Lithium Bis(Trifluoromethanesulfonyl)Imide: New Backbone, Old Hurdles
The Story So Far
Years ago, the world of lithium salts was much simpler. Lithium perchlorate and lithium hexafluorophosphate ruled the roost in laboratories and battery production lines. Most folks outside electrochemistry never heard of Lithium Bis(Trifluoromethanesulfonyl)Imide—often called LiTFSI—until the push for safer and more efficient high-performance batteries turned into a global competition. LiTFSI didn’t just show up to play; it showed up with a different set of strengths. Designed in the late 20th century, LiTFSI became a key player as experts chased stability, temperature tolerance, and solubility that older salts struggled to provide. Anybody who’s spent hours behind a fume hood knows the edge a good electrolyte brings. In the forty years since LiTFSI first drew notice, its reputation has grown thanks to hard chemistry, not just clever marketing.
What LiTFSI Brings to the Table
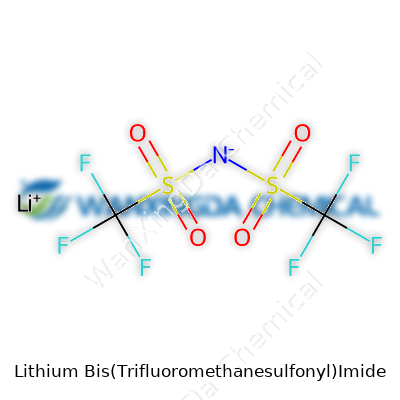
The physical looks of LiTFSI won’t turn heads—a fine white powder most days—but what matters happens on the molecular level. With the formula LiN(SO2CF3)2, it delivers a heavy punch to the electrochemical crowd. The TFSI anion, carrying two big sulfonyl groups, refuses to react in ways that trip up batteries or send safety engineers into a panic. In water, LiTFSI dissolves with readiness; in organic solvents, it rarely jitters, thanks to its bulky yet well-balanced structure. The thermal window stretches wide, so you can push your experiment further without watching the salt decompose. Scientists working with sensitive lithium batteries appreciate how this compound shrugs off moisture uptake and thermal drift.
Step-by-Step: Technical Needs and What’s on the Label
Nobody running quality control shoots blind when dealing with LiTFSI. Purity matters more here than with many other additives, since heavy metals or chloride traces punch real holes in cell performance and safety. Standard grades often clock in at over 99.9 percent purity; the best labs sweat over the last decimal point. Labels can seem crowded, listing everything from melting point to residual water content, but most end users care most about particle size, bulk density, and solution conductivity. Over the years, improvements in crystallization and dry room handling have put better LiTFSI at the world’s fingertips, though the industry still wrings its hands over cost and environmental impact.
Recipe for Synthesis
Synthesis of LiTFSI doesn’t bring magic, just hard chemistry and industrial scale. The standard method runs through reaction of trifluoromethanesulfonamide with lithium hydroxide or carbonate, then dances through recrystallizations to chase down impurities. This process tends to require well-controlled conditions, since unwanted side reactions can sneak through if nobody’s paying attention to stoichiometry or the pH balance. In my own lab experience, unwanted moisture or solvent residues can throw off yields or breed stability headaches, so tight procedural discipline saves headaches later on. Most large-scale producers now emphasize high-purity runs, given the market’s taste for cleaner, higher-performing salts—especially as regulatory eyes scrutinize every waste stream and effluent.
Reactions, Tinkering, and the Chemistry Classroom
Chemists love to poke and prod at salts to unlock new tricks. LiTFSI tends to play nicely with organic solvents and polymers. Poly(ethylene oxide) blends, ionic liquids, and other advanced electrolytes pair well with it, showing high ionic conductivity with low risk of unwanted side reactions. Anyone tinkering with new solid-state battery chemistries gets acquainted with LiTFSI. It doesn’t oxidize or reduce under typical battery voltages, so degradative side reactions stay quiet—if the salt’s pure, of course. Folks have explored partial substitution and mixed-salt formulations, hoping to push conductivity up and degradation down, but the core TFSI anion resists change, which in this case counts as a virtue.
Names and Nicknames
Researchers often trip over the mouthful that is “Lithium Bis(Trifluoromethanesulfonyl)Imide.” The abbreviation LiTFSI sticks in most papers and patents; some older literature still calls it lithium triflimide, a holdover from the days when naming conventions danced around molecular symmetry. A few suppliers slap on branded titles, but most labs stick with LiTFSI or its full IUPAC variation when clarity matters.
