Heptafluorobutyric Anhydride: Unpacking a Niche but Critical Chemical
Tracing the Roots: Historical Development
Watching the field of fluorinated compounds evolve over the decades, I’ve seen a pattern emerge—scientists discover a new fluorinated acid or derivative, then find ways to leverage its intense reactivity and stability. The story of heptafluorobutyric anhydride started with the exploration of perfluorinated acids in the mid-twentieth century, at a time when chemists craved sturdy building blocks for new molecules. Early on, researchers saw value in turning regular carboxylic acids into their anhydrides. This push led to the creation of heptafluorobutyric anhydride, a compound that married the harsh strength of fluorine with the crucial reactivity of anhydrides, carving out a unique sector in the world of organofluorine chemistry. University labs and later specialty chemical companies took interest, developing this molecule for cleaner reactions and higher product yields.
What Sets It Apart: Product Overview
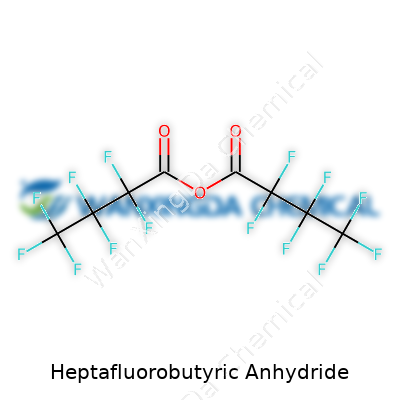
Heptafluorobutyric anhydride—though the name might tangle tongues—packs power in its bite. It’s not a household chemical, but anyone who has worked with it recognizes the signature acrid odor and unmistakable volatility. Perfluorinated anhydrides, including this one, boost chemical reactions with their aggressive acylating nature. You’ll find it almost colorless, giving a false sense of simplicity, but the faint vapor reminds experienced chemists that safety demands respect. I’ve stood in industrial labs and watched it expedite reactions that would crawl otherwise, cutting down on byproducts and improving selectivity, especially in pharma or fine chemical synthesis.
Getting Specific: Physical & Chemical Properties
Every time someone mentions fluorinated chemicals, durability on one side and environmental concerns on the other spring to mind. Heptafluorobutyric anhydride distinguishes itself with an impressive boiling point north of 50°C and a density that hints at its heavy fluorine load. Its structure—a pair of heptafluorobutyryl groups linked by an oxygen—makes it both hydrophobic and resistant to traditional hydrolysis, at least until it hits enough water or an alkaline environment. This stability means the chemical doesn’t break down easily, so it sticks around longer than many peers. It reacts sharply with nucleophiles, transforming basic molecules in seconds, and doesn’t shy away from heat or rough conditions. The intense electronegativity of its fluorine atoms shields it from unwelcome attacks, explaining why it hangs in there when other anhydrides cave.
The Nitty-Gritty: Technical Specs & Labeling
Labels on bottles of heptafluorobutyric anhydride read like a warning more than an invitation. Everything from storage temperature to vapor pressure demands careful management. I’ve seen shipments wrapped in layers of insulation and marked with more symbols than most chemicals. The law reads rightly strict—being a hazardous substance, regulatory authorities want full traceability and detailed hazard communication. Industries dealing with this molecule build their safety protocols around such standards, knowing every drop counts (for cost and safety alike). Handling this stuff out of spec can ruin months of research, not to mention cause serious injury.
Preparation: Step by Step
Chemists in academic settings often prepare heptafluorobutyric anhydride by reacting heptafluorobutyric acid with dehydrating agents like phosphorus pentachloride or thionyl chloride. The chemistry relies on replacing water with chlorine, which then gets swept away by further reaction, bridging two acid molecules. Sometimes, this means setting up cooled reaction vessels and keeping reactions under inert atmosphere to avoid hazardous fumes. In industrial plants, engineers scale things up, using continuous flow reactors to moderate the exothermic character and keep yields high. From personal experience, nothing gets hearts racing faster than a runaway reaction involving fluorinated gases, so process control isn’t just a box to check—it’s a matter of safety for everyone in the room.
