Hexafluoroisopropyl Methyl Ether: An Editorial Commentary on a Modern Chemical
Historical Development
Chemicals often carve their stories across decades, sometimes even centuries. The journey of Hexafluoroisopropyl Methyl Ether stretches back to an era when organic chemists started exploring ether derivatives in search of better solvents, anesthetics, and reaction media. Early curiosity about the effects of fluorination on ethers pushed scientists to investigate structural changes and their impact on boiling points, toxicity, and resistance to chemical attack. As fluorination technologies took off in the late 20th century, the need for specialized solvents in electronics, pharmaceuticals, and even green chemistry paved the way for this compound's wider adoption. Many modern applications came about because researchers refused to settle for the limitations set by hydrocarbon solvents in processes where high performance and safety matter.
Product Overview
Hexafluoroisopropyl Methyl Ether steps out from the crowd of ether solvents for several reasons. Building on the properties inherent to perfluorinated chemicals, this compound rarely gets beaten when dealing with scenarios that call for low reactivity coupled with high volatility. Chemists in both industry and academia note its performance for tasks ranging from serving as a reaction medium in tricky syntheses to acting as a cleaning fluid where contamination poses big risks. Not everyone realizes how much innovation hinges on the right solvent; more often than not, the difference between a failed and a successful experiment in a lab or production line comes down to small changes like these.
Physical & Chemical Properties
The six fluorine atoms joined to the isopropyl group toughen up the molecule, lowering flammability and making it stubbornly resistant to acids, bases, and oxidation. Its molecular structure helps push its boiling point down, so recovery and removal from product streams gets easier—no small thing for high-value manufacturing. A careful look at density, vapor pressure, and solubility shows why it fits specialized extraction, separation, or cleaning tasks better than classic ethers such as diethyl ether or methyl tert-butyl ether. Its vapor is heavy; its liquid, colorless. Anyone who’s worked in a chemistry lab learns to appreciate a solvent that evaporates cleanly, leaves little residue, and resists breaking down at the first sign of heat or contamination.
Technical Specifications & Labeling
Regulatory agencies continue to focus on how chemicals are labeled and handled, especially those with any degree of toxicity or volatility. Hexafluoroisopropyl Methyl Ether doesn’t get a free pass. Standards today call for suppliers to provide clear data on purity, recommended storage temperatures, and safe handling instructions. Proper labeling isn’t a chore or afterthought; mistakes at this stage can ripple through supply chains and end up causing real harm. In my own lab days, I saw how correct labeling averted near-misses by alerting people to reactivity hazards or special storage needs.
Preparation Method
Production starts with fluorinated precursors. Historically, researchers have relied on catalytic fluorination or electrochemical fluorination methods, which involve high skill and careful process design. Some synthesis routes combine perfluoroisopropanol with methyl halides under basic conditions, squeezing out the ether through SN2 mechanisms. Byproducts never stay out of the limelight, so controlling reaction parameters matters as much as the starting materials do. Shifts in environmental standards challenge chemists today to trim waste, recover raw materials, and move away from legacy solvents that create a mountain of compliance headaches.
Chemical Reactions & Modifications
Stubborn stability is both an asset and a hurdle. Confronted with strong acids or bases, this ether shrugs and usually holds its ground. That means it often gets used as a solvent rather than a starting point for further modification. On rare occasions, selective cleavage or substitution can be coaxed, but only with aggressive reagents or under nontraditional conditions. Most research in the field sticks to using it as an inert carrier, not a chemical workhorse. Synthesis chemists know the pain of a solvent that joins instead of watching from the sidelines, often leading them to this molecule as the answer.
Synonyms & Product Names
Names can confuse even seasoned researchers. Along with “Hexafluoroisopropyl Methyl Ether,” the chemical answers to several synonyms, including "Sevoflurane precursor" in anesthesia circles or "HFIME" in technical literature. This can trip up ordering, especially when commercial catalogs use trade names that sound nothing like the IUPAC convention taught in universities. Mistakes get expensive fast when the wrong grade or form shows up at the receiving dock.
Safety & Operational Standards
Industrial solvents always carry a level of risk that calls for respect. Hexafluoroisopropyl Methyl Ether isn’t highly flammable, but its volatility poses inhalation hazards. Exposure limits, even if not strictly regulated everywhere, shape how companies build engineering controls into production lines and labs. Good ventilation helps, as do leak detection and spill kits—lessons learned after seeing one too many headaches from lax PPE rules. Long-term tracking of symptoms in workers reminds us that no solvent is “safe enough” to skip training. Buying the right fume hood might carry a price, but skimping shows up later on hospital bills or regulatory fines.
