Magnesium Trifluoromethanesulfonate: Looking Beyond the Chemistry
Historical Development
It’s hard to ignore the quiet rise of magnesium trifluoromethanesulfonate, a compound that didn’t get its claim to fame right away. Chemists have driven curiosity for decades about harnessing salts with tailored properties, aiming to move beyond the often-used lithium or sodium varieties. The discovery and continued study of this magnesium salt isn’t a fluke; it reflects a consistent push among researchers to meet demands for higher performance — not just in labs, but in devices you find in real life, including batteries and catalysts. Sitting in the shadow of better-known salts, magnesium triflate gained traction in the late 20th century as folks realized its properties could bridge important gaps in existing technology and research.
Overview of the Compound
You get magnesium trifluoromethanesulfonate by pairing magnesium ions with triflate anions. At a glance, it shows off a solid, white, crystalline appearance — a look it shares with many purified inorganic salts. Don’t let this simple facade fool you. Each grain packs a punch: high solubility in polar solvents, non-coordinating behavior in solution, and thermal stability that attracts material scientists and synthetic chemists. Where earlier generations of salts ran into trouble with reactivity or contamination, this one sidesteps many headaches, opening doors to innovation in electrochemistry, organic synthesis, and industrial processing.
Physical and Chemical Standouts
Magnesium triflate displays an ability to dissolve in polar organic solvents such as acetonitrile, DMSO, and methanol, outclassing some traditional magnesium compounds. It offers a melting point well above room temperature, so you won’t find it melting on a warm day in the lab. The triflate anion’s weakly coordinating nature turns this compound into a go-to option for reactions needing a stable, non-interfering counterion. From my experience, you realize its stability under typical laboratory conditions means fewer storage worries — neither humidity nor moderate temperature swings cause notable degradation, and it doesn’t build up static charge or pose spontaneous hazard under normal use.
Technical Specifications and Labeling
Anyone ordering magnesium triflate comes across specifications that matter: batch purity, water content, and precise stoichiometry. Chemists focus on these details because stray ions or moisture can tank a whole experiment. I’ve seen how labeling transparency from suppliers — things like the precise hydration state or crystal structure — saves hours or days, especially in scale-up for industrial runs or academic research. Firms tend to specify for high-performance applications: water content below 1%, micron-level purity, and clear CAS identification, which lines up with regulatory requirements for traceability and safe handling.
Preparation and Synthesis
Synthesizing magnesium trifluoromethanesulfonate commonly takes the route of reacting magnesium carbonate or magnesium oxide with trifluoromethanesulfonic acid, sometimes following a careful neutralization step. The chemistry isn’t particularly exotic, but ensuring purity takes some vigilance, especially on the drying and filtering stage to strip away water or byproducts. Processes often include stepwise crystallization and solvent exchange, since the balance between yield and purity comes front and center if you’re prepping several grams for battery electrolyte research or catalytic screening. A seasoned hand can push this reaction toward high yield without generating messy side products or contaminating the product with magnesium hydroxide or leftover reagents.
Chemical Reactions and Possible Modifications
Magnesium triflate stands out in catalytic cycles. Its ion-pairing characteristics and Lewis acidity allow it to activate organic substrates, particularly in Friedel–Crafts alkylations and acylations. Synthetic chemists put this to work, using it as a gentle activator that doesn’t push side reactions the way stronger acids or alternative salts do. Its solubility and non-coordinating anion open the door for direct use in reactions where other magnesium sources would simply precipitate or lose reactivity. Chemistry departments worldwide test customized magnesium-based complexes made with modified triflate ligands, targeting new selectivity profiles or better compatibility with green chemistry protocols.
Synonyms and Naming Conventions
The naming isn’t the most intuitive, which trips up folks new to the scene. On paper, it goes by several names: magnesium triflate, magnesium trifluoromethylsulfonate, or, for those into abbreviations, Mg(OTf)2. In patent literature, “magnesium bis(trifluoromethanesulfonyl)” sometimes appears, which can create confusion if you’re cross-referencing procedures or comparing suppliers.
