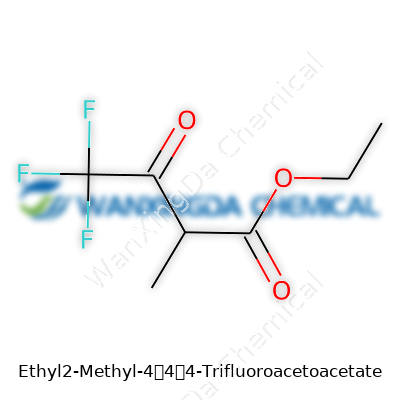
Ethyl 2-Methyl-4,4,4-Trifluoroacetoacetate: More Than Just a Chemical Intermediate
Historical Context and Origins
Looking back at how new molecules change the course of whole industries, the story of Ethyl 2-Methyl-4,4,4-Trifluoroacetoacetate feels like a small lesson in modern chemistry’s ambitions. Chemists have spent decades pushing ketone and trifluoromethyl chemistry into every corner of pharma and materials science. A century ago, the idea of selectively dropping a trifluoromethyl group onto an acetoacetate backbone would have sounded outrageous, bordering on science fiction. Not long after big names like Swarts and Simons introduced reliable ways of making trifluoromethyl compounds in the early twentieth century, research labs began moving beyond fundamental halocarbon synthesis into more analytically tricky and industrially promising esters, acids, and ketones. There’s no single “discovery moment” for this compound, but it traces its roots to the broad push for new building blocks and reagents once fluorine’s influence in organic synthesis became plain. What drives this field has always been less about making novelty molecules and more about giving hands-on chemists new handles on fluorine’s wild set of properties—powerful, sometimes nerve-wracking, always in demand for something.
Product Overview and Real-World Use
For someone working in synthesis, Ethyl 2-Methyl-4,4,4-Trifluoroacetoacetate stands out as a practical intermediate. Its role isn’t glamorous, but it’s the kind of molecule that opens the door to tailored pharmaceuticals, fine chemicals, and crop protection agents. Similar trifluoromethylated esters lend extra muscle to drug research because they help slip fluorine into drug candidates. Fluorine changes a molecule’s behavior on every level—making it more stable in the bloodstream, helping drugs resist digestive breakdown, skewing lipophilicity, even just dodging troublesome enzymes. Many bench chemists know this molecule either as a starting material for a medicinal project or as something you order for a custom synthesis. The companies who make it know the demand is shaped by practical research, not mass commercial processes. In labs, you find bottles labeled with CAS numbers, ready for small-scale or pilot work that lives at the borderline of chemistry and biology.
Physical and Chemical Properties
The structure of Ethyl 2-Methyl-4,4,4-Trifluoroacetoacetate brings together a set of properties you don’t see every day. Its clear, sometimes faintly yellow liquid form hints at volatility and the need for thoughtful handling. You can smell many esters, but the trifluoromethyl group gives this one a sharpness and resistance to easy breakdown. The high density compared with hydrocarbon esters and its notable boiling point carve out a distinct “feel” when pipetting, weighing, and mixing. In solution, it shows the push-and-pull of electron-withdrawing CF3 groups up against an active methylene site. The molecule stands tough against hydrolysis at room temperature, but acid or base shifts change things quickly. In my experience, this ester feels like it’s living in two worlds—it’s tough enough for industrial settings, but reactive enough that a chemist can modify it in a hundred different directions.
Technical Specifications and Labeling
Labeling for this ester follows a cautious pattern because of the trifluoromethyl group's potentially hazardous nature. Good practice means paying attention to percent purity, acidity, moisture content, and even trace metallics, since pharmaceutical and crop scientists have little patience for impurities slipping into their products. In the lab, attention turns to lot traceability and secure containers, often amber for light sensitivity. The global shift toward detailed digital documentation—barcode scanning, electronic notebooks—takes on extra weight when hazardous materials like trifluoromethyl esters stack up in the fridge. Operationally, every bottle comes with warnings about vapor and splashes, and it makes sense—this isn’t the molecule you want loose on your bench.
