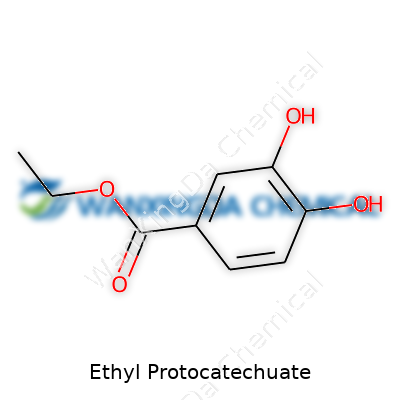
Ethyl Protocatechuate: Reflecting on a Chemical With Roots in Research and Industry
Historical Development of Ethyl Protocatechuate
Chemists first described protocatechuic acid in the 19th century, opening a path for the development of related esters soon after. Over the years, synthesis of ethyl protocatechuate came from efforts to tweak natural phenolic compounds and learn how slight molecular changes could shape their biological and physical properties. Through decades, interest has grown in the broader class of hydroxybenzoates as both nature-inspired molecules and as industrial resources. Ethyl protocatechuate stands out as a product born from this movement, often mentioned in connection with antioxidant studies, synthetic chemistry, and attempts to capture subtle benefits offered by modified natural molecules. Bench chemists, often motivated by curiosity or ongoing trials aimed at finding new preservatives or antioxidants, shaped much of what we know about this compound from the ground up.
Product Overview
Ethyl protocatechuate looks like a simple ester on paper, yet its ties to both laboratory research and industrial applications give it unique relevance. People who work with plant extracts, pharmaceuticals, and food additives may run into this molecule when reviewing antioxidant candidates or testing solubility enhancements in various formulations. Unlike bulk commodities, this compound often occupies a niche space, used most by researchers or small-scale specialists investigating antioxidant activity or developing chemical libraries for further screening. Its origin—derived from the condensation of protocatechuic acid and ethanol—serves as a reminder that even minor tweaks to core structures can have a measurable impact both in the lab and in practice.
Physical and Chemical Properties
Under standard conditions, ethyl protocatechuate forms white or off-white crystals. These tend to dissolve well in alcohol and organic solvents, but water solubility drops off. This trait becomes obvious on the bench: adding even a small volume to water yields only a cloudy suspension, yet it disappears quickly in ethanol. The molecule carries hydroxyl groups off a benzene ring, making it reactive under oxidation conditions yet fairly stable during storage if moisture and light are kept at bay. Its melting point tends to cluster around the moderate range—neither so low as to melt in the hand, nor high enough to resist simple purification steps such as recrystallization. In the presence of metals, the molecule chelates, creating complexes sometimes used in studies tied to metal-induced oxidative damage. These subtle chemical features guide how people approach handling, storage, and application.
Technical Specifications and Labeling
Bottles of ethyl protocatechuate often arrive labeled with purity ratings, typically above 98%. Chemists pay attention to appearance, noting a crystalline structure and checking for discoloration as an early sign of decomposition or contamination. Labels must list CAS numbers, molecular formula C9H10O4, and sometimes provide safety data to guide basic handling practices. The most diligent suppliers give extra analytical details—things like trace metal content or specification of residual solvents—especially if the compound is headed for use in food science research or high-sensitivity biochemical work. These technical details can spell the difference between a failed assay and a reliable result.
Preparation Method
Laboratory synthesis of ethyl protocatechuate typically involves reacting protocatechuic acid with ethanol in the presence of a strong acid such as sulfuric acid. Heating the reaction shifts the equilibrium and favors ester formation, and subsequent washing and extraction steps remove the acid along with leftover starting materials. Recrystallization in an appropriate solvent produces the purified product. The method has not changed much in decades, a sign of how direct and effective classical organic reactions remain for small molecules. Those who need this compound in greater quantities may lean on slightly larger reactors, but the basic chemical approach does not stray far from this textbook protocol. The simplicity of the process appeals to lab workers who appreciate both reliability and predictability in their synthetic steps.
