Ethyl Trifluoromethanesulfonate: An Exploration Beyond the Bench
Historical Development
Ethyl trifluoromethanesulfonate, sometimes referred to among chemists as “ethyl triflate,” has earned a reputation for its reliability in lab syntheses. The journey to today’s use started with the early days of organofluorine chemistry in the mid-20th century. Back then, researchers started noticing that triflate-related compounds could open doors that felt closed in conventional organic synthesis. The first discussions of ethyl triflate appeared in foundational fluorine chemistry journals, with researchers tweaking sulfonate groups and exploring the electrifying effect of CF3 clusters. Each tweak and experiment chipped away at barriers, pushing chemists closer to compounds that could deliver direct, clean transformations. That’s the kind of progress that only comes from patient, sometimes messy, hands-on bench work. Chemists quickly recognized the unique reactivity triflates could offer, especially in building complex molecules for pharmaceuticals and advanced materials.
Product Overview
Ethyl triflate doesn’t belong in the collection of casual chemicals. It’s an elite tool for anyone who’s ever faced that stubborn substrate and needed a real problem-solver. At its core, ethyl triflate pairs a robust ethyl group with a trifluoromethanesulfonate group, making it both reactive and selective. Its sheer power as an alkylating agent comes from this union, making it useful for adding ethyl groups under mild conditions in ways that other reagents rarely manage. A classroom textbook may glide over its importance, but in industry labs or academic projects shooting for something big—like the next blockbuster medicine—ethyl triflate emerges as a vital player.
Physical & Chemical Properties
Open a bottle of ethyl triflate and you’re faced with a clear, almost colorless liquid, though you’ll often keep it sealed since it hydrolyzes quickly if given a chance. Its boiling point sits above what you find in most simple ethers, thanks to the stubborn, electron-rich triflate group. The smell doesn’t announce itself as loudly as some other organofluorine substances, but that doesn’t make it welcoming. Density and solubility can trip up chemists unaccustomed to working with perfluorinated compounds. More importantly, its high electrophilicity brings both opportunity and risk, amplifying its reactivity far beyond standard alkyl sulfonates. This strong alkylating nature gives it a certain danger; its solutions must be handled with unwavering attention.
Technical Specifications & Labeling
Bottles of ethyl triflate rarely sit unmarked on any shelf. Labels come printed with serious warnings: corrosive, lachrymatory, and moisture-sensitive. Even a seasoned chemist feels the pressure to double-check their PPE before opening a fresh sample. Certificate of analysis data for each lot highlight purity that rarely dips below 98 percent, since even a small impurity can throw off key reactions. Batch numbers, shelf-life estimates, and production details stack up next to international transport icons, reminding everyone in the chain to respect the substance. The message remains clear: Ethyl triflate commands the room. It asks users to show skill and discipline from receipt to disposal.
Preparation Method
Everyone who’s tried to prepare ethyl triflate in a glassware-packed fume hood knows the challenge. Traditional lab methods build it from ethyl alcohol and trifluoromethanesulfonic anhydride. You mix the two under dry, chilled conditions, almost always using a non-nucleophilic base such as pyridine to soak up any stray protons. These reactions don’t tolerate sloppy technique; water ruins yields. Large-scale makers enforce tight process control, watching reactor temperature and drying agents like hawks. Solvents must be distilled and dried again. The margin for error shrinks as production scales up. Each successful synthesis feels like a small victory against entropy.
Chemical Reactions & Modifications
The crowning achievement of ethyl triflate’s chemistry is its unmatched ability to alkylate a wide range of nucleophiles. Take an alcohol or an amine that shrugs off weaker alkylating agents—ethyl triflate swoops in, delivering rapid, nearly quantitative conversions. Chemists lean on these properties to build out carbon-oxygen or carbon-nitrogen bonds quickly and under milder conditions than older sulfonate esters could manage. It takes part in substitution reactions where even a slight variation in reactant reactivity can send yields plummeting. Triflate esters, including ethyl triflate, have powered the development of valuable medicinal molecules, electrolytes, and advanced polymers, all thanks to their robust, predictable reactivity. Attempts to tame or modify ethyl triflate often center on tuning its leaving group properties for selective modifications, particularly as the synthetic toolkit expands to include more complex, functionalized systems.