Safety on the Job and After Hours
Chemical safety officers dedicate a lot of memos to lithium salts like LiTFSI. This compound enjoys a reputation for being much less touchy than lithium perchlorate or hexafluorophosphate. Yet the trifluoromethanesulfonyl groups introduce their own hazards. Reliable evidence suggests acute oral and dermal toxicity lands on the mild side for most exposures, but contact can irritate skin and eyes. Chronic health impacts remain under-studied, so gloves, eye protection, and local ventilation remain the norm. Waste streams deserve careful handling; fluorinated organics carry persistence in the environment, and nobody wants to drink yesterday’s electrolyte downstream. Operational standards keep an eye on dust suppression, as airborne LiTFSI may irritate lungs, and every chemist learns to respect how lithium interacts with moisture and acids.
Battery Roots, Research Growth
LiTFSI gained fame powering lithium-ion and lithium metal cells, with the push for longer-lasting, safer, and higher-voltage batteries. Top universities and industry labs keep it at the center of electrolyte development, searching for ways to curb dendrite growth, widen thermal evacuation windows, and keep cell swelling at bay under high charge and discharge rates. Its stability pushes researchers into fresh ground, like battery architectures with solid electrolytes or flexible polymers. A look through the past decade’s journals turns up a steady swell of papers investigating LiTFSI’s solubility profiles, conductivity, and chemical compatibility. Pilots and startups branching into large-scale electric vehicles and energy storage slowly dip their toes into this salt, though supply chain issues tied to sourcing raw fluorochemicals limit overnight adoption.
Health Questions and Toxicology
Any new chemical that commands market share draws inevitable safety research. LiTFSI, on the surface, seems less noxious than other lithium salts, especially for those who recall the explosions and fires tied to lithium perchlorate. Early animal and cellular studies point to limited acute toxicity, though long-term chronic data remains scarce. Labs worry about the environmental fate of TFSI anions, given their persistence and mobility in water. Regulatory agencies turn a slow but steady eye to compounds of this kind, emphasizing the need for full life-cycle impact studies. Those of us used to working with potent organofluorines watch this debate with caution—past experience with Teflon and related substances taught us chemical stability means legacy in nature. Any large-scale adoption of LiTFSI will have to walk a tightrope between innovation and environmental stewardship.
Looking Ahead: Where the Road Leads
Demand for reliable battery electrolytes turbocharges research on LiTFSI. Electric vehicles and grid storage applications depend on stable, safe, and high-conductivity compounds, and today few contestants match LiTFSI’s resume. The future may see even purer grades, improved process recycling, and new co-solvents built around the TFSI core. Environmental scientists chase answers about the long-term fate of fluorinated byproducts; industrial chemists hunt for alternatives that cut costs and reduce ecological risk. Much of the future for LiTFSI hinges on regulatory comfort, raw material sourcing, and the balance between performance and sustainability. The world’s clean energy transition rests on rocks like this—a simple powder, judged by its chemistry and its costs, not just its promise. Those still grinding out new formulas in labs or hunting for fresh commercial edges know every salt tells a bigger story than just the catalogue number it carries.
The Unsung Hero in Battery Tech
People love to talk about batteries – the relentless search for longer runtimes, faster charging, and lighter gadgets. I’ve spent enough time fiddling with electronics to notice which chemicals keep coming up: lithium, cobalt, some obscure fluorides, and this oddly named one, Lithium Bis(Trifluoromethanesulfonyl)Imide, or LiTFSI. You won’t see it painted across phone commercials, but engineers know it plays a big role. Most folks, and even plenty of techies, don’t realize the challenges facing batteries until they stop working at a bad moment in life. I’ll never forget my old laptop shutting down in the middle of a conference because the battery gave up. Ever since, I’ve paid closer attention to what really affects battery life and safety.
Why LiTFSI Makes Such a Difference
The battery in your pocket probably depends on standard lithium salts, but that’s changing. LiTFSI stands out in the crowd for its stability. I first heard about this compound while chatting with a chemist frustrated by overheating phones. Heat can ruin batteries fast, and that’s where traditional lithium salts run into trouble—they break down or trigger reactions that leave devices hot or even unsafe. LiTFSI, on the other hand, doesn’t break down as easily. In practical terms, this means your device is less likely to overheat or catch fire. That small shift matters more than most people think. We’ve seen enough headlines about battery-related recalls to know this is not a theoretical concern.
Then there’s solubility. Chemists geek out of over this. If something dissolves well in the battery’s liquid electrolyte, electricity moves faster and smoother. Regular lithium salts sometimes struggle with this, especially at higher voltages. LiTFSI just dissolves better. End result: faster charging, less battery wear, cleaner performance. In an age where people want to keep gadgets out of the landfill, every little bit of added lifespan counts for consumers, and for the planet.
