Ethyl Trifluoroacetate: A Closer Look at Its History, Uses, and Where It's Going
Historical Development: From Curiosity to Critical Reagent
Chemical history always has stories of overlooked reagents that later find their stride. Ethyl trifluoroacetate belongs here, first catching the eye of chemists exploring halogenated compounds in the 20th century. The early years focused on what trifluoromethyl groups could bring to the table, especially once folks noticed their impact on chemical reactivity and stability. As research ramped up in pharmaceuticals and material science, this compound carved out a home for itself—no grand marketing push, just good old-fashioned laboratory success. Looking back, the route from bench curiosity to trusted synthetic tool mirrors the broader journey of organofluorine chemistry in modern science.
Product Overview and Properties
A clear liquid, ethyl trifluoroacetate tends to have a sharp, fruity odor that sets it apart from most run-of-the-mill esters. Its chemical backbone, built on a trifluoroacetyl group paired with an ethyl ester, gives it unique properties. You won’t find it mixing neatly with water, but solvents like diethyl ether and chloroform attract it with ease. Melting and boiling points are respectable, sitting comfortably between everyday solvents and more exotic reagents. Unlike substances that fizz away under mild conditions, this compound holds steady in the bottle and in the flask, resistant to slow hydrolysis in the face of basic and even acidic media—though not indestructible, either.
Technical Specifications and Labeling
Labeling for ethyl trifluoroacetate tends to warn of volatility and reactivity. It comes with the expected flammable warnings and precautionary statements, and the industry usually ships it in brown glass bottles to protect it from light and stray hands. Purity levels above 97% are standard in research and industry supply. Not packed with fillers or stabilizers, its purity matters more than it might for other esters, since trace contamination can disrupt delicate syntheses.
Preparation Method
The synthesis of ethyl trifluoroacetate can get a bit technical but often relies on esterification of trifluoroacetic acid with ethanol by acid catalysis, sometimes using sulfuric acid as a driver. Alternatively, factories may use transesterification or other methods, but the key is controlling temperature and removing the water byproduct to boost yields. These reactions demand care—too much heat, and you get side-products or hydrolysis. In my own experience, running the reaction under dry, inert gas keeps the process smooth, and using a Dean-Stark trap to remove water as it forms beats having to troubleshoot muddy mixtures later.
Chemical Reactions and Modifications
Ethyl trifluoroacetate really shines as a building block for further chemistry. The trifluoroacetyl group can slide onto nucleophiles under both basic and acidic conditions. The high electronegativity of fluorine atoms pulls electron density away, activating the carbonyl for nucleophilic attack. Chemists rely on this characteristic to introduce trifluoromethyl groups into pharmaceuticals, agrochemicals, and advanced materials. You’ll often find it used for protective group strategies on amines during peptide synthesis, since it’s relatively easy to introduce and later cleave off with base or acid. In organic synthesis, it acts as more than just a source of fluorine—its ester function brings flexibility to protecting-group techniques, which can make or break a complex synthesis.
Synonyms and Product Names
Chemical catalogs and research publications list ethyl trifluoroacetate under several other names—ethyl 2,2,2-trifluoroacetate and trifluoroacetic acid ethyl ester show up the most. The CAS number remains the universal ticket for procurement and regulatory compliance. In plenty of labs around the world, just saying “trifluoroacetate ester” gets the point across, showing its well-established place in synthetic chemistry.
Safety and Operational Standards
While the compound’s volatility isn’t out of control, you won’t see it handled without gloves, goggles, and solid ventilation. It reacts with water and base, so storage in tightly sealed bottles far from basic solutions is non-negotiable. I’ve had more than one bottle turn if left open to air and moisture, reminding everyone not to treat “routine” reagents as harmless. Regulatory standards echo lab experience: local laws set storage and waste requirements, and international safety data points out the low-to-moderate acute toxicity. The main risks come from inhalation of vapors and possible irritation—the classic organofluorine story. Standards usually recommend using fume hoods and not relying on intuition when handling larger amounts.
Application Area
Ethyl trifluoroacetate finds its sweet spot in synthetic methods where trifluoromethyl groups add metabolic stability or alter bioactivity—something medicinal chemists value. It features in the production of antiviral drugs, pesticides, specialty polymers, and even photographic chemicals. Its ability to protect amines efficiently appeals to those working on peptide and protein-based drugs. In material science, the electron-withdrawing power of the trifluoroacetate group can push electronic effects and color in organic electronics.
