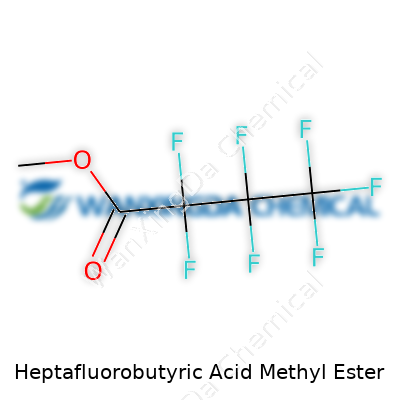
Heptafluorobutyric Acid Methyl Ester: The Chemistry Behind a Story of Progress
Looking Back: How Heptafluorobutyric Acid Methyl Ester Emerged
Back in the 1960s and 1970s, the chemical world started to ask for more specificity, more control, and better tools when it came to separating and analyzing acids, amino acids, and other small molecules. Techniques like gas chromatography and mass spectrometry became more refined, but they demanded sharper contrast between substances. Some of the early chemists in this field turned to the magic of fluorination—adding groups like heptafluorobutyric acid methyl ester to molecules that, by nature, didn’t want to stand out. This molecule, with its string of fluorines, began serving as a derivatization reagent, making otherwise drab molecules like amino acids light up in analysis. The story has always been about making complex molecules easier to handle, easier to see.
In the Lab: What Sets Heptafluorobutyric Acid Methyl Ester Apart
You can’t ignore the way this ester acts. The stuff is a colorless liquid with a faint odor, miscible with organic solvents, and stands out for its volatility. That volatility means handling gets tricky and storing it begs careful sealing, away from any stray sources of moisture. Chemically, the perfluorinated chain shapes the story here—the ester drips with electron-withdrawing power. That plays out in its reactivity and in how it helps derivatize other compounds for methods like HPLC or GC-MS. As for technical specs, its boiling point hovers near 53 degrees Celsius, the molecular weight sits near 230, and its density offers a boost compared to non-fluorinated analogs. I remember opening a vial early in my career, thinking ‘this stuff won’t linger’—its low boiling point made every measurement feel urgent. The label’s warnings about eye and skin irritation weren’t just regulatory noise; a drop on the bench and you knew to clean up, fast.
Preparation and Chemical Reactivity: Real-World Practicalities
The prep work here shows off the value of modern synthetic chemistry—start with heptafluorobutyric acid, introduce methanol, use a strong acid like sulfuric or hydrochloric for catalysis, then distill and purify the final liquid. The process, while straightforward in principle, gets sensitive to humidity and control of temperature, since even small deviations can leave you with a cocktail of unwanted side-products. The result: a methyl ester with a lively carbonyl, hungry for reaction with nucleophiles, especially amines and alcohols. In practice, it works well when you need to tag a molecule without adding bulk or extra color—its perfluorinated nature cranks up detection sensitivity across different detectors, surpassing non-fluorinated esters. Over time, improvements in process chemistry have lowered the cost, but the handling remains as delicate as ever—protective gear and ventilation matter as much as a steady hand.
Understanding the Names: Synonyms Across the Literature
In journals and catalogs, the same substance pops up under a slate of different names: methyl heptafluorobutyrate, methyl perfluorobutyrate, and even HFBME if researchers want to save ink. This spread of synonyms rarely helps clarity, and the confusion underscores the importance of double-checking CAS numbers or structural diagrams. I’ve seen lab mistakes from mislabeled compounds or poor translation between American and European catalogs—a headache that gets solved with careful tracking but never fully goes away. Anyone working across borders or diving into old literature learns fast that a familiar name might hide a fluorinated skeleton underneath.
Safety, Training, and Real Hazards
The push for new analytical chemicals often runs up against the stubborn facts of toxicity and exposure. Heptafluorobutyric Acid Methyl Ester irritates eyes, skin, and the respiratory tract, not through mere regulatory paranoia, but because even tiny spills produce enough vapor to sting the nose and eyes. Accidents usually involve open vials or carelessness during pipetting. Wearing gloves, lab coats, eye protection, and working under a fume hood quickly becomes second nature to those who respect their lungs and nerves. A good training program centers around practical drills, not just paperwork. Even with strong ventilation, I’ve seen colleagues get headaches or dizziness from brief lapses. While the ester isn’t the most toxic reagent on a typical analytical bench, it earns respect through potent volatility. The experience of my own mentor, who prioritizes a clean, organized workspace, drilled into me the lesson: you can’t take shortcuts in chemical handling—not if the goal is long-term health.