Application Area
Most folks outside the industry never hear about this chemical, even though their electronics, medical devices, or pharmaceuticals may depend on its properties. Fluorinated ethers such as this one see service as solvents for extreme conditions, resist developers in microelectronics, carrier fluids in precision cleaning, and as building blocks for advanced anesthetics. The specialty market for semiconductor cleaning has driven up demand, thanks to the quest for ever-smaller feature sizes where chemical purity affects yield. In my professional experience, the right solvent can eliminate days of troubleshooting contamination, swinging a whole production schedule back into line.
Research & Development
Lab groups love pushing boundaries, often finding new uses for chemicals thanks to alliances between academia and industry. Hexafluoroisopropyl Methyl Ether gets studied for potential in green manufacturing, where high volatility aids recovery but its chemical stability reduces unwanted byproducts. The need for sustainable solvents keeps growing, especially in electronic component fabrication and next-generation batteries. Teams keep refining synthesis methods, searching for catalysts or processes that cut fluorine waste and energy consumption—progress that both saves money and lessens the environmental hit.
Toxicity Research
Most solvents sooner or later face a reckoning for toxicity and environmental hazard. Research so far points to relatively low acute toxicity in controlled exposure scenarios, but chronic effects and bioaccumulation still draw cautious attention. Regulatory agencies like the EPA and REACH keep an eye on production volumes, disposal practices, and emissions data to prevent persistent pollution. Chemical toxicity takes years to unravel; public health depends on long-term animal studies and monitoring programs to surface any risks hidden in everyday use. Lessons from similar ethers—both successes and disasters—influence how safety protocols form and change. No lab technician or plant worker wants to learn about health risks only after a lifetime of exposure.
Future Prospects
Application growth for Hexafluoroisopropyl Methyl Ether likely won’t slow down as electronics get smaller and chemical processes grow more demanding. Trends in green chemistry and regulatory pressure will push researchers to develop safer, easier, and cleaner production methods. Companies want solvents that tick every box—performance, safety, recyclability, low emissions—without busting budgets. There’s room for big improvement in recovery systems and waste handling, especially where high-purity demands meet strict environmental goals. I expect new breakthroughs from interdisciplinary teams that mix expertise in chemical engineering, toxicology, and environmental science to close the gap between performance and safety. Chemicals like this one remind us that nothing in industry happens in isolation. Each improvement ripples out, shaping everything from personal safety to the sustainability of our modern way of life.
Why Chemists Value Hexafluoroisopropyl Methyl Ether
Chemistry has ways of surprising the average person. Hexafluoroisopropyl methyl ether doesn’t roll off the tongue, but chemists appreciate the name for what it represents: a specialized, high-performance solvent and reagent that plays a quiet but important role in labs and industry. Unlike everyday solvents like ethanol or acetone, this compound brings a special touch to processes that demand both power and finesse.
The Role in Modern Chemistry
Organic synthesis counts on specialty ethers, and this one stands out. Its structure — bristling with fluorine atoms — gives it impressive chemical stability and low reactivity, which means it doesn’t easily break down or interfere with reactions it's not supposed to be involved in. This makes it an attractive option when working with sensitive or highly reactive compounds that could fall apart if faced with water or alcohols.
From experience in a university lab, small differences in solvent choice can make or break an experiment. Hexafluoroisopropyl methyl ether lets researchers push boundaries with organometallic chemistry, where traditional solvents fall short. Strong bases and nucleophiles, often used in drug synthesis or advanced materials research, won’t chew through this compound. That’s a game-changer, cutting down on failed reactions and inconsistent yields.
Impact on Pharmaceuticals and High-Tech Materials
Pharmaceutical companies see this ether as more than a niche material. When making compounds for new medicines, purity and reproducibility mean everything. Ordinary solvents often invite contaminants or set off side reactions, ruining batches months in the making. This fluorinated ether makes those worries fade — it doesn’t react with key intermediates, resists moisture, and can dissolve stubborn organic matter.
Electronics companies also use this ether for cleaning and manufacturing high-value semiconductors. Picture residues that refuse to budge after a manufacturing run, threatening to lower yields or disrupt new technology launches. Hexafluoroisopropyl methyl ether tackles these residues without corroding delicate components, offering a safer route compared to harsher alternatives.
