Indium Trifluoromethanesulfonate: A Live Wire in Modern Chemistry
Historical Development
Stories behind chemical compounds that disrupt scientific norms always catch my attention, and Indium Trifluoromethanesulfonate doesn’t disappoint. Back in the eighties and nineties, chemists struggled with setting up mild conditions for organic synthesis, and Lewis acids often caused headaches with moisture sensitivity or harsh byproducts. Then, somewhere in this cluttered tangle, indium salts started showing up, and researchers noticed their ability to kickstart reactions without leaving undesired traces. As demand for greener, more forgiving chemistry grew, Indium Trifluoromethanesulfonate began popping up in research that sought softer, yet still powerful, options—especially in the context of Michael additions or glycosylations.
Product Overview
Anyone tracking the evolution of organometallic chemistry will know Indium Trifluoromethanesulfonate by a few names—In(OTf)3, indium triflate, or, among the die-hards, its IUPAC mouthful. What sets it apart is its role as a catalyst and reagent that doesn’t demand a glovebox and five-year training in avoiding water. Commercial labs now reach for it because the compound handles robust transformations with relatively tame conditions compared to more notorious Lewis acids like AlCl3 or TiCl4. The material tends to present itself as a white powder, and though not flashy, it holds real weight in fine chemical, pharmaceutical, and polymer research.
Physical & Chemical Properties
Indium Trifluoromethanesulfonate doesn’t strut with wild colors or dramatic fumes. Typically, it stands as a white, crystalline solid that's soluble in polar solvents from water to acetonitrile. The chemical stays relatively stable under standard ambient conditions, which takes a bit of pressure off lab techs used to babying sensitive reagents. A major kicker for synthetic chemists is its ability to act as a strong Lewis acid without clobbering substrates or generating messy side-products, a feature that's hard to overvalue in high-stakes research or process chemistry.
Technical Specifications & Labeling
In my own practice, accurate information trumps marketing claims every time. With Indium Trifluoromethanesulfonate, content purity and solvent compatibility top the priority list for anyone ordering a batch. Packing lists usually describe water content, trace metals, and whether stabilizers show up. The shelf life stretches comfortably, and labeling practices emphasize correct hazard icons and any recommendations for storage away from acids or bases. Consistency of formulation matters, especially once scale-up enters the picture, but most suppliers catering to R&D provide transparent certificates of analysis that actually match what arrives on the lab bench.
Preparation Method
Rolling up sleeves and synthesizing Indium Trifluoromethanesulfonate fits a model any semi-experienced chemist can appreciate—the meeting point of a direct salt metathesis, typically starting from indium oxide or indium chloride and reacting it with trifluoromethanesulfonic acid. This approach sidesteps unnecessary complexity. I've noted that low water content in final products really influences catalyst performance; so researchers and suppliers often use vacuum drying and water-scavenging agents at the tail end to keep things tight. Most academic groups use protocol tweaks to tailor purity or yield, but the main pathway hasn’t changed much in a decade.
Chemical Reactions & Modifications
Chemists often use Indium Trifluoromethanesulfonate to drive Friedel–Crafts reactions, acetalizations, and glycosylations. Its unique edge over rivals like scandium or zinc comes from high functional group tolerance. It doesn’t bulldoze through sensitive substrates, so medicinal chemists working with fragile intermediates breathe easier knowing yields hold up without a forest of byproducts. Tuning the counter-anion or solvent opens up new territory; swapping in bulkier or less coordinating anions has nudged reactivity for specialty transformations, though In(OTf)3 remains favored when balancing cost, strength, and selectivity.
Synonyms & Product Names
Across publications, the same compound shows up under various names—Indium(III) triflate, indium tris(trifluoromethanesulfonate), and the more concise In(OTf)3. Switching between naming conventions sometimes causes confusion, especially with newcomers to the field. In collaborative research, setting standardized language early prevents the mix-ups that tend to slow down communication between chemists, regulatory staff, and procurement.
Safety & Operational Standards
Indium Trifluoromethanesulfonate isn’t among the nastiest chemicals you encounter, but nobody should skip PPE. In some experiments, fine dust causes mild irritation, and the triflate ion’s environmental persistence demands attention to waste disposal. Labs usually stress solid container closures and spill trays. Training on proper cleanup and containment is emphasized, especially after stories spread about reagents entering municipal waste streams. Some jurisdictions have started treating indium salts with regulatory oversight tied to both human health and aquatic life, and for good reason. It’s not explosive or violently reactive, but that doesn’t translate to carefree use—there's always potential risk in larger-scale applications.
