seen patent filings balloon across Asia, Europe, and North America over the past decade, usually pointing to tailored small molecules that would be impossible without this reagent as a synthetic springboard.
Research and Development: Where Academic Curiosity Meets Industry Ambition
Research continues to chase two frontiers: safer, greener synthesis methods, and new applications demanding ever-more-complex fluorinated motifs. Collaborations between academia and industrial labs highlight new catalytic systems designed to minimize toxic byproducts and to operate under milder conditions. The literature is rich with strategies that cut down on hazardous waste or use alternative halogen sources, often spurred by mounting pressure for sustainable practices. I’ve had colleagues run pilot projects aiming to recycle bromine emissions and transform spent solvent streams into less hazardous effluent. The compound attracts attention for more than its utility—it’s become a flashpoint in the conversation about responsible chemical innovation, where progress won’t come at the cost of environmental or workplace health.
Peering into Toxicity and the Push for Understanding
Lab notes fill up quickly with observations about how potent many organohalogen compounds can be, especially those carrying both bromine and fluorine. Toxicology data points to irritation of mucous membranes, risk of chemical burns, and the classic challenge of assessing chronic low-level exposure, which often trails behind new uses. Animal studies suggest that difluorinated esters don’t accumulate in tissues like some perfluorinated counterparts, which offers slight reassurance—but uncertainty lingers. Regulatory agencies have started ramping up expectations for robust tox profiles before new production routes or applications receive approval. From my own perspective, erring on the side of caution feels smart, because the long-term impact of novel fluorinated fragments on both human health and the environment remains murky. Routine monitoring of workplace environments and regular review of handling procedures pay dividends far beyond regulatory compliance.
What Comes Next: Hopes and Pressures on the Horizon
Looking ahead, ethyl bromodifluoroacetate stands in a tricky spot—crucial for current research and industry but tethered to questions of sustainability and risk. The chemistry community keeps searching for ways to substitute the more hazardous steps in synthesis, such as developing flow processes and less hazardous halogen sources or dialing down the reliance on energy-intensive purification. On the innovation side, the demand for smarter, more active drugs and greener agrochemicals practically guarantees a steady need for fluorinated building blocks, with this ester as an affordable starting material. As international regulation tightens—thanks to environmental concerns about persistent organic pollutants—labs everywhere feel the pull to keep improving safety, tracking emissions more closely, and defending the business case for continued use. I’ve watched young researchers tackle these challenges head-on, blending creative synthetic thinking with eco-conscious protocols—a shift that could shape the next chapter for this molecule and for organofluorine science at large.
Chemical Formula and Why It Matters
Ethyl Bromodifluoroacetate comes with the formula C4H5BrF2O2. That means you get four carbon atoms, five hydrogens, one bromine, two fluorines, and two oxygens in every molecule. At first glance, that might seem like just a set of letters and numbers, but behind each symbol sits a story. Chemistry isn’t just for labs or textbooks—these formulas spill into daily life, affecting everything from modern medicine to how crops are grown.
Beyond the Letters: How Structure Shapes Use
This compound is not some rare lab curiosity. Chemists working in pharmaceutical and agrochemical industries use Ethyl Bromodifluoroacetate as a building block. That’s because its bromine and fluorine atoms act like a toolkit. You can swap pieces in and out, letting you build all sorts of new molecules, some of which might show up in medicines, pest control formulas, or specialty materials. The presence of two fluorine atoms isn’t just for show—fluorine’s high electronegativity shapes how the whole molecule reacts with other chemicals. Fluorine can make drugs more stable in the human body or help a pesticide stick around long enough to do its work.
Getting Personal About Chemistry
My time in undergraduate chemistry labs made these ideas hit home. We’d spend hours working with tricky reagents, many of them cradling halogens like bromine or fluorine. Someone might ask, “Why do we need these oddball molecules?” The answer tends to sneak up in project after project. Changing a single atom completely alters a compound’s behavior. A fluorine here shields a drug from being broken down too fast, or a bromine there makes a new link in a molecule possible, opening routes to new treatments or materials that do jobs nothing else can manage.