Weighing Environmental and Health Factors
Specialty chemicals can’t escape questions about safety. Fluorinated compounds build up in the environment over time. It’s easy to gloss over this concern, but we’ve seen what unchecked chemical use can do — stories of groundwater contamination don’t fade fast. Chemists have a responsibility to track disposal, minimize use, and look for greener alternatives when possible. A single lab may use tiny quantities, but large-scale production brings risk closer to home.
Potential Paths Forward
Innovation means looking for solutions that meet tough performance demands without leaving a scar on the environment. Alternatives to heavily fluorinated ethers won’t appear overnight, but chemists keep hunting. Simpler molecules, recyclable solvents, or greener synthesis routes can lighten the load. Until then, careful handling and transparency about chemical use stay essential. Working with advanced chemicals like hexafluoroisopropyl methyl ether opens doors in research and manufacturing, but responsibility follows every beaker.
The Realities of Handling Strong Chemicals
Hexafluoroisopropyl methyl ether isn’t some everyday material you’d find under the kitchen sink. Instead, this chemical shows up in research labs, specialty manufacturing, and places where people work hands-on with solvents and high-performance compounds. Anybody working around it, myself included, knows that strict safety routines protect both bodies and reputations.
Understanding Risks
Hexafluoroisopropyl methyl ether vaporizes quickly and packs a punch both for what it can do and what it can do to you. I’ve seen colleagues take shortcuts while believing a little exposure won’t hurt, only to end up coughing or complaining of headaches by the day’s end. Breathing its vapors can irritate the lungs, and skin contact may cause burns or rashes. Getting complacent leads to accidents, and, from what I’ve witnessed, the difference comes down to approach: those who treat every transfer as routine versus those who remember why we have rules in the first place.
Learning from Experience
A lab coat on its own doesn’t keep you safe. Proper gloves, face shields, and chemical-resistant aprons actually stand between you and real harm. I learned early that splash goggles beat regular safety glasses every time—splashes follow their own rules, and eye injuries stick with you. Working in a fume hood isn’t optional. Ventilation isn’t just about comfort; it can be the line between a routine task and a dizzy spell or something worse. Too many people underestimate good airflow. Moving away from busy traffic areas also helps reduce distraction and bumps, especially with volatile chemicals on the bench.
Spills, Storage, and Simple Discipline
Many accidents, in my experience, involve storage shortcuts. Leaving a bottle of hexafluoroisopropyl methyl ether uncapped or settling for a container not designed for aggressive solvents can turn a quiet lab into a disaster zone. Flammable storage cabinets and tight seals keep people safe. Labeling matters too, even on long days when grabbing whatever is closest seems easier. I can't count the number of times an unlabeled flask forced the entire lab to stop and double-check what was inside.
Preparedness Beats Luck
I always review safety data sheets before working with new chemicals, not out of bureaucratic habit, but because a single missed detail — like water reactivity or incompatibility with other solvents — can trigger nasty surprises. Emergency showers and eyewash stations are only useful if they remain clean, unblocked, and regularly checked. Training everyone on what to do in case of exposure — not just reading procedures once, but running real drills — turns frozen confusion into confident action if something goes wrong.
Open Communication and Team Culture
A culture of speaking up defines safe labs. If I spot someone handling something dangerously or skipping personal protective equipment, I say it right away. It’s not about policing; it’s about everyone going home unscathed. Open communication, regular training updates, and a willingness to learn from small mistakes keep complacency at bay — and, from what I’ve seen, they create workplaces people trust.
Looking Forward: Better Habits, Fewer Accidents
Lack of respect turns strong chemicals from helpful tools into serious threats. The solution comes down to practical training, real teamwork, and prioritizing safety over speed. With vigilance, clear storage, and a community that values caution, working with hexafluoroisopropyl methyl ether — or any hazardous chemical — gets safer for everybody involved.