Preparation Methods
Among the multiple routes to making Ethyl 2-Methyl-4,4,4-Trifluoroacetoacetate, most rely on the availability and cost of trifluoroacetate sources and specialized alkylation chemistry. Typically, synthesis flows from trifluoroacetic anhydride or acid chloride joined with enolate chemistry, tacking on the methyl group at the alpha position in the presence of strong base. Sometimes, direct condensation reactions with trifluoroacetoacetate salts and alkylating agents simplify the process, but skilled hands and good temperature control make the difference between a clean crude product and a mess of by-products. Most chemists prefer mild acid work-up and low temperatures for isolation, since heating can break down the product or fill the flask with volatile fluorinated side-products. Small labs cut their teeth on batch reactions, while big facilities reach for flow processes or continuous reactors to handle scale with less waste and more reproducibility.
Chemical Reactions and Modifications
The real value of this ester shows up in how it can be pushed and pulled to build new frameworks. Its acidic hydrogen at the methylene site, sandwiched between two carbonyls, begs for alkylation, condensation, and coupling. The trifluoromethyl pulls electron density, stabilizing the resulting enolate and helping drive selectivity in follow-up synthesis. Chemists use this to push the molecule toward more complex heterocycles, sometimes trying to mimic bioactive structures or develop completely fresh scaffolds for pharma leads. The ester’s chemistry supports both nucleophilic addition and electrophilic substitution, and the CF3’s bulky, electronegative influence stubbornly resists reduction or other harsh conditions—a double-edged sword depending on the project. You might see this molecule transformed into pyrazoles, pyridines, or fluorinated alcohols, all with dramatically different properties but still bearing the fingerprint of that original trifluoromethyl “engine.”
Synonyms and Other Product Names
Chemists are rarely satisfied with a single name for anything, and this ester is no exception. The literature abounds with synonyms, mostly derivatives on the IUPAC backbone or historical nomenclature that highlights the functional groups. You might see it called Ethyl trifluoromethyl-β-methylacetoacetate, or simply as the corresponding acetoacetate with trifluoromethyl and methyl substituents noted in the formula. Sometimes, supplier catalogs opt for shorter, sharper names—trifluoroacetoacetate esters—with or without explicit mention of the methyl group, unlocking confusion unless the CAS number backs up the bottle. This patchwork of naming becomes a headache in international research or regulatory discussions, where getting everyone on the same page about which molecule sits on the bench makes the difference between success and waste.
Safety and Operational Standards
My hands-on experience with trifluoromethyl compounds demands respect for their hazards. Vapors from esters like this carry risks to eyes and lungs, and the molecule’s volatility can flood a room if protocols fall short. Safety data sheets warn against skin exposure and recommend fume hoods, gloves, and goggles at minimum. The trifluoromethyl group isn’t just an inert passenger; when mishandled, it can break down to stubborn, difficult-to-remove residues or even fluorinated fumes. Waste handling becomes tougher than with many esters, often requiring specialized disposal or even incineration at facilities equipped for fluorinated waste. Clean-up after a spill is more complicated; standard detergents sometimes fail, and catching residues early stops them from making their way into the environment or the next reaction mixture. Institutional safety culture—regular training, clear signage, and well-maintained workspaces—protects not just researchers but everyone downstream in the waste cycle.
Practical Application Areas
Pharmaceuticals draw the biggest share of demand for this ester, due to fluorinated compounds’ tendency toward metabolic resilience and unique biochemical effects. It’s a favored intermediate or building block in the hands of medicinal chemists searching for ways to tweak potency, bioavailability, or target selectivity of new drugs. Outside drug discovery, agrochemical scientists test it for making herbicides or insecticides that survive sunlight and rain, with tailored breakdown profiles to avoid long-term soil contamination. Even specialty polymer companies find value in its structure, especially for introducing trifluoromethyl functionalities that toughen up coatings or improve weather resistance. These practical needs are driven by a demand for performance, safety, and intellectual property protection, as the molecular fingerprint of this ester can support patent claims and regulatory filings. For every batch sold into a research program, there’s usually a story of trial, failure, and sometimes world-changing results, especially if a pharma team finds an unexpected path to a safer or longer-lasting medicine.