Keeping It Safe and Smart
No chemical compound operates in a vacuum. Stories from working chemists highlight how handling chemicals like Ethyl Bromodifluoroacetate demands close attention to safety. Brominated compounds often require fume hoods, and protective gloves are non-negotiable. Fluorinated chemicals risk accidental release of hazardous gases if mixed with the wrong reagents. The push toward greener chemistry is still finding its feet with halogenated organics, though—these compounds typically resist breaking down in the environment.
Looking for Smarter Solutions
Ethyl Bromodifluoroacetate stands as an example of how even small molecular tweaks drive progress across industries. Better handling procedures, smarter waste management, and closed-loop recycling systems offer ways to keep these benefits rolling without piling up environmental costs. Some research labs already pull off elegant reactions with fewer toxic byproducts, using milder reagents and less energy. The next step means joining those many small innovations together, so chemistry can keep solving problems without creating bigger ones down the line.
A Formula Full of Stories
It’s easy to lose track of what these formulas really represent. C4H5BrF2O2 isn’t just a collection of atoms; it's a gateway to progress, a source of challenges, and sometimes a spark for environmental concern. Anyone who has spent time mixing chemicals or reading up on reaction mechanisms walks away with respect for these seemingly simple structures. Each formula marks a meeting point between raw discovery and applied science—always with an eye on finding the next smarter, safer way forward.
Synthetic chemistry tends to create stars out of the oddest compounds. Take ethyl bromodifluoroacetate: it isn’t a household name, but for chemists tinkering with drug designs or building out new molecules, this stuff means business. My own work in the lab drew me to tools like ethyl bromodifluoroacetate because there’s a constant hunt for agents that can neatly slip a difluoromethyl group onto a molecule’s backbone. That simple twist in a structure can turn a dead-end lead into the kind of drug that resists breakdown in the body or slices through disease targets more effectively.
What Makes It Special in Synthesis?
This compound brings a double punch: bromine and two fluorines. Essentially, you get a molecule that serves as an excellent building block for adding carbon-fluorine bonds, which are a bit of a golden ticket in pharmaceuticals. A lot of medicines loaded with fluorine get better bioavailability and metabolic stability—big wins if you’re hoping to get a drug approved or keep it active in the bloodstream long enough to actually do something.
In everyday research, ethyl bromodifluoroacetate turns up in processes like nucleophilic substitution and cross-coupling reactions. Chemists often grab it because it’s primed and ready to deliver that —CF2— group into all sorts of frameworks. I’ve helped collaborators who use it to assemble potential new antivirals and anticancer molecules, where the difluoro effect makes the difference between “maybe” and “publishable.” Published studies in organic chemistry journals show that, over the past ten years, use of this intermediate has accelerated as more chemists focus on fluorinated motifs. In fact, about one in four new small-molecule drugs today either contains fluorine or was made using a fluorine-bearing intermediate like this one.
Beyond Pharma: Other Frontiers
Ethyl bromodifluoroacetate doesn’t just crop up in drug research. Agrochemicals, for example, benefit from its knack for helping design pesticides that hang around on crops just long enough to do the job, but not so long they cause ecosystem headaches. With the food supply so tightly regulated, researchers lean on molecules like this to thread that needle—knocking out pests while still passing residue tests.
I’ve also talked to material scientists who pull up ethyl bromodifluoroacetate to build out pieces of fluorinated polymers and specialty coatings. These fluorinated materials often resist water, oils, and even harsh acids, so their properties make them welcome in everything from non-stick cookware finishes to protective surface treatments on electronics.
Environmental and Safety Considerations
The demand for fluoro-organic compounds keeps rising, but there’s a catch. Handling chemicals like ethyl bromodifluoroacetate brings real safety stakes—skin contact, inhalation, and environmental disposition require lab teams to stay sharp. Persistent organic pollutants remain a concern, so it feels right that more companies and chem labs face tough questions on safe use and disposal.
One practical solution focuses on building more efficient syntheses, so less waste and lower risk stick around during scale-up. Green chemistry efforts push for milder conditions and safer alternatives where possible. Meanwhile, thorough training and robust fume hoods help keep my colleagues safe in the trenches. As chemists, we’re handed powerful tools; what we build, and how responsibly we do the job, matters just as much as what ends up on the shelf.