Transformation: Chemical Reactions & Modifications
The real beauty of this compound shines when it jumps into action as an acylation agent. In organic synthesis—especially in peptide or polymer work—it reacts with alcohols, amines, or even water, driving forward processes that would otherwise demand more time and effort. The high fluorine content not only makes it reactive but also slaps unique properties onto the resulting molecules: greater chemical stability, resistance to metabolic breakdown, and novel biological activity. In the pharmaceutical sector, this means sharper tools for drug modification. Its derivatives end up in advanced imaging agents or specialty materials. But every time I see a novel fluorine-rich molecule hit the journal covers, I remember it started with sturdy, reactive chemicals like heptafluorobutyric anhydride doing the heavy lifting.
Other Names: Synonyms & Product Names
The chemical world doesn’t often settle on a single name for any substance, and this one wears a few disguises—heptafluorobutyric anhydride, perfluorobutyric anhydride, and sometimes just “HFBA anhydride” in shorthand notes. These names show up in research papers, patents, and purchase orders, and anyone in chemical procurement learns the subtle difference each iteration implies. Making sense of these synonyms keeps supply chains running smoothly and avoids costly mistakes in the laboratory.
Staying Safe: Handling & Operational Guidelines
Direct exposure to heptafluorobutyric anhydride comes with real danger—skin burns, eye irritation, and the threat of inhaling noxious vapors. I remember my first brush with improper fume hood use, and the sharp bite in my nose seared the lesson home. Today’s operational guidelines draw on years of filed accident reports and better gear: full PPE, rigorous fume extraction, and clear training. Keeping it locked in fluoropolymer containers cuts down on risk, since glass or metal sometimes reacts or gets damaged. Safety data sheets read more like field manuals than simple warnings, reflecting the lessons chemists and technicians learned, sometimes the hard way.
Where the Action Happens: Application Area
Ask industry veterans where heptafluorobutyric anhydride matters most, and fingers point quickly to analytical chemistry, pharmaceuticals, and electronic materials. In labs, it pops up refining chemicals for gas chromatography or fine-tuning molecules for drug development. Pharmaceuticals value it for its role in making metabolites “stick out” for tracking inside the body. Environmental chemists use it to derivatize persistent pollutants, making them easier to monitor and address. The electronics sector leans on its ability to modify polymer surfaces, imparting water and chemical resistance. It’s rarely the headline act, but heptafluorobutyric anhydride enables breakthroughs behind the scenes.
Pushing Boundaries: Research & Development
The pressures of stricter environmental laws and shifting market needs push research forward. Much of the excitement in fluorine chemistry comes from the search for new, safer, and more sustainable reaction partners. Heptafluorobutyric anhydride remains a fixture in efforts to build smarter drugs or better imaging agents, but labs worldwide dig into greener synthesis routes and alternative reagents. Conversations I’ve had with R&D teams often circle back to reducing waste and cleaning up effluents, given how persistent fluorinated chemicals tend to be. The shared sense among researchers is that each improvement edges the field closer to a safer and more responsible future.
Staying Healthy: Toxicity and Biological Impact
No one working around this chemical shrugs off its potential harms. Studies show significant toxicity with prolonged exposure, especially through the skin or lungs. Like many fluorinated compounds, heptafluorobutyric anhydride resists breakdown, so it raises concerns about accumulation in the environment or in workers’ bodies. Toxicologists have called for careful control of emissions and regular health monitoring for those in frequent contact. Across labs I’ve visited, robust ventilation, strict spill protocols, and emergency showers stand ready—not as overkill, but as protection against the real risk this molecule presents. Calls for further study continue, as the industry tries to balance the benefits with unknown long-term health and ecological effects.