Synonyms & Product Names
Step into any synthetic lab, and you might hear “ethyl triflate,” “ethyl trifluoromethanesulfonate,” or even “trifluoromethanesulfonic acid ethyl ester” used interchangeably. Catalogues list all these names together, helping avoid confusion. The standard CAS number attaches accountability to distribution and purchase, keeping things transparent for regulatory bodies and scientists looking for consistency. Despite the varied terminology, chemists quickly learn to recognize not just the names but the distinctive bottle shapes and warning labels sitting in the cold room.
Safety & Operational Standards
A good day in the lab relies on respect for ethyl triflate’s hazards. Gloves, goggles, lab coats, and a fresh change of nitrile gloves become as regular as morning coffee. Fume hoods aren’t optional. Experience teaches you that a splash or a stray puff of vapor means more than an inconvenience—ethyl triflate reacts with skin and eyes, so any exposure means a trip to the safety shower. Spill protocols get drilled into every new lab member, because it won’t wait for indecision. Waste streams collect triflate-contaminated materials in specially labeled containers destined for professional neutralization. Regulatory agencies in North America, Europe, and Asia demand strict adherence to safe handling, waste management, and documentation. Companies face audits and researchers update protocols, searching for improvement in worker protection. Each generation of scientists learns from the accidents of the last.
Application Area
Ask synthetic chemists what alkylating agents they trust for demanding jobs, and ethyl triflate makes the list. In the search for new medicines, triflate-derived intermediates sometimes unlock scaffolds that were off-limits before. Medicinal chemistry departments in pharma houses lean on ethyl triflate for rapid exploratory synthesis. Its value carries over to specialty polymers, where it helps introduce precise functional groups that tailor the conductivity or stability of emerging materials. Beyond pharma, research in energy storage materials—lithium-ion batteries and novel electrolytes—often circles back to compounds in the triflate family for their thermal and chemical stability. Some graduate students spend years exploring new applications, always circling back to the reactivity and selectivity that ethyl triflate delivers. Its performance in precision jobs, despite the hazards, keeps it relevant in an evolving technological landscape.
Research & Development
The world of research never sits still. Chemists continue to look for ways to harness ethyl triflate’s power with fewer risks. Labs run experiments on alternative synthesis routes using renewable resources, hoping to decrease the environmental footprint. New reaction protocols focus on minimizing solvent use, incorporating innovations in solid-phase synthesis, or finding recyclable reaction partners. Some efforts look toward integrated process chemistry, where triflate chemistry meshes with flow reactors to improve safety by minimizing the volume of hazardous materials in play. Computer modeling and simulation try to predict side products or new applications, streamlining the trial-and-error that defined earlier decades. Research teams submit papers and patents, hoping their tweaks translate into cleaner, safer, or more cost-effective chemistry. The constant debate between performance and safety pushes the field forward.
Toxicity Research
No story about ethyl triflate has credibility without facing up to toxicity. Lab accidents over the years, documented in safety bulletins and medical journals, have driven demand for better risk data. Animal studies and careful human exposure monitoring provided much of the current understanding, pointing to severe eye and skin damage, respiratory irritation, and the risk of organ effects from chronic or high-level exposure. Regulatory frameworks shape how it’s stored, used, and discarded, and experienced chemists treat it as a potential hazard even when numbers look manageable. Institutions fund ongoing animal and cellular research, probing for long-term effects and potential metabolites. Lessons from accidents create new procedures and inspire improvements in personal protective gear.
