Heptafluorobutyric Acid: The Unseen Force in Modern Chemistry
Tracing the Roots: Historical Development
Back in the day, heptafluorobutyric acid (HFBA) didn’t start off as a standout player in the world of chemistry textbooks. Its roots tie closely to the broader exploration of organofluorine compounds. In the mid-20th century, chemists set out to tackle the shortage of analytical reagents—HFBA caught interest because of its deep fluorination, offering both strong acidity and resistance to breakdown under tough lab conditions. Older research journals show that as chromatography spread across the pharmaceutical and environmental sciences, folks needed acids that wouldn’t gunk up their machines or react unpredictably. That’s where HFBA began showing its worth, outlasting earlier analogs, carving out a spot as both a niche and mainstream tool by the time the late 1900s rolled around.
A Real-World Product: What Sets It Apart
HFBA doesn’t grab headlines, but ask an analytical chemist and you’ll see a different story. In chromatography, especially high-performance liquid chromatography (HPLC), HFBA throws its weight around as an ion-pairing agent, helping separate compounds that don’t play nicely with others. Its backbone—seven fluorine atoms linked to a butyric acid base—lets it handle extreme chemical environments without falling apart or causing safety scares that come with harsher alternatives. I remember early in my lab career seeing how HFBA cut down sample run times and delivered crystal clear readings in tricky peptide and small molecule separations. It solved real headaches with noisy backgrounds and unpredictable retention times.
Under the Microscope: Physical & Chemical Properties
You won’t forget the pungency of HFBA if you’ve ever cracked open a fresh bottle in a poorly ventilated room. This colorless liquid, with a boiling point hovering around 130°C and a density heavier than water, stands out for its volatility and heavy fluorine presence. The seven fluorine atoms attached to the carbon chain don’t just make the molecule stable—they also supercharge its surface-active properties. You get a strong, non-nucleophilic acid, meaning it lends protons without jumping into unwanted side reactions. The molecule’s structure means it behaves predictably, which matters for both safety and lab consistency. Its strong acidity, blended with a stubborn resistance to hydrolysis, makes it a fixture in analytical workflows that rely on tough, unyielding reagents.
Preparation: The Art and Challenge
In practice, preparing HFBA isn’t something you’d take on in a high school lab or even a routine university class. Synthesis happens at an industrial scale, typically harnessing telomerization reactions or by fluorinating precursors like butyric acid or its perfluorinated cousins. The challenge lies with the intense reactivity of fluorine and the need for tight atmospheric control—people working these prep lines need substantial training and robust protective setups. The finished product arrives tightly sealed, flagged for both its corrosive character and its tendency to permeate seals if left unattended.
Chemical Reactions & Modifications: Beyond the Basics
Where HFBA really shines is in its role as more than just a simple acid. The molecule steps into modification chemistry, often transforming amines into more stable derivatives for GC-MS analysis. Fluorinated acids like HFBA lend themselves to subtle tweaks, accommodating a range of substitutions when researchers chase very specific reactivity or detection routes. Spend any time among analytical chemists, and you’ll hear how HFBA sidestepped roadblocks where other acids fell short. Its ability to help derivatize compounds for mass spectrometry sits at the heart of why it gained a reputation as a workhorse.
Keeping Track: Synonyms & Product Names
Anyone digging through catalogs should know that HFBA may pop up with several names. Some call it perfluorobutyric acid, others lean on the systematic name, 2,2,3,3,4,4,4-heptafluorobutanoic acid. Trade labels vary depending on supplier, but the backbone stays the same. For those who grew up navigating old chemical supply lists, these shifting terminologies do more than confuse—they remind us that staying current means double-checking every compound’s identifiers before running expensive experiments.
Safety and Operational Standards: Staying Smart in the Lab
Anyone who’s handled HFBA respects its bite long after a safety seminar ends. It doesn’t demand full HazMat suits, but good luck ignoring the warnings about its corrosiveness and volatility. Direct skin and eye contact stings, and long-term inhalation earns you a trip to the health office. Proper fume hoods, acid-resistant gloves, and a no-nonsense attitude turn routine handling into a matter of muscle memory. Disposal draws special attention—regions have grown more particular about waste containing perfluorinated compounds, reflecting broader concerns about the persistence of fluorinated materials in the environment. Those lessons drilled into every chemist: treat every drop like it matters, because it does.