Safety and Operational Realities
Most lab and industrial settings see magnesium triflate as moderately safe, without the pyrophoric or highly toxic profile found in some metallic organics. Folks still use gloves, protect eyes, and manage dust, since all fine powders pose inhalation risks and can trigger skin irritation with prolonged contact. There's no strong evidence of acute toxicity in low doses, but that doesn’t excuse careless handling. Labs working around sensitive electronics or in scale-up fill out the required risk assessments, mainly for splashes, spills into drains, or chronic exposure that isn’t entirely studied.
Applications Stretch Across Many Fields
You spot magnesium triflate in organometallic synthesis, but its reach extends further. Battery researchers have tested it as a magnesium electrolyte salt, hunting for safer alternatives to lithium-ion cells. Some of the earliest promising tests in non-aqueous rechargeable magnesium batteries leaned on magnesium triflate for reliable ion transport and suppressed side reactions. Catalysis remains another playground, with organic chemists reaching for it in Diels–Alder synthesis or selective protection–deprotection sequences. Industrial processes, especially those relying on fine chemical synthesis, benefit from its predictable behavior and the lack of byproduct generation compared to chloride or acetate salts. Surprising to some, a smattering of pharmaceutical research includes the triflate as a counterion to help drug candidates remain stable in solution during screening.
Ongoing Research and Development
Ask around in electrochemical R&D circles and you’ll hear magnesium triflate mentioned in projects seeking cheaper, safer, and longer-lasting batteries. European and East Asian research teams have reported steady improvements in performance using this salt over the last decade. On the organic synthesis front, catalytic profiles have improved through better control of solubility and phase transfer, translating to higher yields and cleaner post-reaction separation. Academic literature reflects a trend: magnesium triflate earns respect as an underdog, forming the scaffold for explorations of new synthetic pathways or next-generation material processing.
Toxicity Research
So far, tested doses of magnesium trifluoromethanesulfonate show low toxicity in classic animal models, but regulatory reviews stress gaps in chronic exposure and environmental persistence. Given the compound’s high stability, breakdown in wastewater or soil doesn’t happen quickly, which nudges regulators and industry groups to monitor volume use and urge proper waste treatment. My colleagues tend to play it safe, following good laboratory practices, and closely read each new study that explores either acute or reproductive toxicity, even if the results consistently show low risk at typical workplace concentrations.
Looking Ahead: Where This Compound Fits in the Future
The scientific community’s interest in magnesium triflate isn’t fleeting. Rising demand for sustainable batteries and greener synthesis fuels ongoing investment in the chemistry. Industrial partners hunt for scalable routes that deliver consistent purity and cut down on environmental impact. Among battery researchers, the hunt for greener ionically conductive salts keeps magnesium triflate firmly in the conversation, along with calls for even more robust toxicity and recycling studies. Organic chemists continue exploring modifications to the triflate structure, aiming to refine selectivity or enable compatibility with new solvents. Regulatory bodies, meanwhile, keep pushing for clarity on environmental breakdown and waste management, making sure the progress in labs and factories doesn’t blindside the world outside. Looking at the landscape, one thing becomes clear: as long as research keeps aiming for safer, cleaner, and smarter chemical approaches, magnesium trifluoromethanesulfonate remains hard to ignore.
Why Chemists Turn to Magnesium Triflate
Magnesium trifluoromethanesulfonate, or magnesium triflate, isn't the kind of chemical most people keep tucked away at home, but it gets plenty of attention in the world of research labs. Chemists put it to work not for its name, which usually gets shortened, but for how well it behaves as a catalyst and as a part of more advanced materials. I still remember my days shadowing a doctoral student, watching as she dived into reactions that simply refused to go forward until magnesium triflate hit the flask. That was my introduction to how one ingredient can turn a headache into a breakthrough.