Application Area
Indium Trifluoromethanesulfonate shows up everywhere from green chemistry pilot projects to medicinal molecule synthesis. I’ve watched it transform sluggish carbonyl chemistry in classrooms, and in more serious settings, it enables construction of glycosidic bonds vital for antiviral drugs. Companies scaling up specialty polymers use it as a polymerization catalyst where moisture and temperature sensitivity would kill productivity if other Lewis acids were involved. Its broad compatibility makes it attractive for custom syntheses—especially when high throughput and low impurity profiles are at stake.
Research & Development
Adoption of Indium Trifluoromethanesulfonate in academia and industry reflects a continued push for milder, less wasteful synthesis. Every year, emerging research articles point out new ways to trim down reaction times or access novel scaffolds, using tunable properties of indium-based catalysts. Some academic groups dig into hybrid catalysis by pairing indium triflate with transition metals, seeing upticks in reaction rates without harsh conditions. Meanwhile, pharma R&D focuses on chiral induction and greener processes. Not every experimental push ends in tech transfer, but consistent evidence shows this triflate complex leads to clean reactions with less purification pain.
Toxicity Research
Concerns about indium travel beyond the triflate itself. In occupational settings, chronic exposure to indium compounds linked with certain pulmonary issues prompted recent safety reviews. The triflate doesn’t fall into acute hazard categories like some alkylating agents or heavy metals, but prudent researchers still flag indium content for both short- and long-term toxicity. Recent animal studies raised eyebrows, prompting regulatory agencies to re-examine powders’ permissible exposure limits. Waste stream monitoring deserves attention too because indium traces, while small, can build up over time and disrupt aquatic life. Strong guidance on engineering controls and respiratory protection helps keep these hazards in check.
Future Prospects
Looking ahead, the demand for selective, recyclable Lewis acids points straight at compounds like Indium Trifluoromethanesulfonate. Researchers across Europe and Asia seek catalytic systems that avoid precious metals or environmentally harsh partners; indium triflate’s resilience in air and water fits this agenda. There’s talk of integrating indium-based catalysts in continuous flow setups—aiming to cut chemical waste and improve scalability. Green chemistry journals regularly feature studies comparing indium triflate with emerging bio-inspired catalysts, but its unmatched tolerance and straightforward handling keep it a staple in both fundamental and applied research. As manufacturing processes get leaner, the role of catalysts that strike a careful balance between effectiveness and environmental responsibility grows larger—making indium triflate an increasingly important player in the next wave of sustainable R&D.
High-Tech Chemistry for Real-World Progress
Indium Trifluoromethanesulfonate rarely makes headlines, but in modern chemistry labs, this compound does more than lurk in the background. For most folks, the name might sound like a tongue-twister, yet within the research and development field, this catalyst pushes boundaries across several sectors, especially in synthetic chemistry and electronics. Yet its real influence often gets drowned out by more familiar buzzwords like “semiconductors” or “green technology.”
Helping Chemists Build New Molecules
One reality that stands out: chemists count on reliable catalysts to speed up reactions and create products more efficiently. Indium Trifluoromethanesulfonate acts as a strong Lewis acid, which means it nudges certain reactions along that would otherwise crawl forward at a snail’s pace. I’ve watched organic synthesis projects get stuck without the right catalyst, then spark to life with the right chemical help. Scientists working on pharmaceuticals and specialized polymers often turn to this compound because it produces cleaner results and typically releases fewer side products than some older, less selective acids. Cutting waste matters as regulations around chemical manufacturing tighten worldwide.
Electronics and the Push for Miniaturization
Chemical research spills directly into technology. As devices shrink and demands for faster processing rise, makers need reliable materials and cleaner manufacturing routes. Indium-based compounds lend themselves to certain microelectronics applications, especially as companies search for alternatives to traditional and sometimes toxic substances. Indium Trifluoromethanesulfonate, thanks to its thermal stability and its knack for promoting chemical transformations, plays a role in producing electrical components, improved coatings, and sometimes even new-generation display tech.
Cleaner Chemistry: Not Just a Bonus
Green chemistry gets talked up a lot, but putting it into practice means finding substances that lower environmental risk. Many older reagents linger in the waste stream or raise red flags for toxicity. Indium Trifluoromethanesulfonate, compared to conventional strong acids, tends to leave a lighter environmental footprint in many key processes. My colleagues and I have watched labs prioritize this shift, both for the health of researchers and to meet stricter regulations. When you can get the same or better results with a safer catalyst, that’s progress.
