Lithium Trifluoromethanesulfonate: Where Energy Storage and Chemistry Meet Daily Life
Tracing the Story: How Lithium Triflate Became a Go-To Salt for Science and Industry
Lithium trifluoromethanesulfonate—most folks know it as lithium triflate—never started with much fanfare. Its history weaves right through the steady march of lithium research, especially after the global push for more reliable and safer battery technology in the late twentieth century. Chemists looking to boost conductivity in electrolytes kept running into the same roadblocks with older salts: moisture sensitivity, poor solubility, and unpredictable performance when the weather or temperature changed. Lithium triflate broke through that rut thanks to its strong stability in a world that seemed full of wobbly options. It planted itself early in solid-state applications because it does not break apart even if you sit it under humid air for a spell. This made it a favorite not just in research labs but on the factory floor, from Japan to the US, as energy storage ramped up throughout the 2000s.
Looking Closely: What Makes Lithium Triflate Worthy of Attention?
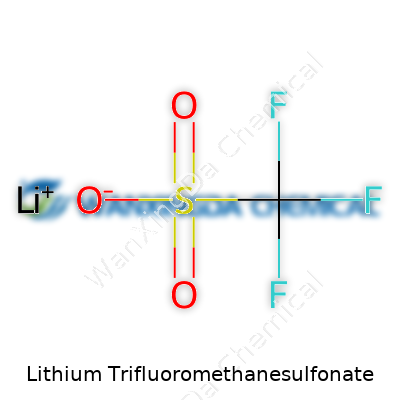
Open a jar of this salt, and you find a white, crystalline powder—nothing fancy to the naked eye. This simplicity hides a bit of chemistry magic. Its formula, LiCF3SO3, spells out why it gets so much attention. That trifluoromethanesulfonate group clings tightly to its lithium ion, but not too tightly. This helps lithium leap around easily in a solution, which anyone designing batteries or electrochemical devices needs. You can stir it into polar solvents or water, you’ll watch it vanish without a fuss. It loves both organic and inorganic partners. The wide electrochemical window—so it doesn’t break down at high voltages—gives it an edge in devices being asked to do more.
Preparation: Getting the Salt Right
The laboratory prep isn’t full of surprises for folks who know their way around strong acids and lithium basics. Most routes start with triflic acid. Neutralization with lithium carbonate or lithium hydroxide completes the trick. Careful workers keep it dry and pure—the acid’s strength means any leftover moisture or impurity gets in the way. Large production follows the same logic, but with cleaner lines and better air controls. There’s no fancy trickery, just patience, good ventilation, and steady handing of caustic agents.
Names and Labels: What Else Do We Call It?
Lithium triflate crops up under a few names depending which catalog you glance at. Some call it trifluoromethanesulfonic acid lithium salt, others prefer lithium trifluoromethanesulfonate. The acronym LiOTf pops up in scientific papers, but folks rarely use it in casual conversation. The CAS number keeps institutions on the same page, but for working chemists, it’s just “lithium triflate”—no fancy branding, just a straightforward identity.
Getting Down to Use: How Lithium Triflate Builds and Powers Devices
This salt earns its keep in solid polymer electrolyte membranes, especially for lithium batteries fighting to keep up with the ever-growing power needs of modern gadgets. Its high ionic conductivity and reluctance to absorb water help battery components last longer—even in humid climates that used to cut lifespan sharply. It has found friends in the world of supercapacitors as well, powering research on energy storage that goes beyond lithium-ion. No one likes a battery that leaks or degrades after a dozen charges; lithium triflate’s toughness on this front stands out. It also shows up in organic synthesis as a key catalyst and supporting electrolyte—a small role that keeps broader chemical manufacturing humming. Compared to competitors, this salt shrugs off the conditions that send lesser chemicals back to the drawing board.
Physical and Chemical Realities: Handling and Storing with Care
White, odorless powder hides a tough customer. Lithium triflate doesn’t mind heating to moderate temperatures. With strong solubility in polar solvents, it deals well in nonaqueous battery mixes. Under bright lights or a bit of humidity, it doesn’t break down or spoil quickly. This property saves a lot of effort in environmental controls, bringing costs down for producers and users alike. Stable over long periods, it avoids the seasonal swings that cause headaches further down the supply chain.
