Ethyl Difluoroacetate: Navigating a Niche Compound’s Journey
Historical Development
Ethyl difluoroacetate doesn’t usually grab headlines, but it has taken an interesting route from obscure lab curiosity to trusted chemical tool in research and manufacturing. From the early days of fluorinated chemistry, scientists have chased new compounds that offer unique reactivity and stability. The presence of two fluorine atoms in its structure gave this ester a slight edge, especially during the late twentieth century as organofluorine chemistry became a hotbed for drug development and material innovation. Looking back, experienced chemists would recall its introduction to fine chemical catalogs around the 1970s or 1980s, by which point awareness of selective fluorination effects had drawn enough interest to get this compound into professional hands. Old notes and journals often referenced it as a handy building block, though nobody thought it would overtake its relatives in bulk demand.
Product Overview
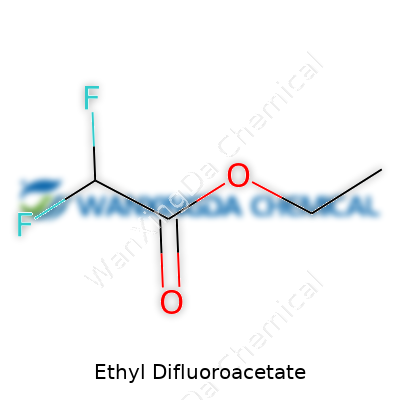
Ethyl difluoroacetate stands as a colorless liquid, carrying a faint fruit-like odor. A simple ester backbone combined with two fluorines off the acetyl group creates subtle but powerful changes in its chemical temperament. I’ve always found that a molecule like this, which slots between the commonplace and the specialized, usually appeals to chemists who operate on the margins of mainstream synthesis. It isn’t found on warehouse shelves in every industrial plant, but those who use it, trust it for specific reactions and standards. Part of its reputation comes from its solid performance in both academic routes and the production of specialty materials, often in the pharmaceutical and agrochemical spaces.
Physical & Chemical Properties
This compound brings a boiling point hovering near 85 degrees Celsius and mixes well with common organic solvents, unlike many other fluorinated esters that often pose either solubility or volatility restrictions. Its moderate polarity and low viscosity ensure easy handling in routine bench work and pilot-plant applications. The fluorines dial up both stability and reactivity, making for a substrate that resists random breakdown yet takes part in predictable transformations under controlled conditions. Its density falls just above that of water, so it doesn’t float or separate too quickly in mixed-phase systems. These features make it a reliable standard for NMR studies involving fluorine, a lesson I picked up early in my career.
Technical Specifications & Labeling
Labels on ethyl difluoroacetate bottles always salute the purity grade, since trace impurities—like unreacted acids or water—can wreck selectivity in fluorination reactions. Reputable suppliers promise content above 97 percent and often test for residual acidity or halide contamination. In practice, those working in analytical settings or scale-up R&D prefer a handful of lab-vetted brands, which often come marked with batch-specific analysis reports. Proper documentation does more than meet regulatory standards; it acts as a trust anchor in a field where a percent or two off-spec can send weeks of effort down the drain.
Preparation Method
Most chemists have learned to make ethyl difluoroacetate using the esterification of difluoroacetic acid with ethanol in the presence of an acid catalyst. In the classic route, difluoroacetic acid, itself derived from controlled reactions between chloroacetic acid and fluorinating agents, meets ethanol under reflux and produces the ester as a distillable fraction. The operation takes careful control, not just to avoid toxic fumes but to maximize yield and cut back on corrosive byproducts. I’ve seen experienced hands veer toward azeotropic distillation to dry their product and neutralize any mineral acid traces with gentle aqueous workups. For scale producers, the need to handle hydrofluoric acid risks upstream is a stark reminder that one compound’s simplicity can hide layers of complexity behind the scenes.