Research and Development Directions
Ethyl 2-Methyl-4,4,4-Trifluoroacetoacetate sees constant study, mainly because every decade brings new tools for making, modifying, or analyzing fluorine-containing molecules. Researchers now harness high-throughput screening, automated synthesis, and advanced spectroscopic techniques to explore faster, cleaner, and greener preparation. In universities and pharma research centers, attention focuses on how modifications at the methyl or trifluoromethyl positions steer biological effects or metabolic fate. There’s heavy cross-talk between computational chemistry, which models molecule-enzyme interactions, and bench chemistry, which validates activity against real targets. As the landscape shifts—especially with rising concerns about sustainability and green chemistry practices—scientists press for less harmful reagents, lower temperatures, and minimal-waste syntheses. The challenge remains: squeezing out maximum performance from each molecule without leaving an environmental mess for future generations.
Toxicity Studies and Environmental Impact
Hundreds of fluorinated molecules have earned a reputation as persistent environmental threats, and every new ester or ketone demands fresh scrutiny before large-scale use. Toxicity studies for this ester hint at acute irritation to eyes, skin, and lungs, and its breakdown products stir up extra concern—especially in wastewater streams. Chronic exposure data lags behind, mainly because widespread industrial use has yet to push this compound into the regulatory spotlight, but caution is the rule rather than the exception. The trifluoromethyl group’s stability means just a small spill or leak can linger in the environment, possibly slipping past traditional filtration or biodegradation. My own experience with similar materials shows that institutional pressure for pre-registration and monitoring grows louder every year, and international treaties on persistent organic pollutants could one day lump compounds like this together for stricter controls. Until then, chemists and regulatory teams need sharp eyes on both acute and long-term risks, feeding data into shared, open-access toxicity databases to close the knowledge gaps.
Looking Ahead: Future Prospects
Rising demand for safer drugs, sustainable agriculture, and tougher specialty materials keeps research on this ester moving forward. If new, even more efficient synthetic routes unlock cheaper or greener production, usage will skyrocket—not just in high-value pharma, but wherever the unique push from a trifluoromethyl group is needed. Emerging research promises clever catalytic cycles, electrochemical activation, or enzyme-based synthesis that might turn this intermediate from a specialized lab rarity into a broader staple of chemical manufacturing. Of course, every expansion brings fresh questions about long-term health effects or environmental fate, and researchers, regulators, and industries must work together, balancing innovation against responsibility. As knowledge grows, open communication and rigorous safety culture will make the difference, letting us build a world where fluorine’s power works for us, not against us.
(Why This Odd-Sounding Molecule Matters)
A name like ethyl 2-methyl-4,4,4-trifluoroacetoacetate doesn’t roll off the tongue, and it sure doesn’t line grocery store shelves. This mouthful grabs the spotlight inside laboratories and research facilities, nudging forward both everyday products and some of the breakthrough ideas in pharmaceuticals. For chemists, this building block makes certain reactions workable that otherwise would stall. In real life, small compounds with big names quietly shape drugs for chronic illness, pest controls that keep crops healthy, and specialty materials in electronics.
Where It Finds Its Niche
Chemists prize this molecule mainly as a reagent—a starting point for bigger, more valuable molecules. Take pharmaceuticals. The trifluoromethyl group, which means three fluorine atoms hung on a carbon, brings a handy trick: It keeps drugs stable in the body and can help medicines last longer or work better. Drug development chases these results, since slight tweaks in a molecule can boost how drugs target disease. For years, researchers have looked for new molecules that carry fluorine because they often improve drug performance. Ethyl 2-methyl-4,4,4-trifluoroacetoacetate steps in during the earliest days of research, providing a backbone for medicines ranging from antivirals to treatments for high blood pressure.
Out in the wider world, agriculture also benefits. Many active ingredients in crop protection draw from the chemistry of fluorinated acetates. Adding trifluoromethyl pieces can change how these products behave in soil and plants. Crops get a fighting chance against pests, and farmers keep yields healthy. This connection matters because it points to my own background, growing up in a rural town where crop cycles kept families steady. Safer, more effective controls mean more stability at harvest season, and families may not notice which molecule delivered that edge, but growers sure appreciate it.
Specialty materials—think polymers or flame-retardant coatings—tap into the promise of trifluorinated ingredients, especially where durability counts. If you’ve ever wondered why some coatings shrug off stains or chemicals or why electronics sometimes resist moisture better, it’s because someone worked behind closed doors with molecules like ethyl 2-methyl-4,4,4-trifluoroacetoacetate. These improvements trickle down to the finished product most folks take for granted, giving tougher, longer-lasting materials.