Driving Innovation in Synthesis
This salt opens doors in organic synthesis. Organic chemists often face reactions that stall out or yield too many unwanted byproducts. Magnesium triflate tends to keep things clean. Its role as a Lewis acid means it helps pull electrons where they're needed, speeding up reactions and bumping up yields. I read a study out of a European university where researchers swapped their old acid catalysts for magnesium triflate and shaved hours off their timelines, with fewer clean-up steps. They pointed to fewer side reactions, which matters when you're not just making samples but trying to scale up a process safely.
Stepping Up Battery and Electrolyte Tech
It’s not just about making new molecules in a fume hood. Magnesium triflate starts popping up in some pretty futuristic applications. Energy storage stands high on that list. People are always talking lithium, but magnesium has been getting more attention as an alternative for batteries. Here, magnesium triflate acts as an electrolyte salt. It works in non-aqueous electrolytes, which helps when designers want energy-dense, safe batteries. Tracing battery research, I’ve seen this salt improve conductivity and support more stable cycling, which brings us closer to safer, more affordable batteries, at least in theory. Improvements don’t always come overnight, though; scaling up remains a hard climb.
Shaping the Future of Green Chemistry
Sustainability in chemistry gets more press than it did a decade ago. Magnesium triflate fits this shift. It’s less corrosive compared to traditional acids and, for certain processes, produces fewer hazardous byproducts. The fact that it stays stable and works efficiently in many solvents means less waste and less fuss during purifications. From green synthesis of pharmaceuticals to more environmentally friendly materials, this salt matters. Of course, sourcing raw materials responsibly and finding smarter ways to recycle and reuse products still needs solving.
Looking at Challenges and What Could Help
Every tool in a lab comes with tradeoffs. Magnesium triflate often costs more than older catalyst salts, which makes penny-pinching researchers pause before pulling it off the shelf. Its solubility, though impressive in some solvents, doesn’t always match up to every need. The bigger challenge lies in public awareness—people fixate on the latest polymers or battery breakthroughs but rarely hear about the small, supporting salts that make those stories possible.
To keep progress moving, more sharing between labs would help. If researchers openly published both their successes and their dead ends with magnesium triflate, everyone could get a clearer picture, avoiding the trap of repeating the same mistakes. Funding for sustainable chemistry should cover not just headline-grabbing inventions, but also the everyday materials and catalysts that keep the machine humming.
The Real Work Behind the Scenes
Magnesium trifluoromethanesulfonate might not show up in daily headlines, but it hums along in the background of real scientific progress. From laboratory benches to new battery designs, it’s often these unsung compounds that quietly drive the most important changes in how our materials and energy solutions take shape.
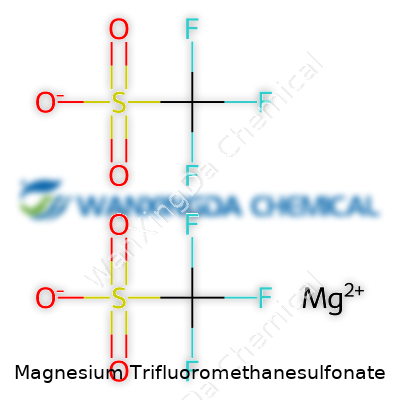
The Numbers Behind the Name
Magnesium trifluoromethanesulfonate carries the chemical formula Mg(CF3SO3)2. This formula tells a story ― two trifluoromethanesulfonate groups paired with one magnesium ion. The molecular weight lands near 322.4 grams per mole. The presence of those CF3 groups adds some heft to the compound, and the magnesium slips into the middle of the mix, quiet but vital.
Why the Details Matter
Getting the formula and weight right isn’t just a chemistry class exercise. Chemistry can feel remote until you realize how the fundamentals link to real work. In my lab days, small errors in a chemical formula led to big headaches ― reactions sputtered or shut down, and nobody likes tracking down why a synthesis failed. Whenever I handled magnesium trifluoromethanesulfonate, even slight uncertainty about the chemistry made complex projects look shaky. Research has shown that overlooked details with similar compounds have derailed years of work, underlining why sparse data still packs a punch.