Looking Forward: Future Prospects
The road ahead for heptafluorobutyric anhydride looks challenging but not bleak. Growing scrutiny over perfluorinated chemicals—especially due to their persistence and bioaccumulative nature—puts pressure on academic labs and manufacturers alike. I’ve sat in meetings where teams wrestled with regulations and sought creative ways to use less, recover more, and release nothing. Some eyes look to alternative non-fluorinated agents, but few match the unique punch packed by this anhydride. Where necessity persists, investments go to waste treatment, recovery systems, and improved worker safety, driven by both social responsibility and regulatory risk. The chemical remains indispensable for now, but how it gets made, handled, and disposed of will continue to change, shaped by innovation, public health research, and the ongoing debate over the future of perfluorinated compounds.
Real-World Chemistry: More Than Just a Strange Name
Walk into any modern analytical chemistry lab and you’ll probably spot a few bottles labeled with names that read like cryptic code. Heptafluorobutyric anhydride is one of those. Even after years working in labs, I never found this compound waiting next to common solvents or buffers, but anyone involved in proteomics or food safety testing would recognize its sharp, almost plasticky odor and know exactly why it matters.
Sharpening Mass Spectrometry with Derivatization
I first ran into heptafluorobutyric anhydride working on mass spectrometry projects checking trace contaminants in food samples. The process usually runs into snags because certain molecules either overlap signals or just don't show up clearly. This anhydride solves the problem. It doesn't just mix in—it changes the chemical structure of the sample by adding fluorine-rich groups. These groups boost sensitivity in mass spectrometry and help molecules separate more cleanly, giving scientists clearer data. Research shows this approach works wonders for analyzing amino acids, pesticides, and even pharmaceutical residues in everything from water to blood plasma. Without it, labs would struggle to hit the strict detection limits that modern regulations demand.
Peptides, Proteins, and Pharmaceutics
The reach goes beyond environmental or food labs. Proteomics researchers often describe the same frustration: small peptides just don't behave during separation. They crowd together, peak shapes fall apart, and quantifying real concentrations becomes guesswork. By treating these molecules with heptafluorobutyric anhydride, chemists boost the hydrophobicity or “water-fearing” nature of peptide fragments. The change means better results for HPLC (high performance liquid chromatography), which many labs rely on for protein identification and quantification. According to journal reports, this reagent’s effect on retention times and resolution can save days of troubleshooting.
Why Precision and Safety Go Hand-in-Hand
I’ve worked near volatile anhydrides before, and they demand respect. Their reactivity makes them useful, but also potentially hazardous. Heptafluorobutyric anhydride is no different; improper handling leads to irritating fumes, chemical burns, or worse. There’s a clear lesson here: as much as this compound unlocks new data, it demands real investment in fume hoods, protective gloves, and up-to-date training. Academic publications remind us that ignoring these fundamentals leads to accidents, even for seasoned researchers.
Balancing Progress and Environmental Caution
Its clear value raises tough questions. Fluorinated chemicals don't break down easily. Throughout my career, disposal of leftover reagents always meant extra paperwork, double-checking guidelines, and special waste collection. This level of care, while tedious, protects water supplies and keeps regulators happy. Recent reports from environmental agencies stress that labs have to review disposal protocols frequently, as improper handling of fluorinated residues hits communities downstream. For any new lab planning to use this material, strong waste management policies aren’t an afterthought—they’re a requirement.
Looking Ahead: Smarter Chemistry
No compound delivers silver-bullet solutions. Still, heptafluorobutyric anhydride plays a real part in today’s fast-moving research world. Developing greener alternatives makes sense, but for now, the focus needs to stay on improving lab handling, investing in training, and updating procedures. This way, chemists can keep pushing analytical techniques forward with the confidence that their practices protect both their results and their communities.