Application Area: From Labs to Industries
HFBA has found its groove, especially in analytical labs and pharma development spaces. Whenever scientists need clean, repeatable results for biological samples or pharmaceutical impurities, HFBA proves its worth. It steps into proteomics, metabolomics, and other ’omics where trace-level detection and separation shape the reputation of entire workflows. Some environmental labs pull HFBA into pesticide and contaminant monitoring, chasing parts-per-billion clarity in water and soil. In the real world, clients rarely ask for it by name, but they appreciate the data quality it supports, with regulatory filings and drug submissions riding on its shoulders. Its grip on these fields only tightens as detection standards grow tougher year by year.
Research & Development: Pushing Boundaries
Recent years pushed HFBA into new territory. Labs keep tinkering with improved methods—lowering reagent volumes, upgrading waste management, revisiting sample prep protocols to trim costs and reduce environmental impact. There’s excitement around developing greener synthesis routes for fluorinated acids, both to cut down industrial emissions and to keep future regulatory hurdles from wiping HFBA off the bench. In some companies, R&D scientists tinker with alternative fluorinated acids that capture similar functional power but break down more cleanly in nature. Journals brim with incremental advances, reflecting how valuable HFBA remains even as the world’s chemistry toolkit evolves.
Toxicity Research: Navigating the Gray Areas
Early on, safety officers flagged perfluorinated compounds as tough customers for waste streams. HFBA finds itself swept up in ongoing concerns about the persistence and toxicity of fluorinated organics. Studies on aquatic life, soil health, and even human health keep growing, as researchers map out the ways these acids resist natural breakdown and bioaccumulate. No one wants a repeat of high-profile stories where industrial fluorocarbons lingered for decades in groundwater. Peer-reviewed studies keep digging into whether HFBA breaks the rules of environmental persistence, and if so, what new disposal or containment protocols look like in an age of tighter regulation.
Future Prospects: Looking Beyond the Status Quo
Chemists talk about HFBA in a way that blends admiration with a nudge of concern. On one hand, no direct substitute offers its combination of acid strength, chemical inertness, and chromatographic versatility. On the other, the steady drumbeat of environmental regulation leaves the future of HFBA in question—a reminder that lab heroes today need to reckon with tomorrow’s constraints. The push for “greener” acids and drop-in replacements is no trend; it reflects the growing demand for tools that clean up after themselves without sacrificing precision. As R&D catches up, and as regulations sharpen their focus on persistent chemicals, everyone who’s leaned on HFBA will be watching both for breakthroughs and for new expectations on the safe stewardship of this unique, potent acid.
A Chemical That Drives Lab Precision
In my time around research labs, simple acids rarely needed much attention. Heptafluorobutyric acid, though, plays a unique role that deserves a closer look—not just for the sounding of its name, but for its impact in analytical chemistry. This strong, fluorinated acid isn’t a household staple, and it doesn’t aim for mass-market fame. Instead, it finds its strength in how it improves scientific precision behind the scenes.
Making Chromatography Work Better
Most folks will never hold a vial labeled heptafluorobutyric acid. In labs, though, you see it pop up in liquid chromatography systems, especially in high performance liquid chromatography, or HPLC. Why? HPLC has changed the way we separate and identify compounds, from blood samples to food safety checks.
I remember running HPLC tests on blood plasma samples where basic acids like acetic wouldn’t cut it for difficult analytes. Heptafluorobutyric acid brought something else—better separation for certain amino acids or peptide samples. The acid works as an ion-pairing agent, latching onto the molecules researchers want to study and helping them apart from others in a crowded mixture. That level of precision holds huge value for clinical diagnostics, doping control in sports, and pharmaceutical quality checks.
Backstage Role in Mass Spectrometry
Labs often couple chromatography with mass spectrometry, looking for absolute answers about drug breakdown products or proteins in a complicated sample. Heptafluorobutyric acid shows up again. It cleans background noise, sharpens the focus, and boosts the researcher’s ability to spot minute amounts of a target molecule. Its fluorinated tail offers a stability you don’t get from simpler acids, and that matters for pushing the limits of sensitivity.