What Could Change Moving Forward
Some challenges need attention. Indium isn’t an endless resource. It mostly gets pulled as a byproduct of zinc mining, and global demand for indium grows thanks to its use in electronics. Recyclability and improved recovery methods will shape the next steps in using indium-based catalysts. Chemists are exploring how to use these materials in smaller quantities or swap in alternatives when possible, but for now, Indium Trifluoromethanesulfonate holds a firm spot in several advanced labs.
Final Thoughts
Indium Trifluoromethanesulfonate doesn’t show up in daily headlines, but it’s a quiet workhorse driving innovation in chemistry and electronics. It makes manufacturing cleaner, reactions faster, and helps open up possibilities for safer commercial processes. As technology keeps shifting, the people behind the scenes searching for smarter materials will keep watching rare chemicals like this one, making sure the science pushes ahead without leaving a mess behind.
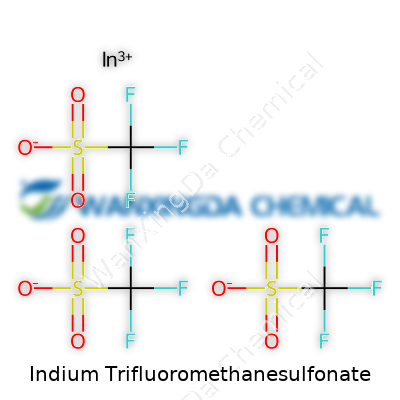
The Backbone Formula: C3F9InO9S3
Ask anyone working in synthetic chemistry about indium trifluoromethanesulfonate and most would blink and reach for the literature. For many, it’s a mouthful—both linguistically and chemically. But break it apart and you get something surprisingly neat: three trifluoromethanesulfonate anions paired with an indium ion, coming together in the formula C3F9InO9S3. This isn’t just a jumble of letters and numbers. It reflects real atoms joining forces, making something useful for a world that never stands still.
So, Why Does This Compound Deserve Attention?
Chemists introducing new methods in organic synthesis often search for catalysts that bring both efficiency and selectivity. Indium trifluoromethanesulfonate steps onto this stage with a lot to offer. Unlike many standard catalysts that lean on rare or highly toxic elements, this one brings indium into play—a post-transition metal you don’t see every day but appreciate when you find it. Its reliability in catalyzing reactions such as Friedel-Crafts acylations and alkylations has been researched and shown to reduce the need for harsher conditions or heavier metals. That matters in the lab and on an industrial scale, where waste and by-products never go unnoticed.
Practical Experience and Reliable Outcomes
Working in a laboratory, efficiency counts for more than fancy purity certificates. Indium trifluoromethanesulfonate stands up to the challenge. It doesn’t soak up water from the air like some other salts, and it tends to dissolve well in polar solvents. In straightforward terms, clean-up takes less elbow grease, and the yields hold steady. A friend in medicinal chemistry once told me about a project where this salt replaced a traditional Lewis acid, streamlining the workflow and actually improving the overall process safety—never something to overlook. These case studies aren’t rare; they echo across the research landscape, from pharmaceutical routes to flavor production.
Environmental and Safety Talking Points
Concerns about heavy metal waste have colored much of recent synthetic chemistry. Here, indium trifluoromethanesulfonate stands on somewhat firmer ground. Indium sits far below the toxicity lines drawn for more familiar metals like mercury or lead. The trifluoromethanesulfonate group, sometimes called triflate, is prized for its stability and resistance to breakdown, even under the stress of laboratory manipulation. The environmental burden, while never nil—since all metal salts can pose waste issues—seems more manageable than many alternatives.
Possible Directions for the Future
Chemical manufacturers and research teams looking to scale up can’t afford to ignore cost and sourcing. Indium isn’t as abundant as some base metals and price fluctuations happen, tied to the demands of the electronics industry. Efforts to recycle and reclaim indium from spent catalysts show promise. Broad adoption depends on practical recovery systems, rather than just throwing away spent material. Working with green chemistry in mind helps push the whole field forward, helping laboratories transition toward catalysts that don’t just work but leave a smaller mark on their surroundings.
Final Thoughts on Formula and Function
The chemical formula C3F9InO9S3 isn’t just abstract—it stands as a stamp on countless reactions where precision and progress matter. As regulations tighten and the drumbeat for sustainability grows louder, indium trifluoromethanesulfonate will keep earning its place in labs that need both performance and responsibility in sync.