Not Your Average Chemical
Heptafluorobutyric anhydride has a complicated name, but it’s got a pretty clear attitude. If you’ve worked around strong acids or handled reactive chemicals, you already know to pay attention. A compound like this doesn’t forgive mistakes, and it makes its presence known quickly with pungent fumes and strong reactivity. Why does storage matter so much? Every bottle in the lab holds potential risk or reward, depending on how you treat it.
Keeping It Contained
I remember my first encounter with a bottle of heptafluorobutyric anhydride. An old-timer in the lab tossed me the safety sheet and said, “No shortcuts.” The biggest risk is its sensitivity to moisture. Water vapor in the air can slip past seals and start breaking it down, turning it into corrosive acids that’ll chew up glassware or worse. Most laboratories store it in tightly sealed containers, usually glass with Teflon-lined caps, to keep the reaction from starting before you’re ready.
Temperature: The Quiet Enemy
Left on an open shelf beside the coffee maker, heptafluorobutyric anhydride won’t play nice. Heat pumps up its volatility and ramps up the risk for uncontrolled reactions or leaks. Dedicated storage in a cool, dry chemical refrigerator makes life easier. Keep it out of the sunlight, and away from radiators or any warming devices. Accidentally warming up a reactive anhydride could endanger not just the person handling it, but everyone nearby.
Neighbor Problems in Storage
Some chemicals keep to themselves. Heptafluorobutyric anhydride is not one of them. It needs distance from bases, water, strong oxidizers, and alcohols. Accidental mixing—even by proximity or vapor—has a way of causing toxic fumes or dangerous heat. Storing this chemical in a designated acidic organics cabinet, separated from incompatible substances, keeps the peace in the chemical world. Anyone who’s seen a storage mix-up knows just how fast a problem can escalate.
Labeling and Personal Responsibility
It’s easy to get sloppy with labeling, but a clear label can be the last line of defense. In one case I saw, a mislabelled anhydride almost found its way into an aqueous waste container. That mistake could have led to corrosive hydrofluoric acid and some panicked phone calls. Good habits protect not just your own skin but everyone in the workspace. Strong labeling, regular checks for corrosion or leaks, and not sticking old containers at the back of a crowded shelf go a long way.
Solutions for Safer Practices
I’ve seen some labs save themselves a world of hurt by investing in quality storage cabinets and proper training. Storage guidelines aren’t just there for new hires; even veterans benefit from fresh reminders and better practices. Automation in environmental controls, like humidity monitors and temperature alarms, really helps busy people catch problems before they spiral. Ordering smaller quantities, rotating stock, and quick reporting of any questionable container can head off disaster.
Final Thoughts
A clear, straightforward approach serves best. If you treat heptafluorobutyric anhydride with the respect it earns, it will do its job without accident or drama. Protecting coworkers and the environment takes simple care and steady attention, not just on lab inspection day, but every day.
Most folks outside of science labs probably haven’t heard much about heptafluorobutyric anhydride. Even those who work in research don’t often stop to consider what happens if this chemical ends up where it shouldn’t. Stashing bottles in a cabinet and donning gloves are everyday habits, but there’s more to the story when a chemical carries risk.
Everyday Safety Isn’t the Whole Picture
Heptafluorobutyric anhydride pops up in labs that work with pharmaceuticals or advanced chemical analysis. Its main job is tweaking molecules so instruments like mass spectrometers can read them better. The strong fluorine backbone that gives it power also raises questions about health and environmental impact.
I’ve seen plenty of chemists treat small bottles of something as “just another reagent.” The reality hits when a drop lands on the glove or a whiff escapes the fume hood. You remember pretty quickly that strong acids or aggressive fluorinated agents don’t ask nicely when they irritate your skin or eyes. I’ve had gloves turn brittle after a brief contact with similar chemicals. Good ventilation and goggles protect you, but lab routines can give a false sense of control.
Hazards Worth Respecting
Direct contact with heptafluorobutyric anhydride can burn skin and eyes fast. The vapor isn’t just another bothersome smell, either. Breathing in the fumes can spark coughing, choking, and sore throats. Over time, regular exposure to strong perfluorinated chemicals could stress the lungs and cause lingering health troubles.