Driving Toward Clean Data
Lab work sometimes feels like an arms race—every molecule counts, every bit of interference matters. Tools like heptafluorobutyric acid help tilt the odds back in favor of reliable data. This isn’t just useful; it’s essential when patient care or legal cases depend on firm answers. Its role in DNA and peptide analysis goes deep, making accurate sequencing or identification possible in forensic or genetic research.
Concerns and Solutions for Safer Handling
Strong acids with fluorine atoms come with downsides. I’ve seen worry arise about environmental impact and technician safety because something that effective also brings hazards. Heptafluorobutyric acid can irritate skin and eyes, and improper disposal adds unwanted fluorine compounds to wastewater. Managing risk demands proper gloves, fume hoods, and safe waste stream practices. Instead of turning away from these powerful tools, smarter training and investment in lab infrastructure can lower exposure and keep science on track.
Innovation and Looking Ahead
Chemistry doesn’t stand still. While heptafluorobutyric acid fills an important role now, researchers look for “greener” alternatives that deliver similar performance with less risk. Until those come along, real-world chemical analysis still relies on precisely these sorts of specialized compounds. With above-average diligence and respect for safety, this acid helps science tackle crucial questions—in medicine, food safety, and almost every field that needs razor-sharp measurement.
Understanding a Complex Chemical
Heptafluorobutyric acid, a mouthful even for folks used to chemical names, shows up in labs and industries that need high-performance separation methods. It's part of the perfluorinated chemicals family. These chemicals have a big role in the science behind liquid chromatography, making measurements sharper and results more reliable for scientists looking at proteins or small pharmaceutical compounds. Still, seeing chemistry at work in medicine or research doesn't mean the chemicals themselves get a free pass. Safety around heptafluorobutyric acid matters way beyond the walls of a laboratory.
Hazards and Exposure Issues
Heptafluorobutyric acid comes with real risks. It’s corrosive, and that shows up fast when skin or eyes meet even a small spill—redness, burns, and pain become real problems. Breathing in its vapors can irritate the nose, throat, and lungs. People who spend time around it in a lab sometimes talk about that sharp, biting odor that sticks around long after a bottle gets opened. Swallowing any amount is a medical emergency, given its ability to attack tissue all the way from mouth to stomach.
Chemical safety data sheets and guides flag heptafluorobutyric acid as an irritant and corrosive. Direct contact damages tissue quickly. Long-term studies fall short on details, but what we do know lines up with most strong acids: low pH, risk of chemical burns, and acute toxicity. There’s not enough long-term data to draw a line between exposure and things like cancer or reproductive harm, but anyone handling it has reason to limit exposure.
Why the Public Should Care
Some people think specialized chemicals only matter to chemists. If it never leaves the lab, does it really touch everyday life? In reality, industrial chemicals can flow, sometimes by accident, into air and water. That’s been true for many perfluorinated compounds. Communities near factories hear about “forever chemicals,” and it’s no longer just background noise. Places with PFAS contamination, even from different cousins in the chemical family, see neighbors lining up for water distribution, property values nosediving, and parents worrying about long-term health effects.
Heptafluorobutyric acid doesn’t grab headlines like Teflon or PFOA, but it fits into that bigger world of persistent, hard-to-break-down substances. The risk may stay small if labs and disposal facilities take care, but accidents happen. Disposing of these acids poses tough questions. Wastewater treatment plants aren’t set up to break down fluorinated chemicals, leaving open space for more of them to trickle out over time.
A Safer Way Forward
People working with heptafluorobutyric acid in labs recognize these risks and use gloves, goggles, and fume hoods—basic safety holds strong as a frontline defense. But strong protocols don’t erase the need for better solutions. Research into less hazardous alternatives for chromatography keeps moving, though some analysts feel stuck using what’s available until greener chemistry catches up.
On a bigger stage, stronger tracking and reporting rules could close the gaps. If every shipment and disposal record matched, leaks and illegal dumping might stay rare. Better training for both researchers and local hazardous waste teams could cut accidental releases. Funding for future science should reward not just what chemicals can do, but how safely they fit into both labs and the environment.
People may not have to keep a bottle of heptafluorobutyric acid under the kitchen sink, but paying attention to where it ends up and how it’s handled counts in every community.