Why We Care About Lab Safety
People work with advanced chemicals daily in research labs and industry. Sometimes flashy names like “Indium Trifluoromethanesulfonate” appear in synthesis directions or patents, often as a catalyst when making pharmaceuticals or specialty materials. But fancy names never guarantee something is safe. Experience tells me, caution always wins out in the lab, especially with less-familiar substances.
What’s At Stake with Indium Trifluoromethanesulfonate?
This compound serves as a strong Lewis acid, popular for helping chemical bonds form under mild conditions. There's no escaping the fact: this sort of capability usually signals reactivity. History with Lewis acids has shown me anything reactive deserves respect. It’s not just about what these chemicals do—they bring risks that some folks brush aside too quickly.
As for toxicity, hard data on indium trifluoromethanesulfonate is surprisingly sparse. No standard toxicology studies get cited in the literature. That alone tells a story: not everything in a glass bottle arrives with complete instructions or a safety scorecard. What we do know gets pieced together from similar substances and general indium chemistry safety sheets. Chronic exposure to some indium compounds has led to respiratory trouble and organ effects in animals. The “triflate” part—trifluoromethanesulfonate—doesn’t ease worries, either. Some “triflate” compounds bring corrosive potential or can irritate lungs and skin. Inhalation and skin contact both spell potential trouble.
Any time I handle an unfamiliar chemical, I watch for spill risks and airborne dust, even when there’s no hazard pictogram staring at me from the bottle. Minor skin contact might not send you running to the ER, but repeat exposure adds up over time. Indium compounds sometimes collect in organs, and evidence from workplaces handling indium-tin oxides links them to lung effects—“indium lung” hits hardest where chronic exposure persists. No study yet links this exact compound to cancer or birth defects, but the absence of proof doesn’t count as a green light. Someone always turns out to be the unlucky test case, and I don’t want it to be me or my students.
Protecting People and the Environment
Practical steps count for more than blind optimism. I always use gloves, splash goggles, and lab coats around compounds like this. Many researchers keep all indium salts in designated cabinets. Ventilation and fume hoods make up the frontline defense against inhaling dust. Good lab practices mean working on absorbent pads, cleaning spills quickly, and keeping unnecessary folks away. Like plenty of reactive materials, indium trifluoromethanesulfonate shouldn’t get anywhere near drinking water or regular trash. Waste collection and safe disposal by professionals prevent small exposures from building up in the environment.
On the policy side, I’d push for clearer guidance from chemical suppliers and workplace regulators. People deserve to know the risks without translating academic papers or manufacturer bulletins. Better labeling and open incident reporting help others learn before accidents repeat themselves. Suppliers who invest in safety data—testing for chronic exposure, acute toxicity, and environmental impact—help everyone.
Building a Culture of Respect
Lab culture rewards people who respect the unknown and look out for each other’s health. In my experience, the best teams treat every bottle as a mystery until proven otherwise. With chemicals like indium trifluoromethanesulfonate, that approach isn’t just smart—it’s essential. By expecting risk and planning for it, research can move forward without anyone paying a hidden price. Safety isn’t a sideline; it’s the bedrock of serious science.
Why Storage for Specialty Chemicals Demands Attention
Specialty chemicals like indium trifluoromethanesulfonate don’t get a lot of mainstream coverage, but in my experience, the details make all the difference in the lab. It’s easy to overlook how storage impacts the performance and safety of complex catalytic salts. Too often, the rush to innovate runs headlong into neglected safety habits. Proper storage isn’t just a regulatory checkbox—it’s about protecting people, the environment, and the bottom line.
Moisture, Heat, and Chemical Integrity: Practical Lessons
Chemists and technicians who handle indium trifluoromethanesulfonate know it absorbs moisture from the air. Exposure to humidity leads to clumping, reduced reactivity, or even chemical degradation. Past experiences taught me to never trust a shelf over a desiccator when stability matters. Keeping indium trifluoromethanesulfonate sealed tightly in an air- and moisture-proof container, with a good desiccant nearby, extends its usable life.
Room temperature might sound harmless, but this salt reacts poorly to extremes. I’ve seen overlooked bench-top samples lose their edge within days, compared to months or more in a controlled environment. A cool, dry spot, far away from heating vents, does more good than people realize. Even storing it in a spot with lower foot traffic helps, since constant room temperature swings—doorways, windows, busy benches—all nudge chemicals toward early breakdown.