Safety and Environmental Concerns
Reliance on fine chemicals always reawakens questions about responsible development. Nobody wants to see a promising product put out into the world without thinking about its impact. Fluorinated chemicals seem to stick around—a blessing in a medicine, a problem if they wander into water supplies or soil. I remember cases where improper handling led to regrettable outcomes, which brings up the importance of regulation and responsible stewardship. Science keeps inching forward on methods to break down or safely use these chemicals, but the conversation isn’t over.
Looking Toward Solutions
If chemists can keep shifting processes greener and safer while maintaining the benefits these molecules bring, future generations can enjoy the advances without the risks. Industry already began changing how large-scale synthesis happens, using less wasteful methods or swapping in safer solvents. Stronger dialogue between chemical companies, regulators, and communities helps. In practice, transparency from industry and a bit more curiosity from the public can nudge things forward. Where people remember that the small, invisible compounds matter, change follows.
Chemicals Don’t Lie: Real-World Lessons in Storage
Working with chemicals like Ethyl 2-Methyl-4,4,4-Trifluoroacetoacetate isn’t only about fancy glassware or clever reactions. Lab veterans will agree: what happens in a flask depends as much on how you treat your chemicals when they’re on the shelf as anything you might do at the bench. In my years in the lab, I’ve watched good experiments flop just because someone turned a blind eye to how a bottle was stored. People love to think chemistry is magic; in truth, good storage is the real trick.
Keep It Sealed—Moisture and Oxygen Are the Enemy
Ethyl 2-Methyl-4,4,4-Trifluoroacetoacetate brings trifluoromethyl power to organic synthesis, and that means it can be touchy with air and water. Pop the cap, and you invite problems. Open bottles don’t only pick up water vapor; they can pull in air, which often doesn't end well. Water and oxygen can kick off slow, silent reactions. Sometimes the liquid yellows or develops a haze you didn’t see last week. Trust me, pour an eyedropper’s worth into a flask and you’ll know if you’re not working with the pure stuff anymore.
Labs use desiccators and dry boxes for a reason. I learned early that if you leave any ester, especially one with fluorinated bits, in humid air, you pay the price. You can't always see the change in purity right away, but analytic testing like NMR or GC will flag it down the line. It’s easy to say, “I’ll just recap the bottle and get back to work,” but it’s a shortcut nobody enjoys later.
Temperature Matters More Than Labels Suggest
Temperature trouble sneaks up on you. Most organofluorine esters like cool, steady conditions. Most lab fridges run at 2-8°C, which works for many reagents. In my own practice, I used to get lazy—shelving bottles on open storage just to keep my bench tidy. Later, I’d find out the reagent didn’t work the way it did last month. Chemical companies don’t always tell the full story on labels, but those who keep these kinds of compounds cold don’t deal with separation, gassing, or weird smells. Moving the bottle to the fridge, keeping it away from light and heat, fixed a lot more failed syntheses than any glossy purification scheme.
Cleanliness Is Preventative, Not Optional
Residues and dirty spatulas are a recipe for heartache. Shared lab spaces invite temptation: the urge to dip a spatula in and out, cap off or on. I’ve been guilty myself. Cross-contamination always looks minor at first. Small mistakes—putting contaminated tools into the bottle, not wearing gloves—invite unintended breakdown products. No one wants to track impurities all the way through to a failed column or an unexpected TLC spot. Good technique starts with simple routines—clean tools, fresh gloves, and avoiding double-dipping—so the reagent can do its thing.
Why It Matters
Sloppy storage doesn’t just waste chemicals; it wastes time and science. Down the road, someone will have to explain a failed project or an experiment that can't be reproduced. For synthesis or R&D labs, the hidden impacts—like extra purification steps or lower yields—can bust budgets and timelines. An open discussion about storage gets everyone on the same page, so fewer people need to learn the hard way. That’s what keeps the science honest—and the discoveries coming.