Reactions and Modifications: Building Blocks for Bigger Chemistry Projects
Chemists keep lithium triflate close when they want to swap ions with cationic exchange or modify a battery’s operating window. Its ability to keep lithium moving without clogging up the works opens new pathways for both energy and organic synthesis research. In the lab, using it as an electrolyte—rather than bulk reactant—means improved yields and cleaner reactions, stripping out the need for extra purification steps. Its compatibility with a range of polymer matrices sets it up for future-proofing materials that store or deliver energy for longer cycles.
Playing by the Rules: Watching Out for Safety and Health
Lithium triflate performs well, but you don’t want it mixed up with your food or shallow breathing space. Workers need gloves and eye protection just like with any caustic lab salt. It doesn’t catch fire easily, nor does it give off noxious fumes unless handled carelessly. If spilled, a sweep and good rinse will do, but inhaling a fine powder over time risks irritation. Anyone running a big operation sticks to the usual occupational safety rules—no shortcuts, no exceptions—even if the chemical itself feels less dramatic than some alternatives. Storage needs a dry spot, ideally in sealed containers because water can still cause trouble if it builds up over the years.
Pushing the Envelope: Current Research and Where Things Might Go
The energy world is always on the hunt for safer, longer-lasting storage with less environmental baggage. Researchers keep circling back to lithium triflate when standard electrolytes or additives disappoint. Current focus sits on solid-state batteries aiming for both higher capacity and greater resistance to fire risks. The triflate group’s unique chemical grip helps block dendrite growth, which has plagued lithium tech for decades. There’s healthy debate over whether its price, tied to triflic acid production, will hold back wider usage, but new synthetic pathways may ease the pressure. Folks working on fluorinated battery chemistries have flagged it for safer, more recyclable alternatives to PF6- or ClO4 salts. Studies continue in biotech, as researchers see if its stability and low toxicity in certain conditions give it a life beyond just energy. Research dollars keep flowing into alternative synthesis, environmental impact, and end-of-life recycling so this promising salt doesn’t create headaches down the road.
Toxicity and Environmental Perspectives: A Cautious Approach
This isn’t table salt, but it doesn’t carry the heavy baggage of other lithium electrolytes. Short-term exposure data mostly show minimal acute toxicity, which reassures both workers and regulatory committees. Waterways still don’t welcome large doses—fish haven’t signed up for lithium supplements. Efforts to measure bioaccumulation and long-term soil impacts stay underway, especially as batteries see broader end-use and potential landfill leakage grows. Environmentalists, regulators, and companies all demand solid end-of-life plans, not just fire-and-forget disposal. Producers listen: improved reclamation and reprocessing methods are starting to close the loop, keeping lithium triflate safer for the next generation.
Looking to Tomorrow: The Near and Distant Future for Lithium Triflate
Anyone watching battery technology knows better electrolytes can make or break a product line. Lithium triflate stands out as stable, tough, and flexible under real-world conditions. It already powers much of the research on safer, nonflammable solid electrolytes and is considered a strong candidate for replacing older, riskier salts in the next wave of devices. Recyclability and raw material sourcing stay as open questions, but the chemical’s record gives hope that advances will catch up with commercial scaling. What matters is getting smarter not just in using it, but managing it over its whole lifetime, from synthesis to final recycling. The technical journey of lithium triflate traces our needs for both higher performance and responsibility in chemistry—a combination future generations will appreciate as energy becomes even more central to daily life.
People outside laboratories rarely come across the name lithium trifluoromethanesulfonate. Yet this quirky-sounding compound keeps turning up behind the scenes in tech I use every day. Phones that stay charged longer, cars that travel farther on a single battery—all owe some thanks to specialty chemicals like this one. Learning how it fits into the world of batteries and broader technology sheds light on why some innovations take off, while others stall on the drawing board.
More Than Just a Mouthful
The compound handles a specific job inside lithium-ion batteries. Chemical engineers pick salts to help lithium ions move easily between electrodes. In the case of lithium trifluoromethanesulfonate—the name packs a punch, so scientists call it LiOTf—it’s all about providing a stable, highly conductive environment. You’d think, as a cyclist might, that the path has to stay clear and smooth for peak flow. Even a tiny hiccup or clump in the chemical landscape can drop overall battery performance. LiOTf supports this flow better than some classic options, resisting moisture and heat, which often spell trouble for battery reliability.
Boosting Safety and Lifespan
Most folks hear about high-profile battery fires: a cell phone overheating or an electric vehicle sidelined by safety concerns. Lithium trifluoromethanesulfonate fights back on that front, too. Its molecular makeup helps slim down the risk of batteries breaking down under tough conditions, generating fewer dendrites—the needle-like intruders that can short-circuit rechargeable batteries. More resilient batteries mean fewer recalls and less waste. These improvements can translate straight to my daily routines: fewer replacement batteries, less e-waste, and products that last longer.