Chemical Reactions & Modifications
The value of this ester isn’t just in its raw form but in its ability to play multiple supporting roles. Nucleophiles attack the carbonyl or α-carbon, making it a foundation for forming more complex difluoroacetylated molecules. In pharmaceutical contexts, adding difluoroacetyl groups means changing a molecule’s metabolism or receptor affinity, often by introducing metabolic stability. Reactions involving Grignard reagents, amines, or reduction pathways lead to amides, alcohols, or more heavily tailored derivatives. The unique properties imparted by those fluorines, including resistance to enzymatic breakdown, have driven many teams—myself included—to reach for ethyl difluoroacetate over less exotic building blocks.
Synonyms & Product Names
For those searching literature or procurement catalogs, ethyl difluoroacetate sometimes appears under names like "Difluoroacetic acid ethyl ester" or even coded shorthand like "EDFA". Chemical abstracts and regulatory documents assign CAS numbers, but real-world workers more often trade stories of product grades, supplier quirks, or historical batches than catalog numbers. The web of trivial names in organic chemistry sometimes clouds discussions, but a seasoned chemist quickly learns to ask for clarification before committing to a purchase.
Safety & Operational Standards
Anyone with a history of handling fluorinated organics knows to respect their potential hazards. Ethyl difluoroacetate doesn’t usually present acute toxicity in small-scale usage, but its vapors and accidental skin contact need prompt response. Inhalation or spill cleanup means opening windows wide and grabbing gloves and goggles without hesitation. Labs and production sites follow standard containment and fume extraction routines, given that both hydrolysis and combustion can unleash toxic acids and phosgene-like byproducts. Guidance from regulatory agencies lines up with years of anecdotal advice—keep this material labeled, contain spills quickly, and store in well-ventilated, cool areas. Firefighters and hazmat teams get the headline safety info, but at the bench, a proper respect for the reactivity and volatility always pays off.
Application Area
Despite its low-volume reputation, ethyl difluoroacetate pops up across fields as varied as medicinal chemistry, specialty polymers, and crop protection studies. In pharma research, teams use it to add difluoroacetyl motifs to candidate molecules, changing both potency profiles and metabolic lifespans. Agrochemical firms deploy it as a precursor to new herbicides and fungicides, counting on the metabolic resilience that fluorines provide. On the materials side, designers searching for polymers with unique surface energies or dielectric properties often incorporate such esters to inject new behaviors. In each field, those tiny fluorine atoms shift both performance and lifecycle, justifying the extra effort in handling and cost.
Research & Development
Ethyl difluoroacetate rarely headlines patents or blockbuster molecules, but researchers have built libraries of molecules off its backbone since at least the 1980s. Its most creative uses often emerge where teams want to push molecular boundaries—like tuning pharmaceuticals for cross-barrier stability or building fluorine-rich scaffolds that challenge existing synthesis dogmas. Plenty of academic groups still chase ways to introduce or exploit difluoroacetyl groups in bioactive design, and I’ve seen multidisciplinary consortia pick this building block for pilot-scale crop protection screens. Not every attempt results in a market product, but the legacy of scientific imagination built on this molecule continues to ripple out.
Toxicity Research
Fluorinated esters demand a cautious approach to toxicity, and the available studies show a mixed bag of acute and chronic impacts. Small doses typically bring mild irritant effects, but the long-term risks align more with repeated or unintentional exposures, echoing concerns seen with other small organofluorines. Toxicologists highlight the risk that breakdown products, especially difluoroacetic acid, might linger in biological systems or water sources. Most findings so far suggest low bioaccumulation potential, but science hasn’t delivered a comprehensive answer. In the absence of deep longitudinal studies, experienced users stick with the strict safety margin—ventilation, gloves, and limiting exposure—while debates about environmental fate continue in regulatory and industrial circles.