Why Chemistry Shorthand Matters Beyond the Lab
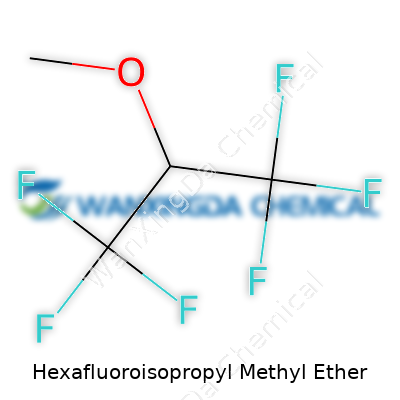
Hexafluoroisopropyl methyl ether, with its formula C4H5F6O, pops up in a corner of science most people rarely think about—industrial solvents and cutting-edge electronics. At first glance, a string of characters like C4H5F6O just looks like codes from a textbook. But these symbols translate to real impact in the world. Each letter means something—not just to chemists, but to anyone who relies on modern devices, clean energy, or even clean air.
Chemical formulas aren’t only for academics. They’re like blueprints. C4H5F6O tells researchers exactly how many carbon, hydrogen, fluorine, and oxygen atoms make up Hexafluoroisopropyl methyl ether. Picture trying to cook a complicated meal and someone tells you only what the final dish tastes like, not what goes inside. That missing info causes confusion. The same holds true in research labs, factories, and even policy offices. A single digit off in a formula can spell disaster when safety’s on the line.
Why This Ether Stands Out
Now let’s talk about fluorine. In the world of chemistry, adding fluorine doesn’t just tweak a formula—it rewrites how a molecule behaves. Hexafluoroisopropyl methyl ether isn’t your run-of-the-mill ether. A big wall of six fluorine atoms gives the ether properties regular solvents can’t match. Think extremely low reactivity, greater resistance to breakdown, stability in the face of acids and bases, and a lower risk of fire. This means in industries where even a hint of instability can threaten a multi-million dollar process, this chemical becomes a sort of insurance policy.
But industry isn’t the only concern. Fluorinated chemicals have a way of hanging around. Communities near certain manufacturing plants have seen the impact of persistent environmental chemicals. I grew up where the river ran brown during storm runoff, and the fish caught warnings from health officials. Even though Hexafluoroisopropyl methyl ether isn’t one of the forever chemicals making headlines, its relatives draw scrutiny. Responsible manufacturing and disposal connect straight to community health.
Why Details in Formulas Build Trust
Anyone who’s spent time in a classroom knows the frustration of unclear lessons. The same applies to chemical transparency. Sharing clear information—including full formulas—builds confidence for regulators, customers, or other researchers. Trust grows when industries put their cards on the table. This can reduce suspicion and even foster partnerships across sectors that wouldn’t usually talk to each other.
There’s room for improvement. Companies can step up their communication game, not hiding behind jargon, and policymakers can back smarter disclosure rules. Schools can lean on real-life case studies to show why these formulas matter. Open access to clear information about chemicals in use—or being replaced—lets people make better choices, whether they run a lab or just care about their home environment. Addressing the full lifecycle and possible impacts shouldn’t feel like a homework assignment. It keeps science grounded, public trust higher, and lets us use powerful compounds like Hexafluoroisopropyl methyl ether with fewer regrets.
If you spend any time in a laboratory or industrial setting, you know organization and safety go together. Hexafluoroisopropyl methyl ether, with its specialty uses in chemistry and manufacturing, brings its own set of challenges. As someone who’s spent a career navigating chemical storage rooms, I’ve seen what proper storage looks like—and what happens without it.
Why Storage Decisions Matter
This compound packs volatility. It earns respect for its strong vapors and possible risks when care slips. People have stories about damaged seals, unexpected leaks, or even fire scares. Overlooking good habits isn’t just about fines; it puts people in harm’s way and risks research goals. The National Fire Protection Association ranks similar ethers as hazardous partly due to their flammability and reactivity with air or moisture. A forgotten bottle on a shelf, sitting near the wrong compounds, can become an accident nobody sees coming.
Best Practices from the Trenches
The best chemical storage rooms feel more like a chef’s kitchen than a closet. Every bottle gets a spot for a reason. With hexafluoroisopropyl methyl ether, you want a cool, dry location. This isn’t just copying a manual—heat makes vapors build pressure, which can push out through caps or crash an experiment. I’ve seen student labs disregard temperature controls, only to face corroded storage shelves and unpredictable fumes later.
Storing this ether in a flame-proof, ventilated cabinet puts a barrier between it and anything that could ignite a fire. Don’t cut corners with plastic bins or leftover shelving material. Steel cabinets, equipped with self-closing doors, don’t warp during small fires or chemical reactions. Ventilation isn’t negotiable either. Standing air lets fumes settle, and these accumulate fast. Chemical-rated exhaust or filtered vent systems carry away what the nose can’t detect—essential after hours and on weekends, when nobody’s around to notice a leak.