I’ve heard stories from colleagues about chemical burns that didn’t heal for days. Many are lucky those stories just involve mild exposure. Spills on clothing carry the risk of delayed burns—what feels like a quick splash can keep reacting with skin until you clean it off thoroughly.
Outlasting Us in the Ecosystem
There is another concern: persistence. Fluorinated substances tend to stick around in the environment long after research has moved on. These “forever chemicals” resist natural breakdown, and scientists worry they slip from waste streams into rivers and soil. Some perfluorinated compounds cause harm far down the food chain.
The Environmental Protection Agency has flagged the persistence and potential toxicity of many fluorinated chemicals, linking some to hormone disruption or organ stress with chronic exposure. Consumer products and industrial waste both add to the load, compounding small lab spills or improper disposal.
Steps Toward Safer Practice
Working with agents like heptafluorobutyric anhydride deserves more than routine caution. Chemical users should push for better training about handling, disposal, and what personal protective gear really stands up to these agents. Disposal programs that track waste—avoiding drains and regular trash—cut risks for workers and wildlife alike.
Replacing persistent chemicals with safer alternatives could shrink risk over the long haul. Not every reaction has a green substitute today, but the push for less toxic and more biodegradable options is growing. Policy support helps, but labs and companies have to prioritize safety and environmental health on their own, too.
Remembering a chemical’s hazards—beyond the label—keeps scientists safer, and cuts down on silent risks to everyone else sharing the planet. The best outcome isn’t just a successful experiment, but leaving as little behind as possible.
Anybody who’s ever worked in a chemical lab knows there’s a unique smell that comes from certain reagents. It sticks in your memory and reminds you of the need for caution long after you’ve left the building. Heptafluorobutyric anhydride fits snugly in that category—a compound that demands respect and careful handling.
The Risk is Real, Not Theoretical
This is not the sort of stuff you want on your skin or in your lungs. I remember my early days in the lab, gloves slightly loose and a small splash that thankfully hit my lab coat, not my arm. Heptafluorobutyric anhydride is highly reactive. It can damage tissue in seconds. The eyes take top priority: a single drop could mean a trip to the emergency room. Inhaling just a few fumes can irritate your respiratory system. These aren’t minor inconveniences. The chemical’s reactivity with water, including humidity in the air, produces corrosive fumes that burn and corrode. The threat isn't exaggerated in the safety data sheets. It punches right through mistakes and inattention.
The Barrier Gear Isn’t Just for Show
Personal protective equipment has saved more than a few hands and faces, including mine. Splash goggles, gloves made of neoprene or nitrile, a proper lab coat—these are basics, not extras. Relying on the idea that “I’m just handling a small amount” has put people in ambulances. Most chemical mishaps begin with small spills, not dramatic accidents. Heptafluorobutyric anhydride leaks or vapors in poorly ventilated spaces cause wider exposure than you’d think. A functioning fume hood isn’t optional. Even for one quick transfer, open-air handling courts disaster.
Storage Lessons Learned the Hard Way
I’ve seen careless storage create chaos weeks down the line. Moisture creeping into a poorly sealed container, corrosion forming inside, vaporized acids filling a storage cabinet. It costs more in clean-up and lost sleep than it does to follow some common-sense rules. Store this compound in tightly closed, compatible containers in a dry, cool place. Glass with Teflon-lined caps usually does the trick. Forgetting to label containers properly can lead to confusion later, and confusion with reactive chemicals multiplies risk. Sort your labels carefully, and never let containers sit in open air longer than necessary.
Training Makes the Difference
Training can feel tedious, especially for experienced hands. The temptation to skip quick refreshers sounds familiar to anyone who’s spent years in chemical environments. People who’ve actually made mistakes become great storytellers, and their experiences serve as warnings worth more than a checklist. A short demo showing the reaction with water or a case study from an actual lab accident leaves a stronger memory than a hundred emails from safety officers.