Electrolytes and Real Value
Magnesium trifluoromethanesulfonate often serves as a go-to source of magnesium for electrochemical experiments and battery research. Since lithium salt supplies face disruptions and cost spikes, some eyes have turned to magnesium. In my experience, magnesium’s supply chain runs steadier and cheaper than lithium. The salt’s manageable weight and clear structure mean researchers can accurately dose it and predict how it’ll dissolve or precipitate. This lowers experimental risk and brings consistency to projects.
Not all chemicals get the same treatment in the literature, and even well-known ones sometimes get glossed over. Students embarking on synthesis projects face the issue when they pull a tub off the shelf, only to squint at the label and ask, “Is this what I’m supposed to add?” Getting the chemical formula printed clearly—and teaching what each element in the formula stands for—builds a foundation for safe and reproducible work. A correct molecular weight guides those prepping electrolyte solutions, not just in academia but also in the labs where batteries take shape and find their way into gadgets and cars.
Bridging the Gaps in Practice
Errors don’t just trip up single reactions; they echo through entire supply chains. I’ve seen team members troubleshoot a failed batch of electrolyte, only to realize the base assumption about the salt’s makeup didn’t match reality. Double-checking the formula and molecular weight sounds basic, but in the heat of an experiment that feels like the last thing anyone does. Chemistry thrives on curiosity, but it runs on precision. Building routines where every step gets checked—formula, weight, label—saves both frustration and cost.
Strengthening the Chain of Trust
Reliable facts turn up in peer-reviewed literature, industry bulletins, and trusted supplier documentation. Over time, you start to spot trusted voices. I lean on reputable sources and stick to the rule—never trust any single fact if it can’t be backed up from multiple directions.
In the end, knowing the chemical makeup and how much a scoop weighs bridges raw discovery with real-world progress. Magnesium trifluoromethanesulfonate doesn’t land the headlines, but for plenty of chemists, batteries, and researchers, those details mark the line between confusion and innovation.
Insights from the Bench: Real-Life Chemistry Lessons
Magnesium trifluoromethanesulfonate doesn’t get much spotlight outside the lab, but this salt shapes quite a few reactions across organic chemistry. Solubility, in my experience, decides success or failure in more syntheses than fancy new reagents do. For researchers and students who’ve wrestled a stubborn, undissolved salt at the bottom of the flask, the question “What dissolves magnesium trifluoromethanesulfonate?” isn’t academic—it’s the key to actually making a reaction work.
What Happens in Water?
In the practical world, magnesium trifluoromethanesulfonate shows pretty strong affinity for water. Its structure brings together a magnesium ion and the triflate anion, which both encourage water molecules to surround and separate them. Anyone who’s mixed this salt with distilled water will notice it goes fully into solution without much fuss—provided the temperature isn’t set to Antarctic levels and you don’t try to dump in supersaturated amounts at once. That ease of dissolution matches up with the underlying chemistry: both magnesium and the triflate group form good interactions with water molecules.
Solubility in water doesn’t just simplify handling. It makes this salt a reliable choice in catalysis for aqueous and mixed-phase reactions. You can fine-tune concentrations without spending half an hour on sonication or standing at the stir plate, which really adds up during long days in the lab.
Behavior in Organic Solvents: Where It Gets Interesting
Solubility isn’t always so straightforward in organic solvents. Flask after flask, researchers quickly notice that non-polar solvents like hexane leave magnesium trifluoromethanesulfonate largely behind. It doesn’t go into solution well, so for work in purely non-polar environments, frustration can follow. This result isn’t surprising. The ionic nature of the salt wants more polar surroundings than hexane can supply.