Chemical Reactions and Modifications
Chemists use ethyl protocatechuate as a stepping stone toward more elaborate molecules. The aromatic ring with two hydroxyl groups on 3 and 4 positions of the ring and an ethoxycarbonyl group at the first position allows for selective reactions. Oxidation generates quinones, while reduction can impact the ester linkage or even the aromatic ring under harsher conditions. Protection or methylation of the hydroxyls creates derivatives used for probing enzyme functions, particularly in studies related to polyphenol oxidase. Nitration, halogenation, and metal chelation reactions expand the utility of the compound for use in both lab research and applied science. People in medicinal chemistry build libraries of analogs by modifying the ester or arene skeleton, hoping to discover new biological effects with medical or agricultural value.
Synonyms and Product Names
Those searching for ethyl protocatechuate in scientific catalogs see an array of names: ethyl 3,4-dihydroxybenzoate, ethyl PCA, and even a less common “ethyl protocatechuic acid” to describe this same structure. Sometimes, foreign labels use linguistic variations, adding to the long tradition in chemistry of redundancy and regional naming variance. Double-checking synonyms prevents confusion when buying or reviewing literature, especially across international borders or in interdisciplinary settings. Still, the CAS number 1910-42-5 slices through ambiguity, a practice that researchers—including myself—rely on to keep track of exact compound identity.
Safety and Operational Standards
In daily use, many chemists treat ethyl protocatechuate as a low-to-moderate hazard, following the same safety steps as for most low-molecular-weight aromatic esters. Gloves and safety glasses are standard, and good ventilation remains a smart precaution, especially during weighing or transfer. If spilled, it sweeps up easily, and accidental skin contact rarely leads to anything more than mild irritation. Regulatory standards set by European and American bodies guide what safety warnings should appear on bottles. Lab workers stay alert for fine dust that could irritate lungs. Over the years, a growing emphasis on documentation and chemical tracking gives an added layer of protection, further minimizing risk and promoting accountability for proper storage and disposal. From my own lab experience, simple protocols—prompt washing, labeling, and safe goggle use—prove their worth through countless routine operations.
Application Area
Ethyl protocatechuate finds use in more areas than a surface glance might suggest. Researchers rely on it as a reference antioxidant, testing its ability to scavenge free radicals compared to other food or drug additives. Its structure makes it a candidate for enzyme inhibition studies, especially in exploring polyphenolic effects on oxidases. Soil scientists have added it to experiment with plant-microbe interactions, hoping to mimic compounds found in plant roots. In industrial settings, this molecule occasionally plays a role in the stabilization of materials that suffer from oxidation, sprinkled into coatings, lubricants, and plastics in experimental formulations. Pharmaceutical researchers consider it a building block for prodrugs, adjusting its skeleton to optimize transport, solubility, or metabolic stability. Its relatively low toxicity (based on repeated animal studies) also draws attention in developing new preservatives for cosmetics or food, even though regulatory hurdles remain high.
Research and Development
Studies around ethyl protocatechuate keep popping up in searchable journals, especially where antioxidant performance is in spotlight. Groups working in nutritional science sometimes turn to this molecule for its activity against common reactive oxygen species, comparing its scavenging ability in freshly prepared cell cultures or model systems. Enzymologists, aiming to tease apart the intricate interactions at play in complex biological systems, use ethyl protocatechuate and its relatives to probe substrate flexibility and guide pharmaceutical design. A steady trickle of papers explores the antimicrobial and anti-inflammatory properties, trying to link modest lab results to broader medical or agricultural goals. From my viewpoint, the most interesting work happens at the boundary between chemistry and biology, where exploring why nature selects certain phenolic patterns can lead to both practical and theoretical discoveries.