Applications: From Analytical Labs to New Frontiers
The main stage for this compound shows up in separation science. Gas and liquid chromatography both gain better peak shape and higher sensitivity if you’ve pretreated samples with the methyl ester; amino acids, peptides, and carboxylic acids, in particular, reveal their secrets more readily. Environmental monitoring and metabolomics also grab hold of this approach: tracking tiny amounts of organic acids in blood, urine, or water gets a bump in confidence. Not long ago, I worked on a project quantifying trace agricultural residues—a process made smoother by this very ester. Despite its specialized reputation, industries from pharmaceuticals to food safety find ways to use it as regulations tighten and demand for accuracy climbs. The drive to squeeze more data from smaller samples keeps new users experimenting with derivatization possibilities beyond just routine analysis.
On the Edge: R&D and Toxicity Studies
Research and development chug ahead, looking at both the strengths and weak spots. Investigators seek methods to recycle unused reagents, drop waste output, and sharpen both selectivity and stability in derivatizations. Toxicity research doesn’t lag behind. Data show that acute exposure irritates lungs and mucous membranes, and long-term health effects remain uncertain, nudging labs toward ever-better containment and cleanup. One lab group I crossed paths with began developing newer fluorinated tags with shorter environmental lifetimes—an effort that speaks to a slow but steady shift in thinking. Chronic toxicity from regular lab exposure hasn’t been fully mapped, but the whiff of caution keeps windows open and fume hoods on high. There’s pressure from both inside and outside the field to keep pushing for safer, greener replacements, yet this ester holds its ground in the analytical toolkit thanks to its effectiveness.
Future Trajectories: Balancing Performance and Responsibility
Peering into the future, it’s clear that heptafluorobutyric acid methyl ester will stay embedded in advanced analytical science, particularly for labs where ultimate detection limits matter. The challenge lies not just in ramping up performance but in reducing environmental and human impact. Chemists talk more about greener chemistry and sustainable synthetic routes. The trend points to tweaking production to minimize fluorinated waste or even designing analogs that degrade harmlessly after use. Government regulation now sees more public scrutiny around perfluorinated compounds, driving both transparency and innovation. My bet is we’ll see hybrid reagents that keep fluorine’s detection punch but break down safely in the environment. For young chemists, the lesson stays the same: respect the tools, push for more knowledge, and keep safety firm alongside curiosity. Every big change in chemistry traces back to small, thoughtful steps in the lab.
Every so often, a chemical earns attention not for how it looks or smells, but for what it quietly enables behind the scenes. Heptafluorobutyric acid methyl ester sits in that category—a helper deep in the scientific trenches, not something folks keep under the kitchen sink. Most people would never come across it. Scientists in synthetic chemistry, though, know it as a specialty solvent and a tool for making molecules talk.
The Science Behind the Scenes
This compound finds its place on the benches of analytical labs, especially those using gas chromatography (GC) and mass spectrometry (MS). These are the machines that handle all sorts of detective work in the lab. They track down tiny amounts of steroids, pesticides, amino acids, even street drugs. Often, the molecules being measured look too similar, so specialists dress up these molecules with certain chemicals to make sure the machines spot just what they are told to find. That’s called derivatization, and heptafluorobutyric acid methyl ester is a go-to dressing agent. It sticks a fluorinated tag onto compounds, making them easier to separate and detect, boosting clarity and reliability.
Why Go to the Trouble?
The more precise the labs get, the more they demand. Doctors want to know if a patient’s medication is behaving as it should. Farmers need to prove their crops obey pesticide rules. Researchers look for new signs of disease in body fluids nobody can pronounce. If the molecule isn’t dressed up the right way, the machine squints and gets it wrong. Trace detection often depends on making tiny molecules look a bit flashier. Adding fluorine atoms—especially through something like heptafluorobutyric acid methyl ester—makes a big difference. Increased sensitivity, fewer mix-ups, and faster runs keep the industry rolling.