Separation and Labeling Safeguards
No one should treat all ethers as equals. Hexafluoroisopropyl methyl ether needs isolation from acids, bases, and oxidizers. Mixing up these storage rules almost guarantees trouble down the line. In my experience, careless stacking or labeling shortcuts open the door to cross-contamination. Strong, color-coded labels help everyone know what sits where, even during stressful inventory checks. Details on the label offer clarity, not just for full-time staff, but for students and visitors who cycle through workspaces over the year.
Fighting Complacency with Training
Practical training makes the big difference. Written checklists and hands-on refreshers pull everyone into the same safety culture. Too often, new staff inherit old habits—or worse, follow routines designed for less sensitive chemicals. It sounds basic, but a walkthrough every quarter, paired with open discussion about what’s working and what isn’t, keeps accidents to a minimum.
Investing in Redundancy and Monitoring
Backup measures matter a lot more than people like to admit. I’ve seen alarms prevent evaporative loss from ruining months of careful work. Installing spill containment trays in cabinets, pairing chemical storage with real-time vapor monitoring, and making sure emergency supplies stay replenished, all add layers of security. You can’t predict every scenario, but you can control your preparation.
Every responsible step with storage pays off in peace of mind. This becomes especially obvious the first time a sensor goes off late at night and you return in the morning to find your precautions saved not just product, but people too.
Overlooked Dangers in the Lab and Beyond
Walk into any research lab, and you’ll see plastic tubing snaking around fume hoods, flasks bubbling away, and a row of bottles marked with names that would stump a spelling bee champion. Hexafluoroisopropyl methyl ether might look like a mouthful, but in specialized industries, people know it as a workhorse solvent for specific reactions.
Chemical exposure rarely makes the front pages, but every year, professionals and technicians develop ailments linked to the vapors they breathe or the splashes that catch their skin. Health authorities and researchers have flagged many ethers as respiratory hazards or skin irritants. Because hexafluoroisopropyl methyl ether is still new compared to old-timers like diethyl ether, there’s a gap between how much we know and what we guess. But from experience, no volatile ether should be treated casually.
Breathing in Trouble: Inhalation Hazards
Ethers with high fluorine content, including this one, evaporate fast and fill a space with fumes before you smell a thing. That quick vaporization means you can feel dizzy or lightheaded faster than expected in a badly ventilated corner. Common sense, and the CDC’s stance on related ethers, link repeated inhalation to headaches or fatigue. Overexposure can irritate the airways, make it hard to concentrate, or in severe cases, depress the central nervous system.
I once worked alongside a colleague who unknowingly handled a less benign ether all afternoon with a faulty hood. He thought he was just overtired, but by the end of the shift, he could barely drive home. Too often, symptoms like this are blamed on a late night or too much coffee, but ignoring the risk doesn’t make it disappear.
Skin and Eye Contact: Not a Harmless Splash
A splash of solvent rarely feels serious at first, but ethers tend to dry out skin or cause redness and itching. Fluorinated compounds carry a risk of deeper tissue irritation compared to simple alcohols. Most lab workers reach for gloves, but how many check that their nitrile gloves aren’t degraded by strong solvents? Casually handling spills without the right gear turns minor accidents into something that might keep you from working the next day.
Ethers in the eye cause instant pain and demand a fast rinse. Every safety drill focuses on the eyewash station for good reason. Once, after a careless uncapping, I felt a drop flick up – pure luck meant it grazed my cheek and not my eye.
Chronic Exposure: A Long Game Few Talk About
Short-term symptoms are bad, but low-level, repeated exposure builds up quietly. Little research tracks the long-term health trends of those handling hexafluoroisopropyl methyl ether, but studies on similar chemicals show problems like liver strain, kidney trouble, or heightened cancer risks over time. Workers have a right to know the risks before anyone falls ill.
Solutions: Smarter Safety Over Blind Trust
The answer starts with respect and ends with accountability. Good ventilation, enforced training, and real-time exposure monitoring create safer spaces. Employers investing in personal protective equipment send the message that every lab worker counts. Reporting spills, near-misses, and symptoms should never be shameful—it’s the responsible step to protect everyone.
Regulatory bodies and companies must sponsor independent studies on the health impacts of every new solvent. No one should expect workers to treat unfamiliar chemicals as safe until proven otherwise. Transparency beats speed every time when people’s health is on the line.