Switch to more polar organic solvents—think methanol, acetonitrile, or DMF—and the story changes. The salt becomes much more willing to dissolve. This trend mirrors what’s seen with many salts that feature highly delocalized anions, especially the triflate family. It’s this accessible solubility in strong and moderately polar solvents that lets researchers use magnesium trifluoromethanesulfonate as a supporting electrolyte and as a Lewis acid catalyst outside aqueous systems.
What Does It Mean for Chemistry and Industry?
Access to salts that move smoothly between different solvent environments reshapes how labs approach tough problems. The solubility profile of magnesium trifluoromethanesulfonate doesn’t just impact bench chemistry—it guides choices in pharmaceuticals, battery development, and catalysis. Labs designing greener reactions or more efficient batteries rely on salts that won’t create bottlenecks in supply or purification. This magnesium salt’s behavior in solution gives a boost to those hoping to reduce use of toxic or poorly soluble alternatives.
Scale-up teams also depend on reproducible, clear-cut solubility—without this, results wobble or costs rise as teams scramble with workarounds. Keeping solubility data transparent and based on real lab conditions, not just theoretical tables, supports reproducible science and straightforward quality control.
Raising the Bar for Shared Knowledge
Great science grows when experiences move beyond the individual researcher. Sharing practical notes on magnesium trifluoromethanesulfonate—in water, in methanol, or in more exotic blends—lets everyone build on strong foundations instead of reinventing the wheel one flask at a time. Real-world knowledge, rooted in what actually happens at the bench, keeps chemical research honest and pushes results closer to solutions that hold up in both the lab and industry.
Everyday Precautions with Powerful Chemicals
Magnesium trifluoromethanesulfonate doesn’t sound like something you’d keep under the kitchen sink—but in laboratories and some industrial settings, it finds its way into more experiments and processes than one might think. This chemical plays a role in synthesis, batteries, and catalytic reactions, so many labs keep a bottle on the shelf. Safe storage and handling don’t need to be complicated, but overlooking the basics invites trouble no scientist or technician wants on their hands.
Why Moisture Matters
Chemistry comes with hard-earned lessons. There’s nothing quite like the mess from a hygroscopic salt that has soaked up water from the air, caked into clumps, or started reacting in unexpected ways. Magnesium trifluoromethanesulfonate pulls water from the air readily. This sounds minor, but anyone who’s tried to weigh a sample that’s already started absorbing moisture knows how frustrating things can get.
To avoid frustration—or worse, failed experiments—keep this chemical in tightly sealed containers. Glass with PTFE-lined caps works especially well, as experience in shared research labs shows that regular lids sometimes let enough air in to make storage unreliable. Just a couple of experiences with ruined batches drive home the fact that the right container pays for itself.
Temperature and Light: Keep It Cool, Keep It Dark
Magnesium trifluoromethanesulfonate remains fairly stable, but leaving it under a lab bench exposed to every light and heat source makes little sense. Cool, dry cabinets do the job. In my work for a battery startup, storage in a simple climate-controlled cabinet cut down on mysterious failures. Temperature swings speed up chemical changes, and even hardy salts start to break down if they sit in less-than-ideal spots for months at a stretch.
Direct sunlight just makes things worse, as even solid compounds can slowly change under ultraviolet exposure. Stick with the habit of putting away chemicals in a shaded space—the fewer surprises, the better.
Handle with Respect—Even Without Panic
It doesn’t pay to treat any laboratory chemical casually. For most people, gloves and safety goggles seem like overkill for routine transfers, but erring on the side of caution always makes sense. Spills are rare if you use a scoop and support the container, but nobody plans to drop a jar at the wrong moment. Even experienced scientists and students learn the hard way how quickly a clean lab can become a sticky, difficult mess.
Some publications highlight the low acute toxicity of magnesium trifluoromethanesulfonate, but skin contact or eye splashes can still cause irritation. It doesn’t harm to have good habits, especially if other, more hazardous reagents sit close by. Clean up spills with care, making sure powder or residues go into the right waste containers. The best-run labs I’ve seen put fresh labels on every container and keep a written log, so nothing gets mixed up or forgotten.