The Science Behind the Formula
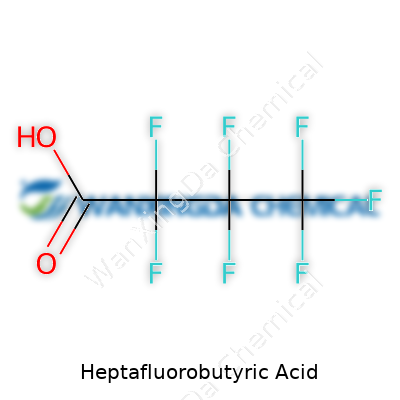
Heptafluorobutyric acid might sound like something pulled from the pages of a dense chemistry textbook, but its formula gives away a story that connects chemistry to real-world uses. The formula—C4HF7O2—boils down to four carbon atoms, a single hydrogen, seven fluorines, and two oxygens. This particular mix shapes a molecule that stands up to tough conditions where most acids would fall apart or react uncontrollably.
Why the Formula Matters
The first time I worked with heptafluorobutyric acid in a lab, it became seriously clear how that group of seven fluorines made a difference. Those tiny atoms pack a serious punch, making the molecule both stable and incredibly reactive where needed. In applications like analytical chemistry, the acid provides a reliable way to tweak how molecules behave during separation and analysis. Labs often rely on it as an additive in chromatography, letting tricky samples break apart for more accurate readings. The recipe itself defines function; you can't swap in just any acid and expect the same performance.
Impact Beyond the Laboratory
Knowing the chemical makeup gets you much further than a quick homework answer. Chemists and technicians often think about ways to control reactivity or even reduce chemical waste. The fluorines in C4HF7O2 give the acid extreme resistance to breakdown, but this same strength brings up environmental questions. Once released, these kinds of resilient compounds can stick around for years, finding their way into water and the food chain. The formula shapes more than a bottle's label—it changes how we think about safety, cleanup, and responsibility.
The Case for Smarter Solutions
As awareness grows about long-lived acids and their impact, researchers and companies have started looking at alternatives—ways to balance the need for high-performance chemistry with the need for cleaner results. Some labs have begun testing organic acids that don't hang around so long or break down into safer pieces. Government agencies, too, have started watching these chemicals more closely, encouraging the development of greener versions.
Heptafluorobutyric acid’s formula captures both the promise and the pitfalls of chemistry that moves faster than our habits. If scientists and manufacturers make it a habit to check not just the application, but also the aftereffects of these compounds, the odds tip toward both progress and protection. The story doesn't end with what sits inside a flask or a chromatograph; it keeps going with the choices chemistry forces us to face.
Why Details Matter with Chemical Storage
Heptafluorobutyric acid doesn’t play around. It’s got a sharp bite as a corrosive liquid, and careless storage means risking a fire department visit or ruined research. I’ve seen people underestimate acids, thinking a simple plastic bottle on any old shelf will do. This particular acid brings more complications: highly reactive, releases fumes, and eats through materials you’d expect to hold up. Every day, I remind new lab members that chemistry isn’t forgiving. Small mistakes snowball fast.
Lessons Learned from Overlooked Risks
Glass looks tough, and it handles a lot in the lab, but not all plastics or glass types stand up to a strong organic acid with fluorine’s added muscle. I watched someone lose an expensive sample once—their storage cap shriveled right off in a month, just from vapor attack. That acid didn’t need a spill; its fumes crept out, filled the cabinet, and left a sticky mess. Damage like that leads to wasted money and time, never mind the hidden safety hazard. This experience stuck with me. Strong acids like this demand chemical-resistant containers approved for long-term storage—usually fluoropolymer or thick-walled glass. Loose-fitting lids and generic plastics just won’t make it.
Temperature and Ventilation: Everyday Neglect, Big Consequences
I’ve worked in labs where bottles of strong acids ended up beside excess buffer solutions or even salts, just because bench space ran out. This always ends in regret. Heptafluorobutyric acid needs a cool, stable environment away from sunlight because heat gives its vapors more energy to escape. Those vapors don’t only threaten equipment, either: prolonged breathing of fumes over time wears down your lungs whether you notice right away or not. Chemical fume hoods aren’t just for handling during use—they’re a wise spot for storage if you can lock cabinets inside. Dry, cool, and well-ventilated—this was drilled into me early on, though shortcuts tempt everyone under pressure.