Beyond Batteries: Chemistry at Work
LiOTf shows up in other pockets of science, sometimes acting as a catalyst, sometimes as a supporting player in organic synthesis. For those of us fascinated by pharmaceuticals or specialty plastics, these roles count. Some drugs only become practical when chemists have access to the right salt at the right time. LiOTf’s ability to handle water without falling apart widens the horizons for molecule-building in labs. Snappy breakthroughs in drug discovery sometimes trace back to humble contributors like this one.
Weighing the Trade-Offs
No story about specialty chemicals sails along without hitting the rocks of cost and supply. LiOTf isn’t cheap. Striking the balance between better performance and affordability pushes research further. Manufacturers constantly weigh the improved stability and conductivity against price tags. Environmental scientists, on the other hand, scrutinize what happens to the compound as batteries get tossed out or recycled. Regulations call for transparency and safety, so every step forward gets measured on more than one scale.
What Comes Next?
I keep an eye on emerging chemistries for batteries, always curious to see which salt will drive the next leap in storage or safety. As devices shrink and vehicle demands grow, lab teams look for even more stable, less expensive chemicals. Continued research into LiOTf’s life cycle and environmental impact stands as a priority. The more data scientists gather and share, the easier it gets to make choices rooted in both innovation and responsibility. In the meantime, this quietly powerful compound keeps doing its job, one battery at a time.
A Closer Look at LiCF3SO3
People don't talk about battery chemistry at the dinner table, but it's part of everyday life. Lithium trifluoromethanesulfonate carries the chemical formula LiCF3SO3. Folks in laboratories and factories use this compound for a reason: it helps move ions inside high-performance batteries — and that's a big deal for the world’s technology.
I saw how important this compound became during the rise of electric cars and high-capacity power banks. Lithium-ion technology doesn't just pop out of nowhere. Chemists chase better salts for safer, long-lasting batteries. That formula, LiCF3SO3, points to one lithium ion and the anion called triflate (CF3SO3-). This salt dissolves in organic solvents better than older salts like lithium perchlorate. Strong solubility means engineers can get better ion transfer, which matters for energy storage.
Why This Matters Outside the Lab
I’ve seen what happens when battery makers take shortcuts with materials. You end up with gadgets that overheat or die quickly. Stability and purity aren’t just checklist items for these chemical salts. The exact makeup — in this case, LiCF3SO3 — determines safety and long-term use. The trifluoromethanesulfonate part gives it good chemical stability and makes it less likely to form dangerous byproducts. That’s not just textbook knowledge. Fire departments and recycling specialists would agree, since incidents from battery failure clog up their schedules every month.
Environmental impact also comes into play. Lithium perchlorate, for instance, has historical baggage due to its potential to cause explosions under certain circumstances. Not all lithium salts share the same risk profile. Trifluoromethanesulfonate salts are less reactive; the formula means fewer environmental drawbacks and lower regulatory headaches for companies that want to go green.
Improvement and Obstacles
Folks in industry push for cheaper, cleaner, and safer materials. Cost sits at the center of every decision made by battery manufacturers. Lithium triflate’s unique balance — solubility, conductivity, and stability — gives it a special spot, but the production price slows its wider adoption. If you’ve ever opened up a device and seen a swollen battery, you’ve seen the results of cheap production. Engineers push for new ways to make lithium triflate that shrink costs and cut toxic byproducts. Streamlining synthesis and researching more recyclable processes could push this salt beyond niche markets.
Some researchers dig into alternatives, but they often end up circling back to this tried-and-true formula. Field tests and academic studies back this up. Its presence in peer-reviewed research speaks volumes. Not every battery salt gets nods from both industry and academia; lithium triflate does.
Looking Forward
People want smaller, lighter tech. Electric cars need batteries that charge faster and last through years of daily traffic. Careful chemistry, rooted in simple but powerful formulas like LiCF3SO3, puts those goals within reach. Applying experience from the lab to the factory floor will shape the next generation of energy devices. That’s why knowing the formula — and understanding what it brings to the table — adds value far beyond the chemistry classroom.
Why the Stuff Shows Up in Labs
Ask a working chemist about lithium trifluoromethanesulfonate, and you’ll find it’s a regular on the shelf, especially in batteries, organic syntheses, or as an electrolyte. I’ve crossed paths with it mostly in electrochemistry and have had a fair share of moments where the label alone made me pause. Its role often deals with moving ions smoothly in solutions where water just complicates things. Despite its usefulness, I've never felt like tossing caution to the wind around anything lithium-related.