Toxicity Research
Toxicity profiles for ethyl protocatechuate mostly reflect low hazard levels in traditional assays. Acute oral toxicity in rats points to high safety margins. Chronic studies still lag behind, especially at larger doses or longer exposure times relevant to environmental release or sustained human consumption. Genotoxicity screens almost always return negative, but as with many low-molecular-weight aromatics, the surveillance for subtle liver or kidney effects continues in regulatory and academic labs. Ecotoxicity remains an open field, as the compound’s tendency to biodegrade in soil and water draws questions about its fate and potential impact on non-target organisms. From my own lab experience, erring on the side of caution and following established disposal rules avoids most risks, but a disciplined research community always pushes for more data—especially as new application ideas filter in from commercial and academic sources.
Future Prospects
Looking ahead, ethyl protocatechuate seems primed for broader roles in biomimetic chemistry, drug design, and green technology. Interest in eco-friendly antioxidants for polymer and food industries runs strong, and molecules like this, which blend plant-inspired structures with direct synthetic access, hold value in that search. New delivery systems—think nanoparticles and encapsulation—may widen its reach as researchers try to harness its antioxidant traits in more targeted or sustained ways. In pharmaceutical research, prodrug strategies relying on slow hydrolysis of the ester group open prospect for precision dosing and metabolic fine-tuning. As environmental standards tighten, compounds with clean degradation profiles move higher on priority lists, further boosting interest. Based on current trends, I expect innovation will come from edges of academia and startup companies, where cross-disciplinary teams see opportunities where others see well-trodden ground. As someone who spends time at the crossroads of chemistry, biology, and materials science, I see ethyl protocatechuate as a symbol of that spirit—refined, revisited, and always ready for a fresh look in a new era of need.
What’s Behind the Name?
Ethyl protocatechuate doesn’t exactly roll off the tongue — most people have never heard of it. This compound pops up in scientific papers and specialty labs more often than on store shelves. Over the years, I’ve learned that obscure molecules like this one sometimes hold overlooked value. Some researchers discovered ethyl protocatechuate during efforts to understand natural antioxidants. It comes from protocatechuic acid, which can be found in foods like olives, grapes, and certain teas.
The Science: More Than Meets the Eye
Ethyl protocatechuate has drawn interest because of its antioxidant activity. Antioxidants slow down or prevent damage caused by free radicals. Free radicals can harm cells, and science ties them to all kinds of health problems, from heart disease to some cancers. I remember reading about how food researchers started looking for better ways to preserve fats and oils. Synthetic antioxidants worked, but there were worries about safety in the long run. So, many eyes turned to natural compounds like this one.
One study out of Asia pointed out that ethyl protocatechuate protected fats in food products better than a few well-known preservatives. That matters for food companies trying to cut down on synthetic chemicals in ingredient lists. Shoppers today tend to be wary when they spot unfamiliar preservatives, and that nudges companies toward more plant-based alternatives.
Medicine: A Glimmer of Potential
Outside the food world, ethyl protocatechuate gets attention from medical research. Specialists saw it could help counter oxidative stress, which plays a role in chronic illnesses, neurodegeneration, and even aging. In some lab tests, this compound helped defend nerve cells from damage. It doesn’t just promise broad “health benefits” — it targets the cellular processes driving real diseases.
Research teams in Europe tested it for broader uses, such as reducing inflammation or even helping antibiotics kill bacteria that normally resist treatment. These are test-tube findings for now, not green lights for everyday use, but they show a possible path forward.
Why People Should Care
Ethyl protocatechuate stands out as part of a bigger shift. Scientists keep pushing beyond traditional preservatives and drug additives, testing natural compounds for safer, more effective options. No consumer wants to trade taste, freshness, or shelf life for extra chemical risks. I see this compound as a key player in the search for safer food additives and pharmaceutical ingredients that won’t bring extra health baggage.
There are hurdles, though. Firms face steep research costs, and natural sources of the compound don’t scale up easily for mass production. If the demand for “clean-label” foods keeps growing, the industry will need to invest in new sourcing and synthesis methods. Funding more research and forming partnerships between universities and private labs can move things along. Choices from regulators and government agencies matter here, too, since clear rules are needed to get these compounds safely onto the market.