Beyond the Gadget: LiTFSI in Other Fields
The role of LiTFSI stretches beyond the obvious consumer tech. Researchers have started using it for grid-level energy storage—the kind of batteries that balance wind farms or backup city power. Here, stability turns into a cost-saving measure. A battery that resists breakdown needs replacing less often, cutting down on waste and long-term expenses.
It doesn’t stop with batteries. Some labs use LiTFSI in electrochemical applications, such as organic synthesis and supercapacitors. Its thermal stability and broad electrochemical window allow engineers room to experiment. Years ago, while helping a friend with a passion project on thermoelectric materials, this compound came up in the context of making certain reactions possible at higher temperatures. It doesn’t sound glamorous, but for researchers pushing boundaries in green energy, these little details make or break progress.
Potential Roadblocks and Honest Questions
No solution fits everywhere. LiTFSI isn’t cheap. Large-scale adoption faces price hurdles. Without economies of scale, prices may hold back new tech for years. I’ve listened to startup founders grumble about the cost of everything with “trifluoro” in the name, including this salt. Environmental questions also linger. Every battery salt leaves its mark, and we need more research into production impacts and recycling.
Engineers need to keep hunting for salt solutions that balance safety, cost, and sustainability. Policymakers and companies can help by supporting research and nudging the market toward safer, longer-lasting energy storage. Consumers should stay aware of what goes into their batteries. Without public interest and demand, progress slows to a crawl.
LiTFSI reminds us that even the most tongue-twisting chemicals can change how we power our lives. Innovation often happens at the edges, among compounds hardly anyone has heard of. The best work happens by putting safety, practicality, and lasting power ahead of convenience—something we could all use a little more of.
Looking Closer at a Popular Electrolyte Salt
Lithium Bis(Trifluoromethanesulfonyl)Imide, known as LiTFSI to most chemists and battery engineers, holds an important spot in rechargeable battery technology. The chemical formula is LiC2F6NO4S2. The structure, at first glance, can seem a mouthful: two trifluoromethanesulfonyl (CF3SO2) groups bonded to a central nitrogen atom carrying a negative charge, paired with a lithium cation. This arrangement delivers stability and efficiency in the right conditions, which is exactly what’s needed from a battery’s lifeblood.
Beyond the Formula: Why LiTFSI Stands Out
Years spent tinkering with lithium-ion cells and reading research papers have shown me how the tiniest shifts in salt composition affect the big picture of power and safety. For a long time, Lithium Hexafluorophosphate (LiPF6) led the way in lithium battery electrolytes. Then came the issue: LiPF6 breaks down under heat or moisture, forming toxic byproducts like hydrofluoric acid. Here’s where LiTFSI steps up. This salt boasts impressive thermal and hydrolytic stability. In simple terms, it doesn’t fall apart or throw a fit when humidity or temperature climbs.
The molecular structure matters here. Each trifluoromethanesulfonyl group keeps the negative charge well-hidden from water and other would-be attackers. That gives the whole molecule a toughness missing in earlier contenders. This directly translates into longer-lasting batteries and a lower risk of catastrophic failure, which matters when these cells power cars, computers, or your phone right next to your body.
Economic and Environmental Trade-Offs
Stability and safety sound great, but there’s always a catch. Large-scale production of LiTFSI costs more than old-school alternatives. Plus, while it performs better under stress, its high fluorine content brings environmental headaches at end-of-life. Fluorinated chemicals, once released, stick around. Waste management and recycling systems haven’t yet caught up to this new breed of battery chemicals. This creates a challenge for the industry and everyone who cares about what happens to yesterday’s gadget.
On the upside, academic labs and companies have started to tackle these gaps. Early-stage research points to better recycling pathways and ways to recover lithium and other elements from spent batteries. Investment here will pay off, once the volume of discarded LiTFSI-based cells matches their rising popularity in energy storage.
Rethinking Battery Chemistry
Electrolyte salts help define the capabilities of a battery. LiTFSI’s reliability and heat resistance give engineers space to push for faster charging and longer lifespans. Consumers won’t need to know the detailed structure—a lithium cation paired with a robust nitrogensulfonyl anion—but the benefits ripple out. If batteries charge safely at higher voltages, portable electronics shrink. If electric cars last longer, fewer batteries end up in landfills. LiTFSI represents a step in the right direction, even as its own challenges spur a call for smarter waste policy and greener chemistry.