Keep the Incompatibles Apart
Storing indium trifluoromethanesulfonate separately from strong bases and reactive organics helps prevent unwanted reactions. Back in my grad school days, a poorly sorted chemical fridge spelled disaster after a minor leak. Since then, chemical compatibility charts have guided every restocking. If your lab doesn’t have clear signs and segregated cabinets, it’s worth pushing for them; just a few minutes of prevention sidesteps costly mistakes.
Labeling: A Small Step That Changes Everything
I still remember reading about an incident where a white powder, left unlabeled on a general shelf, led to hours of confusion. Each one of these mishaps chips away at trust and costs valuable time. Clear labeling—date received, supplier, name, and any hazard symbols—cuts confusion and speeds up safety checks. Training newcomers to double-check containers before and after use creates habits that stick.
Solutions Rooted in Experience
Letting storage slide jeopardizes both safety and research. Smart labs adopt routine inspection schedules. My team sets a monthly calendar to check on desiccants, seals, and temperature logs. Reordering before stocks run low also sidesteps compromised chemicals. Using transparent recordkeeping, instead of relying on memory, ensures small details don’t slip through the cracks—especially in shared spaces.
Investing in proper cabinets, regular audits, and team training builds a safer, more reliable lab environment. The payoff comes not just in chemical stability, but also in fewer accidents, less waste, and better research results.
Ask any chemist who’s wrestled with stubbornly insoluble salts, and you’ll get an earful about the value of a compound that dissolves where and when you want it. Indium trifluoromethanesulfonate steps into the scene, drawing interest across labs for the way it tackles solubility—something that shapes its performance in many reactions. I remember early in my research days, struggling to coax reactions along with indium chloride or acetate. Switching to this indium salt opened doors that remained shut with the other options. The difference comes down to its remarkable behavior in polar solvents.
What Makes Indium Triflate Different?
The chemical structure helps. With three trifluoromethanesulfonate (triflate) groups attached to indium, the whole molecule tilts toward being less tightly bound together than most simple salts. That’s good news if you’re hunting for a source of indium ions in solution. The triflate group spreads out charge thanks to the electronegativity of its fluorine atoms and the resonance in the sulfonate, making the compound much friendlier with polar solvents.
Chemists report this salt readily dissolving in water, acetonitrile, and a range of alcohols. That stands in sharp contrast to indium halides, which might stubbornly sit at the bottom of the flask. In my own work, indium triflate handled high concentrations in acetonitrile—a solvent common in organic synthesis—without any drama. It also dissolved easily in methanol, letting me push reactions or clean up without adding extra heat or exotic procedures.
Why Solubility Matters in the Lab
A handful of facts make the value of this clear. Efficient catalysis and clean product isolation in organic chemistry depend hugely on how well metal salts behave in solution. If the salt won’t dissolve, you either force the issue with excess reagent, boost the temperature, or try to find creative workarounds. Each of those routes means more cost, more risk to sensitive functional groups, or potential loss of selectivity. Indium triflate changes the game by letting reactions run at mild conditions and at lower concentrations. Yields often improve just by being in the right solution at the right time.
This isn’t only about academic curiosity. Research published in peer-reviewed journals like Chemical Reviews and Journal of Organic Chemistry backs up these claims, showing indium triflate driving everything from carbon–carbon coupling to select hydroamination reactions. The fact that you can run these in polar media means less environmental waste, simpler work-up, and often, better reproducibility. That lines up well with the industry push toward more sustainable chemistry and the need for cleaner processes.
Addressing the Downsides
Despite the clear benefits, indium triflate costs more than basic indium salts. Part of that comes from the specialized production, and part from the performance it offers. Labs with tight budgets tend to weigh these factors carefully. Toxicity questions don’t usually top concerns, since both indium and triflate ions have moderate safety profiles. But researchers still respect the usual protocols: gloves, goggles, and fume hoods remain standard. Waste management matters—fluorinated organics, even in low concentrations, don’t vanish harmlessly, and responsible labs must plan for collection and proper disposal.
Practical Suggestions
If you’re troubleshooting low-yielding transformations or struggling with insolubility, it pays to test indium triflate in small-scale screens. Start with acetonitrile or methanol as solvent, since both dissolve it well and rarely interfere. Weigh out only what you need for immediate use—triflate salts can pick up water from the air, which changes their behavior. For greener processes, focus on recycling solvents, and always look for routes that minimize waste. Collaboration between labs—sharing tips, unpublished results, and data—often leads to smarter, more sustainable use. My best results came from cross-pollinating ideas with colleagues who’d faced similar hurdles. The payoff can be significant, both in cleaner reactions and in less time wasted battling solubility problems.