Future Prospects
The fate of ethyl triflate ties directly to trends in synthesis and industrial chemistry. On one hand, modern labs push for greener, less hazardous reagents; on the other, the years have proved there’s no substitute for its performance in some applications. If future generations of chemists develop milder reagents capable of the same alkylation feats, ethyl triflate’s dominance may shrink. For now, ongoing research in risk reduction—like encapsulation, flow chemistry, or robotic handling—offers a path for safer use. Increased regulation and demand for sustainable alternatives have produced a steady trickle of research into similarly reactive but less toxic triflates, and attention also shifts to developing less persistent, more biodegradable options for industrial settings. Whatever shape the chemical marketplace takes, the story of ethyl triflate stays a case study in how innovation, risk, and necessity can shape the toolkit of modern science.
I’ve come across plenty of odd-sounding chemicals during my years in the lab, and Ethyl Trifluoromethanesulfonate—often called “ethyl triflate”—ranks up there with the tongues twisters. Despite the name, the function is straightforward: it acts as a strong alkylating agent, one of those rare chemicals that can stick a short carbon chain onto tricky molecules. In everyday terms, it helps build other chemicals, ones that end up as medicine, advanced materials, or even the electronics many folks rarely think twice about.
Piecing Together New Medicines
Science doesn’t move forward without a lot of trial and error. Cancer drugs and antiviral compounds, for instance, don’t spring up from thin air. Chemists experiment, tweak a compound’s structure, swap out this bit for that, always hunting for a better result. Ethyl triflate steps into this process like a construction worker with a power tool: it makes carbon-oxygen bonds much easier to build, which can transform a dull backbone into a promising treatment.
Some drugs, especially those in the nucleoside family—used to fight viruses or cancer—rely on these specialized bonds. Adding fluorine atoms, as ethyl triflate allows, changes how these molecules act in the body, often boosting stability or allowing them to target cells with more precision. That’s not theory—that’s how lifesaving treatments come to market.
Electronics and Advanced Materials
Flip over your phone or take a look at the screen you’re reading right now: tiny circuits made of specialized plastics and electronic parts make these gadgets work. Chemistry underpins every bit of it, and that includes the use of ethyl trifluoromethanesulfonate. Fluorinated materials, as odd as they might seem, help create plastics stable enough for high heat, strong enough for advanced uses, and sometimes slippery enough for non-stick coatings. Companies use ethyl triflate during the manufacturing process to build these polymers, giving society smaller, faster, and more durable technology.
Challenges for Safety and Sustainability
No one in the lab shrugs off the risks. Ethyl triflate packs a punch. Direct exposure comes with hazards for skin, airways, even the environment. Safety rules matter here. Fume hoods, gloves, and strict waste protocols become daily routines, because nobody wants an accident or an unchecked spill. In my own work, paying close attention during use separated safe days from those filled with regret.
On the sustainability front, the chemical industry faces real pressure. People want safer processes and products that break down after use. Some companies now try to minimize or recycle materials during manufacturing, lessening the environmental footprint of strong alkylating reagents. Research into greener alternatives continues, but for certain applications, ethyl triflate still delivers results no substitute can match.
Looking to the Future
For students and young scientists, learning about reagents like ethyl triflate offers a clear window into the constant balancing act in chemistry: innovation paired with responsibility. It’s not some spooky “big chemistry” ingredient—it is one cog in a vast machine turning basic building blocks into breakthroughs. As safety practices and sustainable methods catch up to the science, maybe we’ll find new ways to harness these powerful tools without sharp tradeoffs. Until then, ethyl triflate quietly powers the discoveries behind medicine, technology, and materials found in daily life. That’s worth paying attention to.
Working with Something This Reactive Changes the Game
People working in chemistry labs get used to handling some tricky substances, but ethyl trifluoromethanesulfonate can catch you off guard. It doesn’t take much—just a splash or a shift in air—to turn an ordinary lab day into something memorable for all the wrong reasons. What sets this compound apart isn’t just its reactivity, but the invisible dangers it brings. Speak to anyone who’s ever dealt with strong alkylating agents, and you’ll hear the same thing: respect this chemical, or regret it.