Why It Matters: Beyond the Lab Bench
In the chemical world, a few drops or an extra degree can make the difference between a safe outcome and an emergency. Anyone using Ethyl Bromodifluoroacetate knows that attention to detail isn’t just for peace of mind; it stands between a smooth process and a dangerous spill. I’ve spent enough time in chemistry labs to notice that rushed or careless storage habits crop up more than they should, especially around specialty solvents or reactive intermediates like this one. So, let’s lay out why people need to pay proper attention to how this compound is handled and stored.
Stability and Safe Storage
Ethyl Bromodifluoroacetate offers a specific chemical niche, useful in a handful of fine chemical applications and synthesis pathways. Its reactivity and volatility mean the wrong storage environment increases the risks of unwanted reactions. I’ve seen cartons stacked too close to heat sources, and every time someone shrugs off proper labeling or skips securing the cap, they jeopardize not just the experiment, but themselves and their coworkers.
Storing this compound away from any source of ignition is more than a box-ticking exercise. It belongs someplace cool, preferably below room temperature, in a well-ventilated chemical storage area—ideally in a flammable liquids cabinet. Direct sunlight or temperature swings can destabilize it. Damp conditions introduce water that can get into the container and react. Moisture always loves to find a way in, so airtight containers matter. Pairing vigilance with the right gear keeps mishaps at bay.
Common-Sense Precautions During Handling
Even years after chemistry classes, simple rules don’t change: never pipette by mouth, and use gloves and goggles for even quick transfers. I’ve seen how a minor splash can sideline someone from work for days. Fume hoods eliminate the risk of inhaling fumes; too many folks underestimate how quickly a sharp-smelling vapor can appear from the smallest spills. Subtle symptoms can show up hours later, especially with more obscure compounds.
Labeling makes life easier, especially during busy weeks or shared spaces. Too vague or peeled labels can lead to mix-ups, which are dangerous in high-turnover labs. I’ve always relied on double-checks and clear records, not just because I was taught to, but because seeing colleagues scramble to account for “missing” chemicals taught me the importance of transparency.
Facts on Accidents and Solutions for Safer Practice
Reports of laboratory accidents—from universities to start-ups—often boil down to overlooked best practices. A misplaced solvent or unlabeled bottle can lead to small leaks or even fires. Data from workplace safety organizations backs this up: most chemical incidents in labs come from human error, not from malfunctioning equipment.
Moving towards safer storage and handling means regular training, clear signage, and respecting those procedures usually included in safety briefings. Quick access to spill kits makes a difference, too. Making sure every team member feels comfortable speaking up about unsafe situations builds a culture that prevents neglect. Encouraging open conversation about near-misses helps spot weak points in habits or storage conditions.
Looking Ahead
Regulatory guidance exists for good reasons, but personal accountability and respect for the risks of Ethyl Bromodifluoroacetate keep accidents low. Cutting corners with hazardous chemicals invites trouble. Real safety comes from a mix of knowledge, the right equipment, and healthy respect—the kind you only get from real-world experience and seeing what happens when those steps get skipped. Industry needs more of that kind of shared wisdom, not less.
A Personal Take on Lab Safety and Chemical Hazards
I remember the first time I handled a volatile compound with two sets of gloves and an uneasy feeling. Even seasoned lab workers get that twinge now and then—especially around chemicals like ethyl bromodifluoroacetate. With its clear, low-key appearance, this compound tricks even careful folks into lowering their guard. Yet, it's got a punch that deserves respect well beyond its looks.
The Health Hazards Lurking Behind the Label
Let’s get straight to what matters: inhalation of its vapors or even lingering mist can send someone to the emergency room. Breathing in this stuff means immediate trouble for your airways. Throat burning, coughing fits, and in worse scenarios, lung inflammation. A splash on the skin, thanks to bromine’s angry chemistry, can burn through to the deeper layers. Your eyes face an even higher risk—painful damage or vision loss isn’t out of the question if you decide to play it fast and loose.
Some people don’t take vapor toxicity seriously until it’s too late. Yet, the long-term side effects of regular exposure, even in tiny amounts, stack up: potential liver impact, nervous system symptoms, and unpredictable reactions in future exposures. While written warnings get ignored on busy days, memories of colleagues who paid the price with lasting health issues always reinforce the need for focus.