Future Prospects
Looking ahead, ethyl difluoroacetate seems poised to hold its niche as an enabling intermediate for inventive chemists. The slow but steady march toward greener and safer fluorination methods might reshape how it's prepared, with more attention paid to atom economy and waste management. If demand rises for small molecule drugs or novel crop protectants incorporating difluoroacetyl motifs, production technologies and supply chains will need to catch up, both in terms of scale and sustainability. From my time in the field, it’s clear that compounds like this—specialized, sometimes underestimated—feed directly into the breakthroughs that drive the next solid leap in chemical and pharmaceutical science. As more research unfolds and regulatory pressures sharpen, the ability to reliably prepare, handle, and deploy subtle compounds like ethyl difluoroacetate will only grow in strategic value.
Ethyl difluoroacetate isn’t catching headlines on major news networks, but anyone with a foot in synthetic chemistry or pharmaceuticals recognizes this compound’s quiet importance. Produced through the esterification of difluoroacetic acid, its clear liquid form holds genuine value, especially in research labs and chemical plants worldwide. Its chemical structure, decorated with two fluorine atoms, makes it more than just another organic ester on a shelf.
Role in Making New Medicines
I’ve watched project after project in pharmaceutical labs speed up thanks to building blocks like ethyl difluoroacetate. Medicinal chemists depend on fluorinated compounds to adjust how drugs behave inside the body. A single tweak—adding a fluorinated group—can totally shift how a molecule interacts with enzymes, how fast the liver clears it out, or whether it irritates the stomach lining. Ethyl difluoroacetate makes it possible to add these tweaks. The growing pile of fluorinated medicines, from antivirals to cancer drugs, shows this ingredient’s value.
Chemistry’s Modular Approach
The demand for difluoro-containing building blocks didn’t just come from the pharmaceutical industry. Agrochemical development teams use these compounds to design pesticides that last in a field but break down safely later. Sometimes, these tweaks help avoid resistance in pests and disease, as changing a molecule’s structure with fluorine can prevent bugs and weeds from adapting so quickly.
The fun part is this ester doesn’t just bring two fluorine atoms for the ride. It offers a pretty reactive platform for further chemical transformation. It became a part of my toolkit back in graduate school, joining the long list of chemicals that help stitch together new, never-before-seen rings or carbon skeletons. Researchers can swap the ethoxy group, break the bond at the alpha-carbon, or use it as a source of the difluoromethyl group. It’s flexible and often saves time compared with older, multi-step routes.
Cost, Safety, and Environmental Impact
Safety and costs still need watching, of course. Fluorinated chemicals sometimes hang around in the environment for a long time, resisting natural decay. That means disposal must be thoughtful and controlled in any lab or plant working with these materials. Early in my career, I saw firsthand how easy it is for waste streams to get contaminated when people cut corners. Responsible companies adopt capture and destruction methods for fluorinated byproducts, but lapses have already left a legacy of environmental pollution in some regions. None of this is unique to ethyl difluoroacetate alone, but every link in the chain matters.
Why It Deserves Attention Now
Today’s chemical toolbox includes more sustainable alternatives for some processes, sparking questions about the continued use of synthetic fluorine chemistry. Still, progress in medicine, agriculture, and materials depends on precisely these building blocks. The key lies in smarter synthesis, tighter waste controls, transparent reporting, and supporting scientists developing greener options. Chemists, regulators, and supply chain managers need to work together, learning from those who saw the pitfalls early and pushed for better oversight. Only then do life-changing drugs and advanced materials justify the risk of complex, persistent ingredients like ethyl difluoroacetate.
Understanding the Basics
Ethyl difluoroacetate comes with a chemical formula of C4H6F2O2. At first glance, it might look like a random string only a chemist cares about. I remember the first time I tried to memorize so many similar formulas in college – always a challenge to keep them straight. Yet, every arrangement of letters and numbers here tells us about function and purpose in the real world. The “difluoro” part points to two fluorine atoms, which completely change the behavior of what looks otherwise like a basic ester. The inclusion of those two fluorines is no simple tweak. They matter for how this chemical reacts, how it resists breakdown, and where it can find a use.