The Risks Go Beyond Skin-Deep
Breathing vapor from ethyl trifluoromethanesulfonate can put your lungs in trouble. Even a tiny spill can burn skin, and it punches right through gloves that work fine against less active solvents. Sometimes the biggest risk isn’t dramatic; it’s slow and quiet—like long-term lung or nerve damage from repeated, low-level exposure. The body doesn’t bounce back from this stuff. Chronic exposure creeps up on you in a way that safety goggles won’t fix. I’ve watched folks lose sense of smell after years in the wrong environment, thinking their practices were fine because nothing happened last week.
Experience Teaches that Shortcuts Don’t Pay Off
Let’s be honest, most chemists think about protective equipment, but there are days when everyone feels tempted to skip the uncomfortable gear, especially during a fast procedure. Over time, you realize those shortcuts don’t just invite burns and blisters. The real cost shows up in mistakes—the sudden burst of toxic vapor that triggers an evacuation or the drop of liquid that burns a hole in someone’s sleeve. Fume hoods and face shields can get in the way, but choosing convenience over safety can end a career in the blink of an eye.
What Makes Real Safety?
No gear beats thinking ahead. Working with ethyl trifluoromethanesulfonate gets safer when you slow down, check labels twice, and never transfer this liquid outside a well-ventilated hood. Pick gloves that resist aggressive agents; standard latex isn’t enough. Face shields help, especially if you ever need to charge a reaction or split batches. Emergency showers and eyewash stations don’t just hang on the wall to collect dust—they turn what could be a life-changing injury into a minor scare if someone reacts quickly.
The best labs don’t treat safety measures as nuisances. Good teamwork means having a buddy on standby during the riskiest steps, so one person never gets left dealing with a splash or a runaway reaction alone. I’ve seen lab culture shift in places where people call each other out for not snapping elastics or for ignoring a fresh glove tear. Real oversight doesn’t come from manuals—it lives in people reminding each other to do things right each time, no matter how small the task seems.
Better Culture, Fewer Accidents
This isn’t about filling out extra paperwork or sitting through more seminars. The safest labs encourage folks to speak up if they’re unsure or stretched thin. If someone’s working late, tired or distracted, everyone understands the risk jumps way up. Pausing a reaction to check on one another can keep an ordinary error from spiraling out of control. No chemical reward matches the relief that comes from everyone making it home healthy.
Chemistry pushes the boundaries of what people can do, but there are some lines nobody can cross safely—ethylene trifluoromethanesulfonate draws that line bright and clear. The more openly people talk about the risks, the safer everyone gets. If there’s one thing worth borrowing from seasoned chemists, it’s the idea that no experiment is really successful unless everyone walks away unscathed.
The Formula Behind a Powerful Reagent
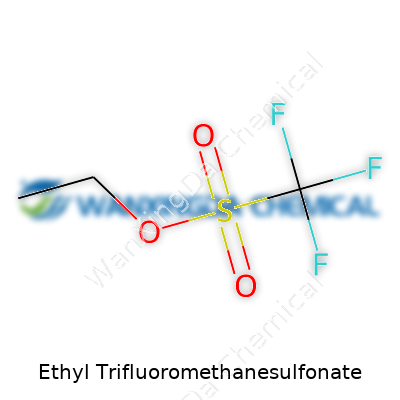
Ethyl trifluoromethanesulfonate. Hearing the name alone calls chemistry classrooms and lab benches to mind, maybe even that rush of uncertainty that comes from handling strong reagents. To understand its reach in the world of organic synthesis, looking at its raw formula paints a clear picture. Chemists refer to it as C3H5F3O3S. Decoding that sequence tells you so much about what it's capable of, and frankly, how it demands respect in any experimental setting.
This compound is built from an ethyl group—two carbons and the corresponding hydrogens—linked to a trifluoromethanesulfonate, or triflate, as folks in the lab often call it. Structurally, it stacks up like this: CH3CH2OSO2CF3. Drawing it out, the ethyl unit connects through an oxygen to a sulfur sprinkled with three highly electronegative fluorines. The triflate segment is well-known for making excellent leaving groups—something you never forget if you've tried pushing sluggish nucleophilic substitutions across the finish line. If you’ve ever dealt with a stubborn substrate, you know exactly why having a transformative ingredient like this makes all the difference.