Research & Development
Interest in ethyl trifluoroacetate continues to grow, especially as researchers chase new drug candidates and advanced materials. Studies focus on improving yields and finding greener synthesis methods. Catalysis and flow-chemistry offer promising routes—less waste, tighter control, and lower temperatures. From what I’ve seen, every major leap in pharmaceutical pipeline development now examines fluorinated building blocks, often circling back to reagents like this one for key transformations. With more regulations on emissions and safety, the research community pays close attention to handling and disposal, pushing for closed-loop systems and effective recycling protocols.
Toxicity Research
Concerns around toxicity of fluorinated esters have grown. Animal studies suggest relatively low acute toxicity but raise red flags about prolonged exposure. Researchers examine metabolite profiles to see if trifluoroacetate acids accumulate in biological systems. Some studies note that excessive exposure can irritate skin, eyes, and respiratory tracts. Evidence so far doesn’t tie it to chronic high-level toxicity, but prudent operators take precautions anyway, echoing the larger debate over fluorinated chemicals and their fate in the environment. Many labs now run periodic monitoring of air quality when using the compound, especially during scale-up.
Future Prospects
Ethyl trifluoroacetate will not fade from view anytime soon. With growing demand for more stable pharmaceuticals and electronic devices that push the limits of material science, chemistry needs the kind of subtle influence that trifluoromethyl groups provide. Research into bioisosteres—a way to substitute functional groups without losing activity—puts this compound in the spotlight for early-stage drug discovery. Sustainability remains a challenge; future developments will likely chase renewable feedstocks, lower-impact processes, and enhanced recovery after use. The next decade might see more efficient catalysts and broader use in fields not yet tapped, as the value of fluorinated chemistry continues to expand.
A Key Ingredient That Shapes Modern Chemistry
Ethyl trifluoroacetate might sound like a name you only hear in a research lab, but it plays a far bigger role outside of white coats and safety goggles. This compound shows up across fields that shape everything from life-saving drugs to the fabrics that keep us dry. Taking a closer look, you find its impact stretches far past a storeroom shelf.
The Building Block in Pharmaceuticals
Pharmaceutical chemists lean on ethyl trifluoroacetate when trying to build complex molecules. It acts as a “protecting group,” which means it shields sensitive parts of a molecule during big chemical reactions. By keeping certain atoms out of the way, scientists avoid unwanted side reactions and keep their focus on the actual target. In my own job, I watched colleagues use it to develop a product for a rare enzyme disorder. They struggled with chemical “side-tracks” until they turned to this compound, which let them finally connect the right pieces.
Fluoro-based compounds also show up a lot in modern drug discovery. Adding fluorine atoms can help drugs last longer in the body and do their job more efficiently. Since ethyl trifluoroacetate brings that fluorine punch, it gives drug designers more control and flexibility over the final product’s function.
Industry Uses Beyond the Lab
Big chemical plants don’t just use this compound for medicine. Producers of herbicides and other crop chemicals rely on it for similar reasons: building complex molecules without accidentally wrecking key parts. Agrochemicals need both effectiveness and environmental safety. Ethyl trifluoroacetate helps chemists shape these products while keeping harmful by-products to a minimum.
Beyond spraying fields, specialty plastics and materials bring this molecule into their mix. The trifluoroacetyl group that comes from it makes certain coatings tougher against moisture and chemicals. Rain jackets and electronics sometimes depend on coatings derived from fluoro chemistry to last longer and work better. Even a few drops of the right chemical can have a ripple effect on how something stands up to daily life.
Why Purity and Safety Need Attention
It’s not all smooth sailing with any reactive chemical. Ethyl trifluoroacetate demands careful handling. Vapors from improper storage or accidents send workers scrambling for ventilation and protective gear. In poorly-ventilated labs, the irritation can hit quickly. From my time shadowing a plant supervisor, I saw how regular air checks and clear safety procedures kept small mistakes from becoming scary emergencies.
Every user down the supply chain benefits from cleaner, purer ingredients. Supply chain managers need to check their sources, not just for quality, but for responsible handling. Tighter training and labeling can help avoid the kind of slipups that lead to contamination or exposure.