Meeting the Needs of Science and Industry
Industries demand precision. Ethyl difluoroacetate has made its way into research labs, particularly in synthetic organic chemistry, thanks to the appeal of fluorinated compounds. Adding fluorine atoms to molecules often improves the stability of certain pharmaceuticals, sometimes reshaping a molecule’s biological profile entirely. In crop science, similar compounds provide certain pest-resistant benefits. The presence of those two small fluorines often means the difference between a promising result and wasted effort.
Knowing the formula C4H6F2O2 lets chemists calculate quantities precisely and predict interactions in a reaction vessel. This is not just for academic satisfaction—dosing even a little too much of the wrong component sends a whole synthesis off track. Mistakes cost time, money, and sometimes safety. My earliest jobs in the lab had a constant whisper from supervisors: the chemical formula isn’t just trivia. It’s a tool, a map, and sometimes a lifeline.
Why Purity and Accuracy Count
Small errors in a chemical’s makeup can ripple through an entire process. In fluorinated chemistry, side-products often introduce unpredictability, which carries over into cost and risk. If a shipment of ethyl difluoroacetate does not match its expected formula, downstream applications run into trouble. Pharmaceutical developers need consistency, or else their experiments collapse. Researchers need trust in their reagents or else work slows to a crawl. Having clear, accurate chemical data—starting with that simple formula—keeps machinery running, supply chains flowing, and discoveries possible.
How We Can Improve Understanding
One practical step includes improved education. Chemistry can intimidate, especially with jargon and numbers, but drawing real-world connections makes it less abstract. If schools and universities bring more context to formulas, future researchers will care more about the connections these formulas create. Investing in clear labeling, standardized certifications, and open channels between suppliers and users helps as well. Chemical producers who make their analysis transparent help build confidence that what arrives in the bottle is what the label promises.
FAQs and outreach from industry leaders would help dispel mix-ups. Often, confusion emerges because people lose sight of what the formula actually guides: real results in the lab, not just theory. Advocacy for open sharing of safety data and real-world stories about how precise formulations change outcomes will inspire more collaboration. Then, the importance of keeping a close eye on even a six-character formula seems obvious.
Conclusion
C4H6F2O2 does more than fill a slot on a shelf. It symbolizes a meeting point of exact science, industrial need, and the hands-on work that shapes modern progress. Chemistry, at its core, rewards anyone persistent enough to ask why each formula matters.
Keeping Chemicals Safe Isn’t Just for the Big Names
Anyone who has spent time in a chemistry lab knows the challenge of handling chemicals that don’t make headlines but can still cause major problems if left unchecked. Ethyl difluoroacetate is one of those quietly tricky substances. I remember walking into a university lab some years ago and finding a shelf cluttered with bottles, some sticky and crusted. A container of ethyl difluoroacetate stood out—not for its prestige, but because someone had scribbled a warning about a strange odor. Small mistakes in storage can lead to big headaches.
The Temperamental Nature of Ethyl Difluoroacetate
This compound brings a mix of volatility and reactivity, making it important to respect its quirks. Ethyl difluoroacetate acts fast—fumes waft out if a cap isn’t tight, irritation hits the eyes and nose, and sensitive electronics sometimes don’t get along with its vapors. Even amateur chemists catch on quickly: this isn’t like handling salt and vinegar. Leak a little, and everyone knows in minutes.
Why Care About Storage?
Poor storage turns a useful reagent into an accident waiting to happen. Having seen a shelf buckle under unexpected weight from leaking solvents, it’s not just a theoretical concern. More than once, students have had to evacuate and scrub out a space because a cap loosened or the wrong plastic got eaten through. Each incident slows down work, wastes money, and sometimes leaves a faint sharp tang behind for weeks.