Why It Matters for Synthetic Chemistry
In labs focused on building molecules from scratch or tweaking sensitive architectures, ethyl trifluoromethanesulfonate becomes a go-to actor. Its reputation as an “ethylating agent” grows from its ability to transfer the ethyl group to other molecules efficiently, especially in conditions where milder reagents would barely get a reaction started. Over my years dabbling with stubborn aromatic systems and quirky heterocycles, getting a clean conversion often feels like winning a small lottery—and hitting it right almost always involves this reagent when other options get ruled out.
Chemists value this stuff not just for its high reactivity, but also for how its triflate group readily departs, leaving behind the desired transformation. It doesn’t matter if you’re tackling a tricky nucleophile or working in the presence of long chains and fragile bonds—the compound finds a way to operate in spaces many simpler chemicals just can’t. In the world of pharmaceuticals, agrochemicals, and specialty polymers, streamlined synthetic routes and efficient processes rest on the shoulders of compounds like ethyl trifluoromethanesulfonate.
Handling, Hazards, and Heavy Responsibility
The story isn’t all smooth sailing. Flip open a chemical safety manual and you’ll see bold warnings about this reagent’s lachrymatory nature and strong irritant risk. If you’ve ever caught a whiff of it escaping from a bottle, you won’t soon forget the sharp sting. It deserves thoughtful respect in the lab, since improper handling introduces real dangers to eyes, lungs, and skin. These aren’t minor annoyances—they force anyone in the lab to slow down and act with care, using gloves, fume hoods, and tightly controlled procedures.
Moving Forward: Safer Science, Smarter Synthesis
Finding ways to replace such hazardous reagents isn’t just an academic exercise. Green chemistry initiatives push researchers to develop newer, friendlier ethylating agents that trade brute force for finesse. Inline detection systems, safer substitutes, and better ventilation have started turning hazardous steps into manageable ones. Sharing these workarounds and improvements, from one research team to another, makes the whole process safer and more reliable for the next chemist who picks up a bottle.
There’s No Shortcut With Serious Chemicals
Walk into any chemistry lab, and you’ll spot bottles marked with bold hazards. Ethyl trifluoromethanesulfonate—often called triflate—comes with more warnings than most. Why? This isn’t just another bottle tucked on a shelf. Triflate packs a punch: it’s a powerful alkylating agent, easily reacting with moisture, skin, and just about anything unprotected. I’ve watched new researchers freeze in panic when confronted with a leaky stopper or an unexpected whiff escaping from a flask. There’s no shrugging off these risks.
Moisture: The Unseen Enemy
Triflate’s biggest foe is water vapor. The reaction kicks off instantly, leading to decomposition and sometimes flammable byproducts. Nobody wants a cloud of potentially hazardous fumes floating through the lab. Keeping the chemical bone-dry isn’t just a detail; it’s the difference between a smooth experiment and a reportable incident. For years, I’ve seen experts insist on airtight containers, using glass with secure Teflon seals—no rubber stoppers, no cracks, no exceptions. In some labs, triflate sits inside a desiccator alongside drying agents, locking out every drop of humidity.
Cool and Dark: Not Just for Fancy Wine
Warm storage speeds up unwanted reactions. Sunlight and bright overhead lights raise the risk by supplying the energy triflate spices up to react. For this reason, chemical refrigerators—not the regular office fridge—keep the temperature steady and low. Forget the office break room: lights stay low, the door’s opened as little as possible, and inventories run tight. More chemicals go bad from rough storage than from anything else.
Separate It—For Peace of Mind
Accidents love company. Crowd strong acids, bases, or oxidizers together, and you’re inviting trouble. My first glance at a high school stockroom burned in my memory for all the wrong reasons: incompatible substances shoved side-by-side, the air tinged with something ‘off.’ Real labs take storage compatibility seriously. Triflate usually gets a dedicated spot far from acids, bases, anything nucleophilic, or chemicals with active hydrogen. Shelves carry clear labels, and containers never migrate for convenience. Emergencies demand clear separation—risking cross-contamination isn’t just a rookie mistake, it’s dangerous.