The Road Ahead
Ethyl protocatechuate isn’t some miracle cure or silver bullet, but it’s another example of how complex natural compounds continue to reshape what we eat and how we treat disease. Progress happens step by step — a new molecule adds another option, which helps companies and doctors offer safer and healthier products. Every move toward natural, well-studied additives keeps people better informed and more in control of what ends up in their bodies.
The Basics and Where It Turns Up
Food and health products pick up all sorts of additives and antioxidants these days, and Ethyl Protocatechuate pops up often. This compound isn’t some futuristic concoction. It comes from a simple origin: the esterification of protocatechuic acid, which itself has roots in edible plants. In practical terms, that means you’re unlikely to run across it in raw form at the grocery store, but it slides into processed foods and sometimes nutritional supplements.
Why Everyone Wants to Talk About Safety
Not every unfamiliar name means trouble. Still, healthy concern makes sense. The food industry leans hard on science for assurance, yet every so often, a new additive slips through before we know everything about it. Ethyl Protocatechuate gets attention because it’s been researched for its antioxidant abilities, which could help protect cells from damage. Scientists have looked into it for helping reduce inflammation or supporting certain body functions in animal studies, but humans aren’t giant rats, and biology isn’t that simple.
What the Science Actually Shows
Reliable safety data matters. In published studies involving rats and mice, Ethyl Protocatechuate didn’t set off alarm bells. At realistic doses, researchers didn’t find evidence of toxicity or alarming side effects in the animals’ organs. The compound broke down in a normal fashion, and the test subjects seemed to go about their rodent business without issue.
Still, human trials are missing in action. The scientific community has not yet lined up volunteers to swallow capsules full of Ethyl Protocatechuate for months. We just don’t have enough controlled, large-scale studies to call it completely safe for people. Food regulators in the United States and Europe demand thorough toxicity and metabolism research before granting a green light. At this point, Ethyl Protocatechuate isn’t sitting on any big industry-approved lists as a food additive, at least not with broad recognition.
Trusting Common Sense and Established Guidelines
Every one of us eats dozens of different chemicals every day—some added; some natural. Products already on our shelves often meet strict standards for what’s safe and what’s not. The trouble comes from the gaps that sometimes exist between new research and policy. History offers plenty of cautionary tales about things that seemed harmless at first. Saccharin, once touted as the miracle sweetener, faced years of health debates before settling into the public's trust. The lesson carries forward: don’t cut corners on safety, especially when people’s health is on the line.
What Steps Could Build Trust?
The clear path forward calls for more studies with real people. Long-term consumption results, details on how the compound is absorbed, and effects on sensitive individuals can turn curiosity into confidence. Transparency from anyone promoting Ethyl Protocatechuate matters, including publishing all findings—both good and bad.
People deserve plain answers about what goes into their food. Until large, peer-reviewed clinical trials on humans fill in the missing pieces, a careful approach wins. Healthcare professionals and regulatory agencies should share straight information and set limits based on the full picture, not just early optimism.
Looking Deeper Into Its Structure
Ethyl Protocatechuate holds a place that not many people talk about unless they're digging into natural compounds or food research. What you're dealing with here is a molecule made by adding an ethyl group to protocatechuic acid, which itself comes from the benzoic acid family. The chemical formula lands at C9H10O4. If you take a closer look, the backbone is a benzene ring with two hydroxy groups at positions 3 and 4, and an ethyl ester attached to the carboxylic acid group. In more relatable terms, this structure makes it an interesting cousin in chemistry's world of esters and phenolic acids.
A Structure That Punches Above Its Weight
Growing up with a keen interest in coffee and plant foods, I saw words like "phenol" get thrown around in textbooks. Ethyl Protocatechuate doesn't just look good on a molecular diagram; it shows up in real-world situations. You’ll spot esters like this in various fruits, teas, and fermented foods. Their use in the food system taps into questions of flavor, shelf life, and even health. The antioxidant power comes straight from those hydroxy groups on the benzene ring. Scientists have published about how this kind of structure helps stabilize free radicals, which shows why these natural chemicals matter much more than just numbers on a chart.