Broader Impact on Health and Safety
Public health and environmental labs push for lower detection thresholds every year. This compound, because of its reliability in derivatization, keeps water and food safety programs on target. It reduces the time it takes to get an answer and improves confidence in the numbers labs report to regulators. School lunch programs, imported food inspections, forensic toxicology tests—these all rely on chemistry tools, and heptafluorobutyric acid methyl ester quietly props up those systems.
It’s Not All Sweet Smell
Fluorinated chemicals stick around. Their persistence has caught plenty of concern from regulators and watchdog groups. Even a specialty compound like this deserves close attention when used in bulk. Best practice says use just enough, collect waste properly, and keep hands and lungs safe. Some labs now seek alternative derivatization agents, chasing both high performance and lower environmental risk. It’s a fair goal, especially as demand climbs and the world worries more about persistent chemicals in the environment.
Moving Forward
Smart use of chemistry opens new doors. Heptafluorobutyric acid methyl ester earns its keep by making hard problems manageable—helping analysts see what matters in a messy world. As science pushes for greener, less stubborn chemicals, the industry needs clear conversations between researchers, safety officials, and manufacturers. Balancing sharp detection with environmental care sets the course for the next generation of lab tools. For now, this ester keeps watch as a humble, effective member of the chemical toolkit.
Anybody who’s done real work in a chemistry lab knows you get a sixth sense around certain chemicals. Some names just stick out, and Heptafluorobutyric Acid Methyl Ester fits right into that group. It’s not the sort of compound you want splashing on your skin or making its way into your lungs. This isn’t scaremongering—it’s learned from days in the lab, where one sloppy move can mean hours in an eyewash station or much worse.
Why Respecting This Compound Matters
The strong smell of anything fluorinated hits hard. That should set off alarms. Heptafluorobutyric Acid Methyl Ester is volatile, and it can irritate eyes, skin, and the respiratory tract. I remember one time as a grad student when someone uncapped something too quickly, and the whole room went from friendly banter to eye-watering silence. We cleared out right away. That was with all the fume hoods running—and the warning still wasn’t enough for some folks, at least not until they experienced it themselves.
This ester works in analytical chemistry, especially in derivatization for gas chromatography. Research has real consequences, so does exposure. Not using protection puts careers and health on the line. Skin absorption risks serious burns, and inhaling vapors can trigger coughing fits, dizziness, and worse, depending on exposure.
Simple Habits Prevent Big Problems
Proper gear is the starting point. Some old-school chemists used to wave away gloves for quick work—nobody I know who’s stayed in the lab long keeps that habit up. Nitrile gloves, lab coats, and tightly fitting goggles go on before uncapping a single vial. Face shields help during bigger batches or when mixing.
Skip any part of that, and you’re asking for trouble. Think about one small splash. Nitrile stands up better than latex; I’ve seen latex buckle under strong acids. Keeping a few pairs of reliable gloves within arm’s reach makes a difference. After spills, change gloves right away. That inconvenience is nothing compared to skin damage.
Ventilation Makes or Breaks Safety
Good fume hoods aren’t a suggestion—they’re law in any chemistry setting. Even outside regulated workplaces, running ones are the gold standard. Some modern labs run regular airflow checks; others fall behind and get away with it until an incident. At home or in less formal labs, setting up a local exhaust system or always working near a window should be the bare minimum. Open jars mean invisible fumes, which hang around for hours. I’ve watched careless workers double over with headaches for the rest of the day from just a short mishap.
Chemical Storage Needs Discipline
I used to joke that the only thing scarier than the chemicals was a messy chemical storeroom. Fluorinated esters like this acid methyl ester need real shelving—metal racks and labeled, sealed containers. Never store next to bases or oxidizers, since even a few stray drops can ignite violent reactions.
Checking for leaks and using secondary containment keeps everyone honest. Broken seals or tiny hairline cracks on a bottle can mean days of hunting around for the source of a mysterious chemical odor. I learned early on to log every bit in and out, and not to trust my memory over actual notes.
Treat Safety as a Mindset, Not a Burden
No rulebook ever saved anybody by itself—it’s people paying attention that change outcomes. Bringing up near-misses helps. Rushing or making exceptions always leads to regrets down the line. Newcomers in the lab learn best from experienced folks who take this seriously: treat every container with respect and keep your space clean. Accidents rarely announce themselves in advance. Real safety starts with never taking shortcuts or assuming “it’ll be fine just this once.”