Practical Experience in Keeping Hazards at Bay
Experience beats reading a label every time. Glass works better for stashing ethyl difluoroacetate than cheap plastics. Some containers look airtight but let fumes slip out, especially after a few uses. A solid glass bottle with a reliable seal cuts down on complaints and keeps the shelf below from turning into a sticky mess.
Nobody wants a storage room that smells like a chemistry set gone wrong. A chemical like this sits better inside a metal or vented cabinet, away from acids or bases. Ordinary steel shelves rust quickly when airborne chemicals react. Chemical storage cabinets with ventilation and spill-containment trays show their worth in the long run. The cost might seem steep upfront, but not having to replace nearby gear or scrub floors pays off.
Chemical Safety Isn’t a Static Checklist
With ethyl difluoroacetate, I’ve found that checking containers every few months makes a real difference. Even a perfect initial setup can fail: labels fall off, glass cracks, caps loosen after repeat openings. A periodic review—especially after heavy use—keeps potential hazards in check. Routine makes storage part of the culture rather than an afterthought.
Accident stories circulate through every lab: a small spill leads to an alarm; a vapor leak sparks worries about who breathed what. Some labs now pair hazardous storage with clear training so even the newest staff knows what not to mix and how to spot trouble before it escalates.
Building Trust and Accountability
Trust in a research or teaching environment has to be earned through consistency. I value places where staff speak out about storage, check one another’s work, and put time into equipment upkeep instead of rushing for short-term savings. A good culture doesn’t take heroic effort—just attention.
Safe storage turns out to be less about chemical formulas and more about careful habits, plain communication, and simple steps repeated again and again. A well-run lab proves its worth every day through these unshowy choices.
Everyday Chemistry, Real Concerns
People think chemistry belongs in labs, out of sight and out of mind. That’s not the reality for many of us who live and work near places where industrial chemicals get used or stored. Ethyl difluoroacetate doesn’t make headlines unless something goes wrong, but understanding whether it’s hazardous matters—especially when consumer safety, worker health, and environmental responsibility are at stake.
Understand What We’re Dealing With
Ethyl difluoroacetate is a specialized chemical, popping up mostly in research labs and factories that work with custom organic molecules. The way it smells—sharp and biting, similar to strong cleaning products—should clue anyone in: this isn’t a bottle you want to handle carelessly. Many fluorine-containing chemicals have a reputation for being more reactive and sometimes more harmful than their non-fluorinated cousins. Anyone who has worked with solvent-like compounds can recognize the warning signs, even without seeing skull-and-crossbones labels everywhere.
Health Risks: Not Just in the Fine Print
Direct contact with ethyl difluoroacetate can irritate skin and eyes; that’s common sense for anyone who’s spilled so much as vinegar on their hands, much less something engineered for lab work. Breathing in its fumes goes beyond mere discomfort—prolonged exposure has the potential to cause headaches, dizziness, and other symptoms. Fluorinated compounds sometimes break down into even nastier byproducts, such as hydrogen fluoride, which has a well-documented history of causing severe burns and lasting respiratory damage. The risk grows with larger spills or accidents, especially where ventilation is poor or protective equipment goes neglected.
Environmental Impact Gets Overlooked
Folks working in waste management or environmental testing will tell you: once small specialty chemicals hit the water or soil, they rarely stay put. Ethyl difluoroacetate doesn’t naturally biodegrade fast, so improper disposal can lead to buildup. Wildlife faces risks we often forget about, especially aquatic organisms that can suffer from chemical run-off. Chlorine, bromine, and fluorine-based organics have made their way into drinking water sources before—most infamously in the case of PFAS—which teaches us to pay attention before another problem takes root.
How Do We Protect Ourselves and Our Communities?
One answer lies in supporting good training and awareness for anyone who works with such chemicals. No amount of paperwork beats actual familiarity with safe storage and proper ventilation. Companies bear responsibility here; regular audits, spill drills, and clear labeling policies are more than red tape—they’re what keep routine work from turning into a crisis.