Importance Goes Beyond the Lab
In day-to-day life, food shelf stability and potential health benefits often trace back to tiny molecular changes like the switch from acid to ester. The ethyl group here might seem like small potatoes, but it shifts the way the molecule works in a living system. Stepping away from pure chemistry, food researchers keep exploring how such structures could help preserve food naturally, reducing the need for synthetic additives.
Medical and nutrition fields also pay attention because the two hydroxy groups signal antioxidant activity. Plants evolved with this kind of chemistry for natural protection. For anyone who cares about what goes into their food or wants alternatives to harsh food preservatives, ethyl protocatechuate and its chemical relatives play a part.
Solutions Rooted in Science
The next step lies in focusing on how these natural esters can find more space in food science and health technology. With antibiotic resistance and cancer still big threats, interest has grown in compounds from everyday food that might offer benefits. Not every natural molecule will be a magic bullet, but supporting more peer-reviewed research into how substances like ethyl protocatechuate work in living systems builds a stronger case for their use.
Another unique angle: encouraging transparent ingredient labeling. The more people know about what goes into processed foods, the easier it becomes to demand smarter choices from big manufacturers. Food and drug regulators should support research and policies that reward safe and effective use of natural compounds.
Rethinking Ingredient Lists and Priorities
Maybe the biggest shift will come at the cultural level. Deep dives into the nuts and bolts of natural compounds keep revealing the benefits of chemical complexity in plants. Instead of looking for one-size-fits-all solutions, encouraging innovation that respects both nature and evidence-based science feels like the right path forward.
Why Proper Storage Really Matters
People often focus on the flashy breakthroughs in chemistry, but few talk about the basics that keep research and industry running smoothly. Simple mistakes turn experiments sour and put people at risk. Talking about storing Ethyl Protocatechuate may not sound exciting, but this basic step protects everyone handling the stuff and keeps its quality intact. I’ve seen enough ruined batches and wasted resources to know that sloppy storage adds up to big costs and bigger headaches.
Understanding Ethyl Protocatechuate
This compound, used mostly for research or as an antioxidant, holds up well enough under the right conditions. The trouble starts once someone gets careless or assumes anything that “looks like a powder” will last no matter how it’s kept. Ethyl Protocatechuate, like plenty of lab chemicals, reacts to air, moisture, and light. All three chip away at its purity and strength. Skip the usual precautions, and it can clump, break down, or even spark unwanted reactions. Rarely do chemicals turn dangerous on their own, but surprises crop up once they're neglected on a lab bench or stashed in a damp corner.
Let’s Break Down Storage Basics
Behind the scenes, seasoned lab workers use a mix of common sense, experience, and basic science to avoid disaster. Keep it tightly sealed. Exposure to air knocks down its stability, drawing in unwanted moisture or oxygen. Small containers work better than large ones, since each opening reintroduces outside air. Use containers made of glass or high-grade plastic—ordinary jars invite trouble if they react with the contents.
Don’t park it near direct sunlight or heat sources—store it somewhere cool and dry, out of the sun’s reach. A shelf away from radiators, windows, and lab hot spots slows down harmful chemical changes. Labs that set up their own dry cupboards or keep desiccants handy see a lot less hassle with sensitive materials. If you’re serious, throw a silica packet in with the stock to absorb extra moisture. That small trick, taken from old-school research habits, consistently pays off.
Health and Safety: Not Just Paperwork
Some rules sound like overkill until you see what happens without them. Wearing gloves, keeping workspaces tidy, labeling everything—even the most experienced chemists double-check themselves. Accidents stack up from small lapses. Mislabeling or mixing up containers risks ruined results or worse, so it never hurts to check twice before putting chemicals away. Keep emergency information and safety sheets handy, both for storage and in case someone needs to clean up a mess.