Taking Safety Seriously – Gloves On, Goggles Down
Say you open a jar of lithium triflate. The powder looks harmless, almost boring. That can be deceiving. Every Material Safety Data Sheet flags it as an irritant. It doesn’t explode at a glance or scorch like concentrated acids, but touching it or breathing in dust finds trouble. I know people who picked up headaches or coughs working too casually around lithium salts, and skin can dry out or get red in no time. Even small exposures like forgetting a dust mask during a quick weighing can lead to a tickling cough that lingers through the day. Under UV light, it glows faintly, a reminder of its charged personality.
Lab safety is about habits, not heroics. Every time I suit up, it’s because complacency has no upside when chemicals enter airways or eyes. Googles, gloves, and fume hood work aren’t for show. Long sleeves, closed shoes – the basics – become second nature the day a splash lands. Lithium compounds, even when not explosive, don’t mix well with water on skin or in eyes. Rinsing promptly with running water does the trick, but prevention just saves the hassle.
Waste Isn’t Just Garbage
I’ve seen folks toss lithium residue in the wrong bin – an ugly surprise for waste handlers or anyone down the line. General landfill disposal risks leaching, especially in places where groundwater isn’t well protected. Instead, neutral containers marked for hazardous waste keep municipalities and groundwater safer. Lip service to "green chemistry" means nothing unless chemical waste gets managed at every stop.
The Risk Hides in Small Details
In dry form, it won’t jump out and attack your lungs, but grinding, weighing, or pouring raises dust. Accidental spills never end up where you want them. If you mix lithium triflate with certain strong oxidizers, a reaction can start. I once watched an undergrad slip some into the wrong flask – the fizzing didn’t hurt anyone, but it scared them into double-checking afterward. Reading up beforehand, even after years in the lab, saves dollars and health both.
Smarter Habits, Fewer Regrets
Common sense, plus a bit of preparation, makes handling lithium triflate almost routine. Using sealed dispensers, storing in cool and dry spots, and never trusting expired gloves – these habits keep work incident-free. Good ventilation and an organized workspace – not just clean, but clutter-free – also offer a quiet kind of protection. Building a culture of safety where people look out for each other means near-misses won’t turn into accidents that haunt a career.
The bottom line: lithium trifluoromethanesulfonate won’t fry your hand at first touch, but the risks aren’t imaginary. Science pushes forward with both innovation and responsibility. Treat it with respect and nobody gets burned – that’s a standard worth carrying past the lab, into any workplace where chemistry makes things happen.
Understanding the Real Impact of Storage Choices
Storing chemicals rarely makes headlines, but anyone who's spent time in a university lab or an industrial battery startup knows storage decisions can have outsized consequences. Lithium trifluoromethanesulfonate—often called lithium triflate—seems unremarkable at first: a white powder, not especially volatile, not known for catching on fire by itself. Yet, the storage slip-ups I've seen, or have heard in postmortem stories from researchers, offer a firsthand look at how small mistakes with materials like this shape safety, data reliability, and project costs.
Humidity’s Sneaky Threat
Lithium triflate tends to pull in water from the air. It's called being hygroscopic. I’ve worked with enough hygroscopic chemicals to know how quickly an open jar changes: one moment, clean and free-flowing; a month later, you get clumps, a sticky mess, and measurements that veer off course. Water sneaks into sensitive lithium projects, quietly interfering with conductivity and electrochemical tests. Lab managers repeat the advice, and for good reason: keep lithium triflate in tightly sealed bottles, protected from air, and watch humidity levels like a hawk. A good desiccator cabinet filled with fresh desiccant packs helps, especially during sweltering summer days or rainy seasons when labs get muggy. When the humidity in the bottle spikes with just a few careless minutes exposed to open air, it’s tough to undo the damage.
Room Temperature Is Not Always "Room Temperature"
It’s tempting to toss everything onto an open shelf, call it “room temp,” and move on. From my experience, that’s an invitation for trouble. Heating or freezing extremes don’t hurt lithium triflate the way they harm unstable compounds, but temperature swings encourage condensation and slow, unwanted reactions—especially in shared storage spaces. Consistency counts more than aiming for some magic number. Most guidance points to typical room temperatures, but real practice often means carving out a stable, dry zone, far from the vibration and temperature near windows, radiators, or air conditioners. Store the bottle where staff have clear rules about chemical segregation, so spills or cross-contamination stand less chance of happening.