Separation Prevents Chaos
Accidents don’t schedule themselves. If you store heptafluorobutyric acid near bases, oxidizers, or anything flammable, you set yourself up for trouble. I used to think labeling and physical distance in storage cabinets was overkill until I saw a close call—the fumes from a carelessly stored acid reached a bottle of base, and the whole place reeked for days. A good rule sticks: acids to one side, bases and incompatibles far away, clear markings, and no exceptions for convenience. Proper practices stop small mistakes from turning into major events.
Small Fixes, Safer Labs
I’ve watched labs overhaul safety routines for less, yet chemicals like this one still trip up experienced folks. Double-walled containers only cost a little more. Locks and alarms aren’t a sign of paranoia—they’re insurance. Training matters even more. People can memorize handling protocols, but if they don’t trust the importance of storage, all those rules fade during a hectic day. I bring up examples like these in safety trainings: they put the “why” behind routines, push everyone to respect chemicals that seem stable but hide real risks. At the end of each week, I check our storage cabinets, and I encourage others to do the same—just a simple walk-through can catch problems before they grow.
Why Respecting Heptafluorobutyric Acid Makes a Difference
I’ve learned over the years that unpredictable chemicals carve the rules, not the other way around. Heptafluorobutyric acid isn’t the flashiest bottle in a chemistry stockroom, but you notice its bite when you see what it does to unprotected skin or lungs. With an odorless, colorless presence that hides its danger, more than one well-meaning scientist has discovered too late the costs of not taking it seriously.
This acid’s strong corrosive punch explains the cautions posted on its bottles. I remember a careless late-night project where disposable nitrile gloves were no match for a careless splash. Skin tingled, then turned angry red, reminding me how easy it is to forget why thick acid-resistant gloves matter. No lab coat thin as a bedsheet held its own against those drops either; it soaked through fast, burning skin under my sleeve. Whenever we talk about “personal protective equipment,” it’s these moments that replay in my mind.
Breathable Air and the Forgotten Mask
Working in a ventilated fume hood feels obvious until you don’t have one: humid air carries invisible vapors that sting your throat and leave you coughing through the rest of the day. Exposure isn’t always dramatic—sometimes the burn emerges quietly. The acid’s fumes, heavier than air, like to sneak past open windows or drift to the floor. Standard lab masks don’t always cut it. An acid vapor respirator may sound like overkill, but anyone with sinus pain after a few short minutes wouldn’t call it excessive.
Safe Storage: It’s Not Just About the Label
I’ve seen more than my share of storage blunders—acids lined next to incompatible organics, aging containers leaking onto the shelf. Heptafluorobutyric acid gives metals a run for their money, corroding containers not built for it. Glass works best, and sealing it tight prevents unpredictable reactions with moisture in the air. Even when labels warn of “severe burns,” plenty of users still treat acids like nuisance ingredients. If spills happen, neutralizer and absorbent should be ready. Baking soda won’t cut it for this one; specialized neutralizers handle these spills best.
Training Gaps and Community Solutions
Regulatory guidelines often show up as dry handouts covered in dense text, easy to ignore until something goes wrong. The big gap isn’t always in the rulebooks; it’s in the hands-on knowledge passed between lab workers. I’ve found that on-the-job mentorship leaves a stronger impression than a short online video. Briefings before opening a bottle, showing exactly how acid-resistant gloves feel, explaining why eye protection stays on from start to finish—these actions stop injuries before they start.
In some research teams, monthly “near-miss” discussions give space to talk about close calls. Problems laid out honestly, stories told without fear of embarrassment—these drive culture change faster than regulatory announcements. Labs who invest in training, up-to-date safety gear, and peer accountability avoid the costs and pain of acid accidents. If anything, my experience says talking openly about mistakes teaches more than any warning label can.
Everyday Vigilance Pays Off
Heptafluorobutyric acid carries a reputation earned over years of mishaps and, occasionally, disaster. Respect builds not from fear but from collective memory, a recognition that risks never stay in the manual—they show up the moment you cut corners. Commitment to safety, fed by real experience and plain hard truths, shapes better habits in the next generation of chemists. Because in labs, it only takes one overlooked splash to remind you why these precautions exist.