Looking Past the Chemical Jargon
Plenty of folks glaze over at names like heptafluorobutyric acid methyl ester. The chemistry sounds complicated, like something best left on a lab bench. But every substance—especially one used across labs in research, pharmaceuticals, or analytical chemistry—deserves a closer look.
Breaking Down the Name
Heptafluorobutyric acid methyl ester gets its lengthy title from its structure: a butyric acid with seven fluorine atoms and a methyl ester group replacing the typical hydrogen. The chemical formula is C5H3F7O2, and the molecule weighs about 214.06 grams per mole. In practice, such detail helps chemists predict how the compound acts in real-world experiments or environmental situations.
Why Chemists Care
Heptafluorobutyric acid methyl ester has become a staple in analytical labs, especially in chromatography. Its volatility and strong fluorination help make compounds more detectable by machines that struggle with less reactive chemicals. This function means the compound can shine a light on hidden substances in food, soil, water, or medicine that might otherwise slip under the radar.
But power comes with a responsibility. Heavily fluorinated compounds like this one don’t generally break down easily in the environment. That's caught the eye of toxicologists and environmental scientists. The resistance of these chemicals to degradation means they can build up in soils or move through water systems well after their intended use.
Safety and the Bigger Picture
I’ve seen the tug-of-war between innovation and responsibility up close. In graduate school, our lab loved compounds that cut analysis time, but we faced strict rules about storage and disposal. Heptafluorobutyric acid methyl ester needs special handling not because of immediate toxicity, but because careless storage or spills can turn a quick experiment into a persistent environmental issue.
News about perfluorinated compounds found in drinking water or seafood points to the same challenge. Fluorine-rich technology gave us nonstick cookware and better medical diagnostics, but nature doesn't always play along with indestructible molecules. It’s tough to ignore headlines about bioaccumulation and contamination, which link back to the ordinary decisions made in labs and factories everywhere.
What Moves Us Forward
Chemical manufacturers and regulators constantly assess risk. The solution starts with tighter tracking and disposal systems—thinking through every bottle that arrives and leaves a facility. Labs that purchase heptafluorobutyric acid methyl ester have the best shot at reducing harm when they work with local hazardous waste programs instead of taking shortcuts. Safer replacements would help, but the industry moves cautiously, since established chemicals like this one have long data records and clear behavior.
Education also plays a role. Chemists in training need detailed guidance on the downstream effects of their choices, so they see every vial as more than a research shortcut. Transparency about sourcing and environment-friendly disposal closes the loop between laboratory science and the world outside.
Looking Ahead
Science keeps advancing, driven by a need for speed and precision. Heptafluorobutyric acid methyl ester, with its unique formula and weight, offers tools that make complicated analysis seem simple. But as we use these tools, weighing today’s breakthroughs against tomorrow’s environmental cost, we can keep pace with innovation and public health together.
Keeping Harmful Vapors in Check
Every so often, the shiniest fume hood or the thickest gloves don't fully prepare a person for chemicals like heptafluorobutyric acid methyl ester. Its strong odor and tendency to escape into the air make it clear that plain carelessness isn't an option. Tossing the bottle on a crowded shelf or next to an open flame ends up flirting with risk in a way nobody wants in the lab.
A bottle of this ester prefers a cool, dry spot, away from any heat source and certainly out of reach from direct sunlight. My first lab job involved tracking volatile chemicals like this one. Time after time, storage nightmares began with a lazy bottle left by a window or next to a steam radiator. If the liquid warms up, vapors start to leak, slowly at first, sometimes so subtly nobody notices until the room fills with a stinging odor. It doesn’t take long before headaches and complaints set in.
Moisture Is the Silent Enemy
Moisture never does these chemicals any favors. Leave the lid loose—humidity sneaks in, and you're suddenly staring at cloudiness or unexpected reaction products. I still remember a mislabeled carboy that turned brown within weeks; all because someone laughed off the “keep tightly capped” label. A tight seal keeps out water, dust, and a host of other surprises. Trying to fix a contaminated batch can turn into a full day's headache and lost resources.
Compatible Containers Matter
Not every glass or plastic bottle is created equal. Some people grab any old container, figuring a bottle is a bottle. That’s where things go sideways. Polyethylene warps with harsher solvents. Metal lids corrode or react, risking leaks or new contaminations. Real-world lab stories show that using fluoropolymer or amber glass minimizes these headaches. These containers also keep out light, which helps chemicals like this ester stay stable longer.