Safety Isn’t Just a Checklist
Mixed storage, accidental spills, and absent-minded placements pop up all the time. Some places throw lithium triflate with oxidizers or even food containers by mistake, especially in crowded shared labs. I’ve seen graduate students turn red when they get called out for storing sensitive lithium salts next to open acid jars. Not separating incompatible chemicals sets the stage for accidents—sudden chemical reactions, toxic fumes, and even injuries. Anyone serious about safety builds habits: double-checking labels, using secondary containment (like plastic trays), and making sure spill kits are close at hand. Fire hazards with lithium triflate stay pretty low, but dust and fine powders can get into airways if the bottle’s tossed around, so plenty of folks use gloves and masks even for quick weigh-outs.
Real Solutions for Tidy, Reliable Workspaces
Better training for lab staff gets results. I’ve seen teams cut storage mistakes by setting up shared guidelines and running drills on what to do after a chemical spill or an accidental humidity spike. Regular audits—some old-fashioned clipboard checks—keep people from getting lazy over time. Investing in labeled, airtight storage, plus easy access to desiccators where needed, pays off for any lab working with lithium salts. Skip on the basics, and frustrations—ruined experiments, hidden hazards, even costly replacements—stack up fast.
A few small changes in storage can decide whether research goes smoothly or falls apart. That’s a lesson every chemist learns quickly, often the hard way.
Try searching for lithium trifluoromethanesulfonate in the daily news headlines. It doesn’t scream for attention the way precious metals or rare earths do, but its impact quietly drives some of the most important technology around us. Most folks probably never think about it, even though parts of our modern world would fall flat without this mouthful of a chemical.
Behind Every Battery Story
I remember testing lengthy lists of battery specs in a small electronics lab back in college. We always looked for batteries that ran a little longer, recharged more efficiently, stayed cool under stress. Electrolytes, which let lithium ions shuttle between electrodes, turned out to be the secret ingredient. Lithium trifluoromethanesulfonate, or LiOTf, keeps popping up as a trusted salt in these solutions.
The EV revolution and the surge in powerful smartphones need batteries that last, don’t flame up, and deliver every promised milliamp. Legacy solutions relied heavily on lithium hexafluorophosphate. Chemists started mixing in lithium triflate for its chemical stability, seeing fewer toxic byproducts, especially when batteries are pushed to their limits. The story doesn’t end with consumer gadgets; grid-scale battery storage systems stand to benefit as well. With more communities worrying about energy resilience and climate change, safer, more reliable electrolytes become a big deal.
Polymers: The World Gets Flexible
You’d think plastics are all the same, but try comparing the wiring in a basic lamp to flexible displays or next-generation dental devices. Industries like electronics and medical tech need polymers with specific conductivity or resilience. Lithium trifluoromethanesulfonate finds its way into polymer electrolytes, where its high ionic conductivity opens creative doorways. By adding this salt, labs achieve plastics that not only conduct ions but also stay flexible or withstand heat. These tweaks show up in wearables, flexible solar cells, even certain drug delivery patches.
Chemical Synthesis: Small Salt, Big Job
Not all transformations in chemistry require heavy machinery or exotic metals. Sometimes, a simple salt turns a stalled reaction into a breakthrough. Organic chemists often reach for lithium triflate in their labs. It acts as a catalyst or stabilizer during tricky reactions, like forming carbon-carbon bonds or rearranging molecules just so. These reactions eventually bring us new medicines, performance materials, and sometimes surprising household products.
The Quiet Reality of Supply and Cost
Lab chemicals rarely get as much scrutiny as rare earth elements or lithium for use in batteries, but scaling up brings familiar headaches. Mining and refining are never risk-free ventures, especially as demand for advanced batteries and high-performance plastics surges. The quest to recycle electronics and batteries, or to design greener manufacturing, keeps finding these “fine chemicals” in the spotlight.
Facing the Challenge: Next Steps
Research teams need to work closely with industry to find ways to recycle or reuse lithium-based salts. Developing methods to recover chemicals like LiOTf from spent batteries, expired devices, or even chemical reactors could make a dent in both cost and environmental impact. Public awareness also matters; if engineers and policy-makers stay tuned to such details, smarter regulations and incentives could nudge the market toward responsible sourcing and recovery. As in so many cases, the invisible backbone of technology deserves a little more attention from all of us.