It’s Never “Just a Small Transfer”
Working with ethyl bromodifluoroacetate in research, I never trusted flimsy ventilation or quick washes. The proper PPE routine—thinking beyond single gloves, always wearing goggles, avoiding open shoes, keeping lab coats snapped up—wasn’t about bureaucracy. It was about seeing the aftermath of a spill or inhalation scare in others and not wanting to experience it myself. Chemical fume hoods aren’t “nice-to-have” here. Realistically, a closed hood with good airflow separates a regular shift from a panic call to poison control.
Why It Matters Outside the Lab as Well
Disposal slips get casual treatment in some labs. This catches up later, when someone unknowingly opens a container months down the line. Proper labeling, secure cap replacement, and making sure waste bottles aren’t overfilled or stored in the wrong place become a shared responsibility. Stories of costly evacuations—just from small leaks—teach everyone a lesson the hard way.
The chemistry community sometimes underplays the mental side of handling nastier compounds. Knowing your limits and pressing pause helps more than bravado ever will. Keeping updated Safety Data Sheets, running drills, checking gear for nicks or thin spots, and supporting each other in safety-first habits lets researchers focus on discovery instead of worrying about avoidable trips to the ER.
Building a Safer Place to Work
It only takes a few seconds for complacency to undo years of safe practice. I’ve seen the difference strong mentorship and open conversations about risk make, especially for folks just starting out. Pair that with access to spill kits, eye wash stations, and clear pathways to exits, and you create more than compliance—you build a culture where everyone looks out for one another. In research, chemistry’s real breakthroughs come from safety, not shortcuts.
Why Purity Matters for Life, Not Just the Lab
Chemicals like ethyl bromodifluoroacetate make headlines mostly among scientists and manufacturers. Purity doesn’t just boost a statistic printed on a label—it makes or breaks the process for chemists working to create new medicines, crop protection agents, or specialty compounds. Over many years, working in both academic and industrial labs, I’ve seen that a reagent with lower purity turns a promising reaction into a day wasted on troubleshooting.
With ethyl bromodifluoroacetate, buyers regularly look for the highest possible purity, usually 97% or above. The reason is simple: traces of water, other chemicals, or leftover solvents can throw off the whole synthesis. Low impurity levels spare researchers from repeating failed reactions, which costs time and money. In drug development, a single misstep due to impure reagents can send a whole month’s work back to the starting line.
Packaging Sizes: Options and Real-World Tradeoffs
Ask three chemists what packaging sizes they prefer and you’ll hear three different answers. In everyday lab life, the decision comes down to volume needed, safety, and budget. Ethyl bromodifluoroacetate often comes in glass bottles ranging in size from 5 grams up to 100 grams. Some suppliers offer half-liter or liter bottles for those running pilot plant experiments or scaling up for industrial batches.
At the bench scale, smaller sizes do have more fans. Handling 5- or 10-gram bottles cuts waste, lowers the risk from spills, and lets you keep a fresher supply. When I worked with reactive reagents, even slight exposure to moisture could degrade the compound inside a week, making larger containers more trouble than they’re worth if you only use a little at a time. For large-scale users—think chemical plants or major R&D groups—bulk purchases keep costs down. Handling bigger bottles always means stricter storage, better ventilation, and safety routines to match the increased hazard.
Keeping Trust With Reliable Sourcing
Purity and packaging seem like dry, technical choices. Yet both serve as markers of whether a supplier is serious about your success. In my career, I’ve relied on well-established chemical suppliers for one reason: they back up their claims with clear documentation. For ethyl bromodifluoroacetate, this means a certificate showing purity and impurity levels, ideally verified by third-party labs. If a company hesitates to share this, that’s a red flag.
Big-name catalog companies, as well as smaller specialty dealers, know that professional transparency builds trust. When the difference between 97% and 99% purity can change chemical properties, that documentation matters. Many in the research community spread news—good or bad—about misleading vendors in minutes, and one bad batch can inflict a global hit on reputation.
How Better Choices Start With an Informed Buyer
Labs struggling with budget cuts sometimes reach for the cheapest reagent or the bulk size that looks like the best deal. Experience teaches that a little caution saves far more down the line. Knowing where and how your ethyl bromodifluoroacetate comes packaged, plus demanding careful purity standards, ensures true progress over shortcuts. Science, at its heart, runs on trustworthy building blocks. This one detail often separates a lab that advances discovery from one caught in endless troubleshooting.