Balancing Innovation With Responsibility
Chemists and engineers face constant trade-offs: how to push boundaries without putting people or the environment at risk. Smart training, clear oversight, and next-generation safety gear hold a lot of promise in this field. Using compounds like ethyl trifluoroacetate with respect lets us keep pursuing new cures, stronger materials, and greener solutions without letting short-term mistakes cost us in the long run.
A Closer Look at the Formula
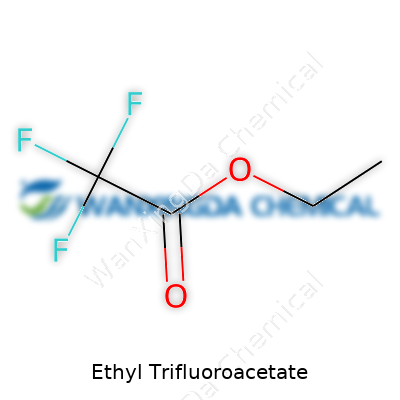
Ethyl trifluoroacetate doesn't make its way into everyday conversation unless you spend your days in a lab coat. The chemical formula—C4H5F3O2—carries more utility than it might seem. It stands for a molecule with four carbon atoms, five hydrogen atoms, three fluorine atoms, and two oxygen atoms. In my work with organic synthesis, the trifluoroacetate group gives this compound its punch. Swapping out regular hydrogens for fluorines changes the way molecules behave: volatility, reactivity, and resistance to breakdown all spike.
Real-World Roles of Ethyl Trifluoroacetate
For decades, chemists have relied on trifluorinated reagents to drive reactions that introduce fluorine into larger molecules. Fluorine atoms create drugs and agrochemicals that last longer, avoid breaking down in a week, and sometimes slip past bacteria that would chew up a less-protected molecule. One example: the active ingredients in some modern antibiotics stick around and work more effectively thanks to fluorinated building blocks in their synthesis.
Ethyl trifluoroacetate offers a route to those building blocks. In pharmaceutical chemistry, every shortcut matters, especially if it avoids toxic or costly reagents. I remember working with similar compounds and seeing firsthand how a single trifluoroacetate group made the purification stage less messy and cut down on harsh waste. That's a win not only for lab safety but for the environment, which always ends up with the leftovers from industrial chemistry.
Learning from Risks
There’s no sugarcoating the challenges that come with handling fluorinated chemicals. Fluorine doesn’t play nice—accidental contact through skin or lungs can sting or worse. That being said, most modern firms prioritize equipment that vents these vapors and reduces spills. I’ve seen labs move to closed-system synthesis over the years, making mistakes less likely and reducing personal risk. It’s not only experienced chemists who benefit. Students and interns working with these compounds get a first-hand lesson in why safety rules aren’t just bureaucratic boxes to check.
Environmental Consciousness Matters
Few topics rile up environmental chemists quite like the long lifespan of fluorinated compounds in nature. These molecules don’t just disappear. If released, they stick around for years, show up in water sources, and can build up in wildlife. This isn’t hypothetical. Monitoring studies in the last decade traced a rise in persistent organofluorine contamination across North America and Europe. As someone who has watched the rise of “green chemistry” approaches, I see the push to invent ways to break down or recover fluorinated residues growing every year.
The lesson from industry and academia: chemical innovation comes with a responsibility to look past the lab. Better disposal strategies and more research on biodegradation should move higher up the priority list. Some younger researchers bring a fresh approach, testing out new catalysts or bacterial strains that might speed up what nature would otherwise take centuries to accomplish.
Finding a Balance
Ethyl trifluoroacetate sits at a crossroad between utility and risk. Getting the benefits of easier molecule building and stable drug ingredients means grappling with hard questions about safety and waste. Based on years in and out of labs, I believe the next leap comes from blending chemistry with smart engineering and oversight. Learn what these molecules can do—but keep an eye on where they end up when the work is done.
The Real Stakes in Chemical Storage
Anyone who has spent time in a lab knows there’s more to storing a bottle of chemicals than sticking it on a shelf and slapping on a label. Ethyl Trifluoroacetate isn’t some backyard solvent—it packs enough punch to demand serious respect. Handling and storage habits, if sloppy, create quiet risks that can turn loud and dangerous.