Ventilation and Spill Defense Go Hand-in-Hand
Leaving volatile esters in cupboards without airflow has never worked out well. Even small leaks add up, especially if salvage and hazardous material disposal require outside help. I keep a habit of checking stored chemicals weekly for sticky residue or chemical ‘sweating’ at the seams. A minor leak rarely stays minor if unnoticed. Absorbent pads beneath storage shelves catch early spills, and labeling containers clearly can prevent one chemical from getting mixed up with another.
Responsible Labs Stop Assumptions at the Door
Proper handling and respect for chemicals grow out of clear lab protocols, not just lectures. New students find out pretty quickly that storing reactive compounds like heptafluorobutyric acid methyl ester carries more weight than a checklist. Chemical Safety Sheets and real lab accidents both teach that a cool, ventilated, secured spot with compatible packaging keeps work safe and limits waste. Keeping the storage area organized and reviewed makes the biggest difference for everybody who passes through those lab doors.
A Tool for Sensitive Analytical Work
Stepping into any modern chemical or pharmaceutical lab, it’s easy to see the hunger for accurate, sensitive analysis. Researchers hunt for tools that make it easier to track down the tiniest traces of complex molecules—think drugs in biological samples or persistent environmental pollutants. Heptafluorobutyric acid methyl ester (HFBME) enters the scene as a strong helper in the hunt for clarity in those challenging analyses.
Boosting Chromatography: Adding Muscle to the Mix
People working in analytical chemistry respect chromatography for its reliability and versatility. Still, not every compound likes to cooperate, and scientists often struggle to get clean separations for substances with similar chemical properties. HFBME finds a purpose here. Researchers add this reagent as a derivatization agent for gas chromatography (GC) and liquid chromatography (LC), especially when handling stubbornly non-volatile or polar analytes. In my own work, tweaking a sample’s chemical structure gives otherwise invisible compounds a fighting chance to show up in the data. Adding fluorinated groups—HFBME’s specialty—makes analytes more volatile and less sticky, so they fly through GC columns with greater separation and stronger signals.
Sharpening Mass Spectrometry Results
Anyone who has spent time cradling a mass spectrometer learns the value of derivatization. Bio-analysts, for example, gravitate toward HFBME for preparing amino acids, peptides, or carboxylic acids ahead of mass spec runs. By swapping hydrogen atoms with bulky, electronegative fluorine through methyl esterification, HFBME bumps up volatility and lowers the chances for tailing or messy fragmentation. These improvements bring sharper peaks and cleaner spectra, which matter when quantifying trace doping agents in athletes or tracking environmental contaminants in food. Poor separation wastes time and materials—a mistake researchers and their budgets want to avoid.
Streamlining Sample Prep in Drug Testing
Labs running toxicological screens for drugs in blood or urine rely on speed and reliability. HFBME gets used to convert carboxylic acids into their methyl esters before analysis, since direct detection can be unreliable for such compounds. This step changes the chemical behavior of the drugs being tested, allowing for smoother passage through GC columns, lower detection limits, and more confidence when regulations or courtrooms demand it. The methyl ester helps hide polar, sticky interactions that typically drag down sensitivity.
Environmental Analysis Grows Up
People depend on sensitive methods to uncover pollutants like perfluorinated compounds or pesticides in drinking water and soil samples. HFBME plays a part in making those pollutants visible, after sample extraction and cleanup, by turning hard-to-detect acids into volatile esters ready for GC-MS routines. Better detection keeps communities safer and holds polluters to account. Labs avoid headaches by using a consistent, robust reagent like HFBME instead of fumbling through repeated failed analyses.
Looking Beyond: Learning and Limitations
It’s smart to recognize both value and risk with this ester. It brings chemical fluency but also toxicity and handling demands. Proper ventilation, careful storage, and safety protocols are non-negotiable. For new analysts, learning this workflow means absorbing attention to chemical compatibility and gradually mastering derivatization’s subtle trade-offs. With environmental rules tightening and new drugs emerging, flexible reagents like HFBME anchor labs’ ability to solve problems without skipping accuracy or safety. As analysis gets more demanding, smart chemists keep proven tools close—and HFBME remains on that short list.