Strong oversight matters as well. Regulatory agencies, from the EPA to the European Chemicals Agency, often track and review chemicals that pose health or environmental threats. Industry should cooperate with these watchdogs, not treat them as adversaries. Open sharing of incident reports and toxicity studies helps prevent harm, and policymakers need to listen to local communities where these substances are used.
Sometimes the best solution involves switching to less hazardous alternatives, or at least minimizing how much ethyl difluoroacetate enters the waste stream. In my own experience, researchers and operations managers rarely look for alternatives until regulations change or public pressure mounts. Proactive steps—using closed systems, strengthening PPE rules, supporting green chemistry research—bring peace of mind for everyone, not just those in the lab.
A Call for Common Sense and Caution
Treating ethyl difluoroacetate with respect and care isn’t overkill—it’s just smart. I’ve seen firsthand what can happen when shortcuts get taken, and the aftermath always hurts more than a little extra effort ever would. By taking these risks seriously and arming ourselves with knowledge, we do right by workers, neighbors, and the environment. Trust in science isn’t just about discovery; it’s about responsibility once that discovery steps into the real world.
Staying Safe Isn't Just a Checklist — It’s a Habit
Anyone who’s spent time in a chemistry lab knows certain chemicals demand respect. Ethyl difluoroacetate fits that bill. I remember early on, watching a senior researcher handle it with calm but steady caution — gloves on, fume hood humming, and goggles never coming off. It stuck with me. This isn’t a compound for shortcuts or guessing games.
Ethyl difluoroacetate serves as a building block for specialty chemicals, pharmaceuticals, and research projects. Its volatility and reactivity can catch even seasoned techs by surprise if they treat it like just any bottle on the shelf.
No Substitutes for Proper Storage and Equipment
I’ve seen clean-up drills that made all the difference later. This liquid doesn’t play nice with open air or heat. Left in a warm lab near direct sunlight, it can break down or even catch fire. That risk alone shows why a cool, ventilated storage space matters. Forgetting this turns a safe workspace into a hazard zone.
Glassware kept free of cracks and only using containers with tight seals greatly lowers the chance for spills or leaks. The right storage isn’t a suggestion—it sets workers up for a safer day, and that affects everyone in the room, not just the person pouring the liquid.
Personal Protective Gear is Non-Negotiable
Goggles, chemical-resistant gloves, and lab coats become basic armor. During a rushed spring semester, I spotted someone skip the gloves thinking they’d be careful. A drop landed on their skin. Even after a fast wash, some irritation lingered for days — a reminder that chemical burns aren’t rare.
The fumes can irritate eyes and lungs, so a working fume hood brings more than just comfort; it cuts down exposure sharply. I’d rather deal with a noisy vent than risk coughing through the day. Frequent checks of the hood make sure it actually pulls vapors away, not just makes noise.
Training and Simple Communication Save Trouble Down the Line
I once joined a project where the safety orientation seemed like an afterthought. Within a week, somebody stored the bottle on a cluttered bench instead of inside the ventilated cabinet. After a heated conversation (and a near-miss with some spilled solvent), we updated our training. That made everyone rethink how important simple reminders are.
Clear instructions need a human touch — “This reacts quickly, so pour slowly.” “Don’t set it down on the heat block.” Safety talks work better if everyone in the team feels invited to speak up. Mistakes often happen because people don’t want to ask, or they assume someone else will clean up.
Addressing the Toughest Challenges
Few workplaces have endless budgets for the latest gear. Even so, careful labeling, double-checks, and shared responsibility keep everyone out of trouble. Some labs rotate the safety leader role, making sure it’s part of daily conversation, not background noise.
For those working with ethyl difluoroacetate, handling it with routine vigilance protects health and research alike. Building good habits, investing in gear that works, and encouraging everyone to take safety personally may sound basic, but these steps turn big risks into manageable tasks.