People Over Protocols
Too many see the protocol but miss the reason behind it. I’ve picked up habits from cautious mentors and sharp-eyed colleagues: never open the triflate bottle outside a fume hood, double-check the tightness on every seal, watch for any leaks. Fast hands create slow disasters. Teaching these habits matters, especially to new scientists. No one forgets a close call.
Safe Storage, Sustainable Research
Good storage practices boil down to respect—for the chemical and for the people working around you. Conscientious scientists document every movement, run regular checks, and update labels at the first sign of wear. It’s not about getting in and out of the stockroom faster. It’s about heading home in one piece and building trust in the lab community.
Ethyl trifluoromethanesulfonate isn’t friendly, but with care, it doesn’t need to be a source of stress. Store it cool, dry, isolated, tightly closed, and always under the watchful eye of someone who understands what’s at stake.
Direct Contact Isn’t Just an Annoyance
Ethyl trifluoromethanesulfonate rarely pops up outside high-level labs, but for anyone dealing with it, safety can’t be just a checkbox. Splash a bit of this stuff on your skin and you’ll feel the burn almost right away. One colleague, tough and usually unflappable, ended up with a hospital trip after working late and skipping gloves for “just a minute.” The irritation didn’t show up immediately—it crept in, left him nursing sores days later. If it gets near your eyes, the risk escalates to permanent damage. You can flush your eyes, but the sting sticks around.
Breathing Fumes Is a Gamble
Labs might have alarms and vents, but most don’t notice how easily fumes can spread. Ethyl trifluoromethanesulfonate gives off vapors that stress your lungs and throat, leading to coughing, sneezing, and more serious respiratory trouble over time. A slip-up with the ventilation left the air thick enough to send a friend home with a tight chest and migraine. Even with face masks, one whiff stands out—a sharp, chemical bite that lingers far too long.
Real Worry: It Doesn’t Just Go Away
This chemical isn’t the sort to just sit in the corner. Ethyl trifluoromethanesulfonate reacts with water, producing fumes that quickly spread and worsen hazards. A single botched cleanup can mean ongoing exposure for the whole workspace. I’ve watched floors stay slick with residue for hours, leading to slips and continued contamination risk. Tidy habits and fast action make the difference here; a missed droplet multiplies the threat. In addition, any spill often becomes a larger headache because it can combine with other common chemicals and create byproducts you don’t want anywhere near your lungs or skin.
Long-Term Impact Lurks in the Details
The real issue grows over time. Repeated, low-level exposure doesn’t always knock you down today, but it adds up. Some chemicals force you to take them seriously after a single mistake. With ethyl trifluoromethanesulfonate, the risk sits in the background. Repeated breathing or skin contact might damage airways or cause skin to crack and heal poorly. I’ve spoken to older chemists who can trace small, lingering health complaints back to their early, less-careful years.
Turning the Tide: What Actually Makes a Difference
Relying on basic gloves or thin lab coats won’t cut it here. Thick nitrile gloves or even heavier butyl options are the go-to. Never skip over eye protection; splashes happen fast. Solid air extraction systems backed by regular checkups keep vapors from circulating. Supervisors have to set the tone—no skipping procedures for convenience.
Training matters more than manuals gathering dust on a shelf. Labs that run regular drills and use real-life scenarios break through complacency. Reporting tiny leaks or splashes without fear of blame keeps everyone safer. Some chemists bring personal stories to orientation, and that resonates better than diagrams or wall posters.
Care Means Counting Every Exposure
It’s tempting to dismiss small exposures, especially on busy days. Still, every encounter chips away at your health, and the compounding effect can’t be overstated. Avoiding shortcuts, insisting on regular checks, and using the right protective tools stop this chemical from setting off a chain of health issues. Experience—hard-earned by many—shapes every safe lab.