Understanding the Details
Getting down to the nitty-gritty, Ethyl 2-Methyl-4,4,4-Trifluoroacetoacetate isn’t the kind of name that rolls off the tongue at the dinner table. Yet, scientists and those in the chemical industry bump into it more often than you’d think. Imagine building a house without knowing the number of bricks you need or their size. That’s what it’s like trying to use a compound without knowing its chemical formula or molecular weight. For this compound, the chemical formula is C7H9F3O3, and the molecular weight clocks in at roughly 198.14 g/mol.
Why It Matters
Even if you don’t scan chemical catalogs for fun, there’s a reason to care about the nuts and bolts of molecules like this. They form the backbone of much of what makes up everyday life — pharmaceuticals, agrochemicals, and materials science. Knowing exactly how many atoms are strung together in a compound can help chemists predict reactions or even decide if an experiment is safe in the first place. I remember trying to synthesize a related compound back in college and missing a single fluorine atom in the formula threw off my calculations. The product didn’t yield as expected, teaching me early about the domino effect one mistake can have.
On a bigger scale, miscounting carbons or hydrogens doesn’t just mess with graduation projects; it can lead to incorrect dosing in drug research or misjudgments in commercial scale-ups. That’s not just a lab headache. It turns into regulatory headaches, safety issues, and wasted resources. The consequences of sloppy formula work sneak out of labs and into production lines.
What’s the Bigger Picture?
You’ll see chemicals like Ethyl 2-Methyl-4,4,4-Trifluoroacetoacetate as stepping stones. They often act as intermediates — ingredients that don’t make it into the final product on the shelf, but without them, nothing gets built. The trifluoromethyl group attached to the molecule makes it more than just another acetoacetate derivative. That group changes the way the molecule behaves. It can influence boiling points, reactivity, and solubility. For drug developers, that means tweaking one group can open doors to new biological properties or improve how a substance gets absorbed by the body.
There’s no one-size-fits-all approach in chemical development. Understanding molecular weight helps chemists calculate how much of a compound to mix, and gives clues about how it’ll act under different conditions. Without this data, predictions morph into stabs in the dark. I’ve seen small research groups waste precious grants chasing down synthesis paths that could have been ironed out with simple formula cross-checking.
Solutions for Smarter Chemistry
So how do we get things right more often? Consistency starts with detailed documentation and reliable software. Tools like ChemDraw or open-source alternatives offer a second pair of eyes to catch simple mistakes. Training matters, too. Young scientists need hands-on experience — recalculating formulas, cross-verifying molecular weights — as part of their routine. Open access to chemical databases would level the playing field, giving labs without deep pockets the same information as big players.
At the end of the day, every formula written on a chalkboard or lab notebook is bigger than the sum of its numbers and letters. It’s a bridge between raw ingredients and real-world solutions — from safer medicines to better-performing materials. Getting that formula and its weight right is where it all starts.
Life in the Chemistry Lane
Spend any time around a chemistry lab, and long, tongue-twisting names eventually become part of the landscape. Ethyl2-methyl-4,4,4-trifluoroacetoacetate is one of those unwieldy terms that often pops up in synthesis work. Beyond the syllables, though, lies a question plenty of folks outside chemistry circles have: Is this stuff hazardous or toxic? It's a fair concern. The reality is that chemicals with a complex structure and fluorinated carbon atoms usually come with risks that anyone—from researchers to industry workers—would be wise to understand.
What the Research Shows
Dig into available studies or material safety guidance, and you’ll see some common themes. Ethyl2-methyl-4,4,4-trifluoroacetoacetate tends to irritate skin and eyes. Maybe not surprising, given how its parent group—trifluoroacetoacetates and similar esters—has a reputation for being harsh if you get sloppy handling them. Direct contact rarely goes unnoticed, causing redness, itching, or outright burns in the worst cases.
That story doesn’t end with touch, either. Breathing in vapor during transfer or cleaning up a spill can hurt airways. Coughing fits, sore throats, headaches—they all show up in records from exposed workers. Ingestion, intentional or accidental in a lab mix-up, risks effects ranging from stomach pain to more serious disruptions with prolonged or high dose exposure. The data on chronic toxicity is still pretty thin, but with fluorinated carbons, caution makes sense since persistent and bioaccumulative chemicals often bring surprises later.