Balancing Safety and Progress in Modern Chemistry
Folks who dabble in battery chemistry or advanced materials know Lithium Bis(Trifluoromethanesulfonyl)Imide—often called LiTFSI—has carved a space as a top-tier lithium salt. Its reputation comes from its chemical stability and solubility, giving lithium-ion batteries and other electrochemical cells their long shelf lives and solid performance. But with every powerful ingredient, there’s a flip side: proper handling and storage matter just as much as performance numbers on paper.
Understanding Why Care Matters
Factories, research labs, and even seasoned chemists have to remember that LiTFSI comes with a few risks baked in. The stuff can irritate skin, eyes, and respiratory systems. If you’ve spent any time in a laboratory, the warnings for lithium salts tend to stick in your mind. My own run-ins with lithium compounds taught me pretty quickly that careless moments lead to rashes, coughs, and some explaining to the lab supervisor. Gloves, goggles, and a sturdy lab coat keep mishaps small and manageable—simple steps, but they go a long way.
I find it’s easy to gloss over the need for a clean, dry workspace—until someone leaves a chemical bottle unsealed and the powder clumps or, worse, starts reacting with moisture. Lithium salts can pull water from the air, and LiTFSI is no exception. Humidity can wreck the quality of a whole batch, leading to wasted money and frustrated teams. Studies show that water contamination degrades performance for batteries and can even produce dangerous byproducts. Airtight bottles with desiccant packs, stored on a stable shelf away from direct heat, do more than protect a product—they protect people’s work and well-being.
Controlling the Environment
LiTFSI likes cold and dry conditions. From experience, the best labs use climate control for sensitive supplies. Cooling to room temperature or lower dries out the risk of decomposition. Sunlight streaming through a window can cause the compound to break down, so a dark cupboard beats out a bright bench every time. It’s not high-tech, just thoughtful.
Spill stories float around any lab: a careless scoop, a bump of the wrist, some powder drifting into the air. Local exhaust—hoods or even simple fans—catch airborne dust and keep it out of the air where people breathe. Cleaning up right after a spill, without letting the area sit exposed, makes a difference for the next person who walks by. Good habits add up.
Fact-Driven Solutions
Clear labeling tops my list of must-haves, along with regular training. If half the folks in a workspace pick up a bottle without checking what’s inside, mistakes happen. Digital tracking for chemical stocks, with reminders about storage and expiry, helps avoid surprises.
Regulatory guidelines from agencies like OSHA and the European Chemicals Agency lay out basic requirements for chemical safety. Their resources don’t just come from thin air—they’re based on years of workplace incidents and near-misses. By following these established signals, companies send a message: lab safety improves results, workplace trust, and keeps folks out of the hospital.
Looking Ahead
Lithium bis(trifluoromethanesulfonyl)imide is a building block for new battery tech and countless innovation stories. The simple routines of keeping things dry, labeling, and working with respect for risk may not make headlines, but they set the stage for real progress. Safe handling and storage aren’t only legal hoop-jumping—they keep projects on track and give research the steady ground it needs to thrive.
Understanding the Boom Around Lithium Salts
Every few months, headlines pop up about lithium-ion batteries powering everything from cars to smartphones. Lithium salts sit at the core of these batteries, and one that’s getting attention lately is Lithium Bis(Trifluoromethanesulfonyl)Imide, or LiTFSI. In labs and industry, LiTFSI has taken off because it helps batteries last longer and charge faster. That sounds great on paper, but stories about chemical hazards always prompt the question: is it safe?
What Makes LiTFSI Useful—and What Raises Eyebrows
LiTFSI has a mouthful of a name, but its role is pretty simple. It functions as an electrolyte salt. Scientists like it because it dissolves well in battery solvents, which means you get better power output and less likelihood of a short circuit. Researchers have praised LiTFSI for its stability at high temperatures and for making lithium batteries perform better in tough conditions.
Yet, anyone who’s worked in a lab knows that better battery chemistry doesn’t automatically go hand-in-hand with safety. LiTFSI contains fluorine and sulfur, and both give off nasty byproducts if they end up in the wrong place—like in your lungs, water supply, or on skin.
The Hazards: Not Just a Lab Issue
Let’s get concrete: LiTFSI by itself isn’t explosive, and it doesn’t burn easily. Drop it into an open flame, you won’t see Hollywood explosions. On the other hand, mix it with water and you can get corrosive, acidic fumes. During my graduate research years, a lab mate spilled a small sample into a sink. The sharp, unpleasant smell that filled the room told us right away that accidental releases, even minor, shouldn’t happen.