Get to Know What You're Dealing With
Working with Ethyl Trifluoroacetate has taught me that this colorless liquid doesn’t throw tantrums, but it does sneak up on you. Consider its volatility and the way it reacts with water or strong bases—forgetting about its reactive nature, even briefly, can cause more than just minor headaches. The fumes catch you first: acrid, unpleasant, and no joke if your ventilation stinks.
Temperature and Environment: Where Small Choices Matter
I learned quickly that warm, humid storerooms just don’t cut it. Flammable liquids like Ethyl Trifluoroacetate don’t belong anywhere near heat sources. I’ve seen more than one lab ignore this rule, with containers sweating in sun-drenched windows and refrigerators jammed with who-knows-what. Storing it cool and dry isn’t some fussy academic rule—it’s a practical way to lower risks. Think under 25°C, away from direct sunlight or radiators, and humidity far from pipes prone to leaks or bursts.
Keep It Sealed, Keep Your Peace of Mind
My first lab gig drilled this point into me: always seal containers tight. Ethyl Trifluoroacetate fumes aren’t just harsh—they spread quickly and irritate eyes and airways. Once, a coworker left a cap loose; the smell hung around, and the whole team started coughing before we tracked it down. Tight caps and vapor-tight containers make a noticeable difference, especially after hours of handling volatile liquids.
Compatible Storage Only
It isn’t enough to throw all your chemicals on the same shelf, hoping for the best. I’ve seen too many storerooms with acids, bases, peroxides, and flammables jammed side by side. Ethyl Trifluoroacetate reacts with strong bases and water, so it belongs with other organic solvents, well away from those troublemakers. Store it with chemicals bearing similar risk profiles. Segregation isn’t bureaucracy—it’s about dodging irreversible mistakes.
Ventilation—The Overlooked Hero
A cramped, stuffy storage cabinet smells bad and breeds trouble. Ventilated cabinets work wonders for flammables, and Ethyl Trifluoroacetate benefits from fresh air just as much as any acetone or ether. I trust a cabinet with a working exhaust system; it means I don’t walk into a toxic fog every time I open the door. That’s about protecting everyone in the building, not just whoever put the bottle away.
Training Counts
No list of safety protocols beats common sense. Every lab tech or chemist needs training—on the risks, the symptoms to watch for, the first aid steps. Accidents drop off when people know what they’re dealing with. That includes clear signage, up-to-date safety data sheets, and regular checks of storage conditions. The best labs run safety drills and keep emergency gear within arm’s reach; that’s not overkill, it’s knowing what’s at stake.
Making Storage Safer: A Collective Effort
What’s at stake stretches far beyond compliance and paperwork. I’ve watched labs that take these steps—cool temperatures, segregated shelves, sealed containers, proper ventilation—work for decades without incident. The difference comes down to culture and vigilance, not just rules. Storing Ethyl Trifluoroacetate with care reflects respect for everyone in the lab and the communities nearby.
Respecting Chemistry in Real Life
Ethyl trifluoroacetate tends to show up where chemists want to add a trifluoromethyl group, especially during specialty syntheses in pharma and fine chemicals. I remember working alongside chemists who valued this chemical for its reactivity, but even the most skilled among them treated it with caution and a healthy dose of respect.
Why Vigilance Is Non-Negotiable
Breathing easy in the lab quickly feels like a luxury if someone cracks open a bottle of this stuff without proper precautions. Ethyl trifluoroacetate gives off vapors that don’t just irritate the eyes and lungs — repeated exposure can linger in your body, and the risk isn’t worth rolling the dice. Common sense goes a long way, but here it’s not just a guiding principle; it stands between routine work and a trip to the occupational health nurse.
Simple Steps: Protecting Skin, Eyes, and Lungs
Anyone who’s spent hours in the lab learns early that gloves and goggles are non-negotiable. Nitrile gloves perform much better than latex when dealing with this ester. Splashes happen — even to the cautious — and nobody wants trifluoroacetate burns or chronic irritation. A lab coat, not just for show, provides a solid layer of defense and keeps your street clothes clean.
Ventilation matters. I’ve seen close calls from haphazard use on an open bench, the sharp scent wafting through the room and setting off headaches almost right away. Fume hoods keep those vapors out of your lungs and prevent the chemical from becoming everyone else’s problem. It’s about respecting the shared workspace as much as protecting yourself.