Regulatory Oversight: What Exists, and What Gaps Remain
Big agencies like OSHA and the European Chemicals Agency carefully track most industrial chemicals, and this compound’s group doesn’t escape attention. Safety data sheets warn against contact, urge proper ventilation, and point professionals to lab coats and goggles. Fire risks rise in concentrated vapor or around open flames, and warnings about environmental release reflect the broader trend of regulators waking up to persistent organic pollutants.
Problems crop up less from the molecule than from the sometimes casual attitude people take toward “routine” chemicals. Some labs still treat these materials like minor irritants, not worthy of real protocols, only to regret it after a spill lands someone in the health unit. Upgrading safety culture doesn’t require fancy gear—just respect for what even small volumes can do if ignored.
Practical Steps Forward
The safest labs I’ve worked in put good habits above everything else. Label everything, keep proper ventilation in place, and make goggles non-negotiable. Any time a new researcher marches in, clear training beats wishful thinking. Emergency showers and quick access to clean water make a huge difference when accidents happen. Chemical fume hoods really aren’t optional, especially with volatile and reactive chemicals.
Waste shouldn’t go down the drain or in regular trash. Local and national disposal rules matter, and tracking containers pays off when regulators check on compliance or when trying to retrace a bad batch back to its source. At the end of the day, simple respect for chemical risk, guided by experience and clear regulatory advice, makes the difference every time.
Looking Out for Each Other
Hazardous potential doesn’t exist just in academic debates or regulatory boards. It shows up when someone forgets gloves while cleaning glassware or isn’t paying attention while transferring reagents. With compounds such as ethyl2-methyl-4,4,4-trifluoroacetoacetate, understanding and acting on risks—backed by facts, not just routine—keeps people safe for the long haul. That keeps both science and scientists moving forward, with no unwelcome surprises.
Why Purity Matters in Chemical Work
Lab work draws a clear line between trial and error and genuine discovery. Ethyl 2-methyl-4,4,4-trifluoroacetoacetate, a mouthful for sure, plays an important role in making pharmaceutical intermediates, specialty polymers, and sometimes even agricultural chemicals. Purity shapes everything from reactivity to safety, so ignoring it means inviting surprises downstream. A chemist who trusts an impure batch might scratch their head when reactions stall or impurities show up in finished product testing.
Reliable Specifications: What Does Purity Actually Mean?
A good supplier’s technical sheet, if they choose transparency, will often list purity upwards of 97% or sometimes even reaching 99%. This comes as a result of careful distillation and analytical testing. The stakes run high, because even half a percent of the wrong material can alter results, create toxic byproducts, or waste precious time retracing steps. For research, a slightly lower purity sometimes passes muster if trace contaminants fall outside regions of concern, but with scale-up or regulated manufacturing, almost nothing but top purity cuts it.
Talking to colleagues in synthesis, nobody wants to gamble with questionable chemicals. The supply chain’s grown even trickier as manufacturing drifts across borders. Trust gets harder to come by, so most experienced chemists demand a certificate of analysis with every shipment. This serves as a paper trail, documenting the exact degree of purity by techniques like gas chromatography, nuclear magnetic resonance, or mass spectrometry.
Challenges in Maintaining Purity
Purity only tells half the story. Shelf stability can erode over time if moisture creeps in or if the bottle ends up under the wrong light at the back of a cabinet. The other trouble comes from invisible residues—solvents that linger or similar fluorinated compounds that barely show up until someone digs through the analytical data. Good chemical hygiene means labeling reagents with open dates and storing them in cool, dry spaces. Small steps like this head off trouble long before an experiment fails.
Practical Steps for End Users
Folks using this compound should start by asking for clear documentation on lot-specific purity every time they order. If uncertainty lingers, a double check at an in-house lab or by a trusted third-party testing company can save huge headaches. I’ve seen teams troubleshoot for weeks, only to discover that the culprit wasn’t their technique—it was a tainted batch of starting material.
There’s power in community, too. Sharing notes with others who buy from the same sources helps root out issues early. And on the supplier side, strong relationships and candid feedback can prompt improvements or recalls before small problems snowball.
Looking Ahead
Progress depends on vigilance and clear information. Keeping a close eye on what goes into projects—whether making new drugs or testing materials—means asking tough questions about source, storage, and handling. The stories I hear from old-school chemists echo that lesson: skimping on purity lets chaos creep in, while attention to detail keeps science moving forward.