What Works—and What Needs Changing
Addressing safe handling isn’t about adding more rules for the sake of it. It means taking the time to check that the basics—the right gloves, working ventilation, clear labeling, proper containers—happen every time, without exception. Facilities that foster open reporting of near-misses end up safer for everyone. Regular walk-throughs and the chance to talk through mistakes in a supportive way build a culture where people think before they act. Inconsistent policies and lack of real-world training cause trouble. Pairing clear procedures with hands-on experience changes behavior fast.
Working with powerful reagents like heptafluorobutyric anhydride calls for focus, not just expertise. One rushed moment can undo years of safe practice. It’s about protecting people, not just ticking boxes. Safety for dangerous chemicals starts with respect, not fear, and it ends with a team looking out for each other every step of the way.
Ask a few chemists about Heptafluorobutyric Anhydride and most will agree: this isn’t the sort of chemical you stash in a corner cabinet and forget. It’s a power tool in the analytical chemist’s kit, and it can help crack tough cases in pharmaceutical research or environmental testing. But there’s a catch: once you pop open the bottle, the clock starts ticking. Over the years working in analytical labs, I’ve seen more than a few stocks of specialty reagents degrade—sometimes subtly, sometimes catastrophically. It’s easy to underestimate how this can skew results and waste both money and time.
Why Shelf Life Actually Matters
For Heptafluorobutyric Anhydride, those little things lab veterans stress over—like moisture in the air and cap closures—make a big impact. Its reactivity with water causes it to slowly turn into heptafluorobutyric acid. Run a reaction or a sensitive chromatographic analysis with stuff that’s sat out too long and you’ll start seeing lower yields, strange peaks, or even ruined columns. Anyone who’s had to troubleshoot an HPLC baseline drift after using a compromised batch knows the headaches that come with taking shortcuts on storage.
What’s Really Going On With This Stuff?
This isn’t about bureaucracy. The molecular structure of Heptafluorobutyric Anhydride makes it eager to snatch up every water molecule it can find, even from supposedly dry air. In commercial practice, the unopened shelf life usually hits the one-year mark if you keep it sealed and cool. Open the bottle, though, and you’re dealing with a much shorter window—typically a couple of months before you see signs of hydrolysis. I’ve seen analytical grade material keep for three months with careful use and storage, but I wouldn’t bet important data on it past that.
How to Make the Most of Your Stock
Practical tips beat fancy storage protocols every time. Store Heptafluorobutyric Anhydride in a tightly closed, original container. Keep it at low temps, ideally below room temperature. Some labs go the extra mile and split fresh bottles into small, single-use vials under nitrogen or argon. Silica gel and molecular sieves help ward off humidity, though nothing replaces vigilance. I always encourage new lab techs to mark the opening date right on the bottle with a permanent marker—easy to forget otherwise. Once opened, plan to use the remainder quickly for critical assays.
What Happens When You Push the Envelope?
Labs with tighter budgets try to eke out every last drop, but the result is often frustration. When samples start failing quality control or hints of hydrolysis slip into sensitive analyses, the answer is almost always freshness. Even if the material still looks clear, those hydrolysis products aren’t always visible, but they do show up where it hurts most: in precision and reliability. Big pharma and most compliance-driven operations don’t take chances and routinely replace opened bottles every couple of months. For smaller outfits, it helps to plan projects to synchronize with purchase dates and batch prep to minimize waste.
Moving Forward, Fewer Surprises
Heptafluorobutyric Anhydride isn’t unique in facing a ticking clock, but its rapid hydrolysis drives home the bigger lesson: chemical shelf life isn’t just a box to check—it’s a matter of honest results. Investing the time to store it right, track usage, and build a culture of preventive replacement doesn’t just keep the work cleaner. It keeps the trust in the data you publish or the products you release, which at the end of the day, really does matter the most.