Storing with Some Foresight
A shelf in the corner or a cluttered bench undermines all your safety habits. Sealed containers and clearly labeled bottles store this stuff reliably. There’s no heroism in wrestling with a stuck cap; damaged bottles leak fumes, so careful handling while opening and closing is the only way to go. I’ve had bottles leak after being left half-opened — that quick fix leads to nasty surprises down the road.
Waste and Spills: Mistakes Happen, So Plan Ahead
A spill on the bench can seem minor until you realize how quickly those fumes fill the air. Spill kits save the day, but only if you actually know where to find one. Neutral absorbents work; paper towels don’t. Tossing waste or used rags in the regular trash only shifts the risk. Label hazardous waste and coordinate proper disposal — no need to gamble with downstream safety.
Bigger Picture: Culture of Safety
Regulations from OSHA and REACH don’t just appear on paper; they reflect lessons learned the hard way. Training refreshers, clear signage, and a willingness to slow down help everyone avoid learning those lessons firsthand. Trusting your nose or eyes for early warning isn’t enough, since vapors don’t always give fair notice before effects set in.
Safe habits around ethyl trifluoroacetate mean less time worrying about what could go wrong. Focusing on basics each day grants time for more meaningful work in the lab, and as someone who’s seen shortcuts backfire, I don’t miss a chance to remind newcomers that in chemistry, caution isn’t optional — it’s how you earn your next experiment.
Understanding What We Work With
Ethyl trifluoroacetate turns up in labs everywhere, from pharmaceutical research to chemical synthesis. People sometimes overlook the grade of a reagent, but there’s no shortcut here. Purity means more than just getting good yields—it’s about trust in your results and knowing exactly what goes into your reaction flask.
Seeing Beyond the Label
Most bottles of ethyl trifluoroacetate will list a purity percentage. Typical lab-grade batches hover around 97% to 99%. Some suppliers push for 99% or higher, flagged as "analytical grade" or "high purity." That sounds pretty good, but those numbers don’t tell the whole story. Purity ratings depend on the test methods used. Two bottles labeled 99% can still vary because each company watches for different common contaminants.
Minor impurities may seem harmless, but a stray bit of moisture or a leftover reactant from synthesis can knock a reaction sideways. I’ve seen colleagues lose weeks of work tracing strange side products back to an unnoticed impurity. Chromatography and NMR may only spot the big players; smaller contaminants may sneak through until they show up unexpectedly.
Where it Hits Hardest: Research and Production
Anyone running sensitive reactions—especially in medicinal chemistry or materials science—wants reagents that don't invite surprises. Impurities change reaction selectivity, introduce false positives, and scramble reproducibility. In drug development, this means more than a mild headache. Researchers rely on the documented purity to meet regulatory and safety requirements, especially with products destined for clinical work.
Labs working on scale-up face an even stricter world. FDA guidelines in the US, and EMA rules in Europe, push for transparency in trace materials down to parts per million. You can’t fudge a batch with unclear origin—records, certificates of analysis, and validation reports pile up to protect everything downstream.
Chasing Purity and Accountability
Problems are clear, but so are the ways forward. Anyone buying ethyl trifluoroacetate should insist on a detailed certificate of analysis. This means not just a single percentage, but a rundown of contaminant limits and test methods used. An open conversation with suppliers pays off—don’t just accept the standard “high purity” label. Ask if there’s batch data available, and find out if extra distillation or purification was used.
It helps to work with suppliers that invest in documentation and traceability. Some companies back their products with transparent supply chains and frequently updated testing protocols. Open access to these details builds trust and helps labs avoid unpleasant surprises.
Taking Responsibility in the Lab
Checking purity may seem like an annoying extra step when budgets and deadlines pile up. But experience teaches you to put care up front. Test small samples when possible, and compare supplier data with in-house verification. In the long run, reliable chemistry depends on checking the groundwork. Bad reagents waste time, money, and brittle goodwill with collaborators.
If you don’t know what you’re starting with, everything else becomes uncertain. That lesson sticks, whether you’re chasing a complicated target molecule or just trying to build a reputation for good science. In the end, purity means trust—not just in the product, but in your own work.