How Teams Can Do Better
Teams that learn from each other catch mistakes early. In some labs, people swap tips and pass down better ways to handle tricky compounds. Regularly reviewing storage areas, replacing old containers, and tossing out compromised batches keep operations running clean. Anyone working with chemicals—students, researchers, or industry pros—benefits from a culture that values careful storage as much as groundbreaking discoveries. Investing in quality containers, setting up organized cabinets, and following up on best practices earns its keep every single day.
Building Better Habits
It’s easy to overlook proper storage in the rush of busy lab days, but the payoff shows itself in fewer spoiled chemicals, safer conditions, and smoother workflow. Taking the time to do it right, document your methods, and upgrade storage when needed reflects real respect for everyone sharing that workspace. In my experience, the labs that nail down these basics rarely have to scramble to fix preventable problems—their science always reaps the benefits.
Stepping into any laboratory that deals with antioxidants or looks at ways to slow down cell wear and tear, you’ll likely find researchers working with ethyl protocatechuate. This small molecule, an ester of protocatechuic acid, finds its way onto the bench in more places than folks might expect. From my own time in the lab, cracking the lid on a container of white, needle-like crystals was, more often than not, the start of an antioxidant test or a project tweaking the shelf life of natural extracts.
Kicking Off with Antioxidant Research
You can’t really talk about ethyl protocatechuate without mentioning its popularity in studies that aim to understand how substances handle oxidative stress. Many researchers turn to it for one simple reason: it packs a reliable antioxidant punch. In food labs exploring how to keep oils fresher for longer, this compound shows up again and again. A published study from the "Journal of Agricultural and Food Chemistry" points out that ethyl protocatechuate protects fatty foods from spoiling, which matters for producers who want to ship products farther or let them sit on grocery shelves that bit longer.
Nudging Natural Products Toward Better Health Benefits
Natural extracts—think herbal supplements or fortified foods—often fade in strength because they can’t stand up to exposure from air, light, or heat. Mixing in ethyl protocatechuate not only keeps flavors stable but helps lock in healthy compounds. Many of the scientists I’ve spoken to use it to compare natural protection with synthetic preservation. The compound consistently helps botanicals hold onto their original power, especially anthocyanins and flavonoids, which are hot topics in nutrition research.
Smoothing the Road for Drug Delivery
The field of medicine has a growing interest in safer, less harsh ways to improve drug uptake in the body. Ethyl protocatechuate shows promising results in preclinical studies because it guards sensitive molecules and boosts stability. A group at a medical research institute taught me about encapsulating some tough-to-handle drugs with this compound to steady them and target them more effectively. Clinical data isn't broad yet, but these early findings are hard to ignore. The molecule appears in patent filings for novel drug delivery schemes, reflecting both curiosity and cautious optimism among developers.
Fine-Tuning Cell and Tissue Studies
Cellular models and tissue samples don’t always survive stressful chemical treatments in lab settings. Ethyl protocatechuate, because of its antioxidant capabilities, has been tested to help rescue cells exposed to toxins or environmental challenges. In several recent papers, researchers mention using this compound to map out protective pathways inside human or animal cells. They want to know just how far you can push cells before damage sets in, and how much compounds like this can postpone trouble.
What Needs Fixing in Practice?
One challenge with ethyl protocatechuate comes down to transparency about sourcing and consistency. Some studies wind up with different results simply because two research groups end up with batches from different suppliers or under varying storage conditions. Regulating storage and handling, documenting provenance, and sharing standardized benchmarks in publications can boost reproducibility. In the wider industry, more real-world data about safety in complex mixtures such as foods or supplements would quiet doubts from both regulators and consumers.
The takeaway is clear: where there is need for preservative power and antioxidant research, ethyl protocatechuate continues to be a proven, if somewhat underappreciated, tool that bridges the gap from bench science to commercial kitchens and manufacturing lines.