Wearing gloves and goggles helps, but disposal brings its own set of headaches. Water treatment plants aren’t set up to handle chemicals like this. If LiTFSI runs off into rivers, both the lithium and the fluorinated parts can stress aquatic life. Over time, those perfluorinated compounds build up and don’t break down in nature, and scientists have linked many of these “forever chemicals” to health problems in wildlife and humans alike.
Symptoms like eye and skin irritation pop up first. Industrial exposure can also affect breathing. The danger raises its head not just during big accidents, but through the small, chronic exposures of factory work or improper storage and transport.
Why Transparency and Oversight Matter
Companies love touting their battery innovations, but honest talk about chemical safety often sits in the fine print. Workers and consumers should know exactly what’s in the batteries that power their daily lives. Regulatory bodies such as the Environmental Protection Agency and similar groups overseas place restrictions on handling and disposal, but gaps show up as production ramps up. One major battery plant in Asia faced fines not long ago for improper waste handling—evidence that regulations don’t always catch issues in real time.
Safer Paths Forward
The solution isn’t to panic and halt research. Instead, focus should land on containment, clear labeling, and enforcing responsible disposal. Closed-system production lines, personal protective equipment, and real consequences for companies that sidestep safety rules can help. On a bigger scale, investment in alternative salts with fewer environmental dangers and more data sharing among battery makers make sense. Labs need funding for long-term toxicological studies, not just patent races for better battery life.
For those of us who see the promise of greener tech: the gadgets and vehicles powered by modern batteries shouldn’t leave invisible marks on workers or ecosystems. Being honest about hazards leads to smarter use, and that’s the only way this technology will truly serve us all.
Understanding Purity for Real-World Applications
In labs or at production scale, people expect their chemicals to be reliable. With lithium bis(trifluoromethanesulfonyl)imide—often called LiTFSI—the need for consistency feels especially strong. Battery makers and electrochemical researchers know this salt by reputation because every impurity can throw off the delicate balance inside lithium-ion cells. Most reputable suppliers offer LiTFSI at purities of 99.9% or higher. There's a reason for that. Trace water or metallic contaminants degrade battery performance, leading to lower capacity, faster degradation, or dangerous situations like short circuits. Real-world chemists invest effort in tracking these numbers, not to show off, but to trust their measurements tomorrow as much as today.
Industry standards keep people honest. Quality gets verified with methods like Karl Fischer titration to check for moisture or ICP-MS for metals. Field experts will sometimes push for “battery grade” material—this means rigorous limits on impurities, not just a high number on the label. Anything below about 99.9% purity tends to introduce unpredictability in high-end applications. The more demanding the field, the more scrutiny folks give to every decimal point.
Packaging: It’s Not Just About the Size
Bulk buyers and researchers look at more than price per kilogram. With LiTFSI’s mild hygroscopic nature, packages don’t just hold the salt—they protect it from the air. Manufacturers and suppliers usually offer a range: you can find this salt in glass bottles as small as 5 or 10 grams, ideal for research setups where waste is costly and shelf-life matters. Go up a notch, small drums or plastic containers in 100-gram or 500-gram volumes become available. These often come vacuum-sealed or in foil-lined bags. The biggest buyers, like battery plants or electrolyte formulators, order by the kilogram—sometimes in double-lined drums to minimize air and moisture contact over longer storage. I’ve seen smaller labs split up big batches with a dry box, just to save on costs without losing quality to humidity.
Why Detail Matters
It’s tempting to overlook the little things, but the packaging can make or break a project. Even the purest LiTFSI will take on water in a humid room, and that’s enough to scrap a sensitive test or slow down a production line. Lab veterans know to open supplies under dry atmosphere or inside glove boxes. There’s no point in paying for high-purity material just to let it degrade on the shelf. It helps to share this advice with colleagues, especially newcomers who might not have tripped over this the hard way.
Pushing for Better Quality and Less Waste
The world wants more high-quality batteries, so demand for clean, reliable LiTFSI isn’t shrinking anytime soon. Experience shows the push for better packaging and stricter impurity controls always comes from both ends—the scientist frustrated by unexpected cell failure, and the supplier who doesn’t want complaints or wasted returns. Some folks in the field push suppliers to offer clear impurity breakdowns and tighter moisture controls, not just vague claims. Smart labs use up what they buy quickly, share bulk shipments between departments, or ask for custom packaging to avoid leftovers degrading on the shelf.
Trusting your chemicals comes down to both purity and how they arrive at your door. In battery research and production, that trust matters as much as the voltages and currents themselves.