Building Lasting Laboratory Habits
Safe storage and smart handling aren’t just rules in the lab—they make up the rhythm of working around chemicals day after day. Training young researchers, I’ve noticed mistakes usually pop up right as routines get sloppy. Take the time to run through best practices now, and you’ll spend less time dealing with preventable problems. Sharing what works from personal experience helps drive home why it matters to follow the steps, label things, and never take shortcuts.
Magnesium trifluoromethanesulfonate deserves the same caution as any specialized reagent. Time spent setting up storage right, wearing proper gear, and staying alert during transfers keeps projects on track and people safe. With a little discipline—learned the good way or the hard way—these everyday moments become second nature. That kind of reliability is worth aiming for, no matter how advanced the science in the room.
Magnesium Trifluoromethanesulfonate rarely gets any attention outside of chemical circles, yet it finds its way into countless labs and some cutting-edge industries. Those who work around it can’t ignore the real risks, even if it doesn’t sound as volatile as some other reagents. I’ve spent enough time with strange chemicals to know hazard signs rarely shout. Sometimes you find out the hard way that ordinary-looking powders demand respect.
The Real Talk on Handling
Most chemists treat Magnesium Trifluoromethanesulfonate as a dry, crystalline salt—heavy in the hand and not outwardly aggressive. But don’t let that vibe fool you. Tiny grains end up everywhere. If you track them home under fingernails or in cuffs, you’ll carry traces where they’re neither expected nor wanted. Always use gloves and a reliable dust mask or respirator if the material becomes airborne. Safety glasses aren’t optional just because the risk isn’t obvious; a chemist once told me he learned that after a routine day left his eye watering for hours from an unexpected splash.
Working in a fume hood helps a lot, especially during weighing or mixing. Even if you feel confident handling other magnesium salts, this one needs similar precautions—gloves rated for chemical resistance, careful containment, and minimizing skin exposure.
Breaking Down Exposure Risks
Bodily exposure to Magnesium Trifluoromethanesulfonate isn’t usually dramatic, but repeated contact dries out skin, sometimes cracking it wide open. That’s more than a nuisance: open skin offers a path straight inside, and I’ve seen cases where chronic handling leads to allergies or skin sensitivities. Eyes sting painfully if dust lands there, and inhaling fine particles could irritate airways.
Accidents—spills, dust clouds on a breezy day, or a dropped jar—play out faster than anyone expects. Wet things down immediately to keep dust from rising, and vacuum rather than sweep. Washing up after handling isn’t just habit; it’s a way to end small exposures you don’t realize happened. I’ve caught myself with streaks of white dust on my wrists hours after thinking I’d “scrubbed in.”
Thinking Long-Term and Responsibility
Some older safety sheets barely covered long-term toxicity. Modern understanding recognizes the environmental angle—waste disposal really matters. Water systems don’t filter out every fancy new compound, and something as persistent as a triflate ends up downstream. Label and store this chemical well away from food, and don’t pour anything down normal drains. Landfill isn't much safer. Instead, check if your institution has a specialist disposal program. Personal responsibility adds up, especially when a growing crowd starts working with niche compounds.
I talk a lot with up-and-coming chemists about what “good lab technique” actually means beyond passing inspections. Preparation makes mistakes less likely. It means keeping a tidy bench, knowing the quirks of the material you’re using, never skipping labels, and making sure anyone else working in the same space knows what’s in use.
Moving Toward Safer Habits
Speaking from experience and from watching mistakes in real labs, the big change in safety doesn’t only come from stricter rules. It comes from building habits—regularly checking for cuts on your hands, never setting an open jar where it could tip, logging waste carefully, refusing to work without personal protective equipment. It’s not dramatic, it’s steady, and it’s how people keep one another safe.