Mannose Triflate: A Deep Dive Into Its Past, Present, and Potential
The Path Taken: From Basic Sugar to Indispensable Reagent
Mannose triflate sits at a strange crossroads in the world of chemical reagents. It starts life as something ordinary – mannose, a simple sugar found in a host of natural sources from cranberries to aloe vera. That regular sugar carries biological weight in its own right, but chemists figured out decades ago that attaching a triflate group turns dull carbons into reactive handles. The story of mannose triflate stands as another example of how the tiniest changes in a molecule can open up fresh routes in research labs worldwide. People sometimes forget the winding paths these molecules take on their journey from raw materials through custom synthesis, each step shaped by experimental frustration and sudden realization. The earliest reliable preparations started cropping up during the expansion of carbohydrate chemistry, piggybacking on advances in organic synthesis and the rising popularity of triflate leaving groups. What matters today is not just the recipe itself, but the way it unites fields once thought separate: medicinal chemistry, glycobiology, and the hunt for better pharmaceuticals.
Physical Substance and Chemical Attitude
At room temperature, mannose triflate shows up as a white to off-white powder, powdered enough to glimmer between a chemist’s fingers if the jar tips during weighing. Moisture gets along poorly with it; most labs find themselves storing it under dry, cold conditions for this reason. In solution, especially in polar aprotic solvents, the compound dissolves well and then waits for the next step. The triflate (trifluoromethanesulfonate) group turns it into a strong electrophile, meaning carbons once reluctant to accept newcomers suddenly greet nucleophiles with open arms. This property prods chemists to reach for mannose triflate when they want to build up sugar chains, create libraries of glycomimetics, or tweak the sugar backbone for drug development. Ordinary sugars never offered this type of reactivity.
Crucial Details: Specifications and Labels Aren’t Just for Boxes
Every bottle of mannose triflate comes with fine print, pointing to purity, content, and appearance. That’s not about bureaucracy but about protecting experiments from derailment. Purity above 97% protects ambitious synthesis from hidden side products. Melting point ranges do more than satisfy curiosity—they alert researchers to unexpected impurities or troublesome by-products. If a batch picks up moisture or biological contamination, the end result swings wildly, causing hours of wasted troubleshooting. That makes attention to labeling, storage temperature, and supplier reputation more than formalities. In a business that turns on the presence or absence of single percentage points, these technical details affect everything from pharmaceutical trials to basic bench research.
Making Mannose Triflate: Lab Techniques That Make or Break a Reagent
Behind the bottle, a story of finicky glassware and persistent hands unfolds. Synthesis usually begins with protecting groups on the parent mannose, keeping all but the target carbon tucked away behind chemical shields. Adding triflic anhydride under controlled, cold conditions triggers the transformation as the triflate group replaces a selected hydroxyl. Each step invites complications—overreaction, loss of selectivity, unwanted rearrangements. Routine work in carbohydrate chemistry turns surprisingly unpredictable once triflation enters the mix. After the reaction ends, careful workup and repeated crystallization pull the wanted product from a noisy mixture of close relatives. The recipe may run just a few steps but mastering the details takes years off a chemist's patience.
How Chemists Use It: Transformations and Tweaks Across the Board
The triflate handle isn’t an end in itself. Researchers lean on mannose triflate during glycosylation, swapping sugars to alter biological recognition, or tacking on unusual groups to chase down new activities. The triflate leaves easily, letting nucleophiles like thiols, alcohols, or amines step in and make new bonds. Each time a new derivative shows biological promise, more hands reach for another batch of triflate to scale up or iterate further. The willingness of mannose triflate to step aside brings flexibility that sugars rarely offer on their own.
Other Names, Same Molecule
Literature reflects the story of a compound through the names it gathers along the way. Mannose triflate is known just as often as mannopyranosyl triflate or mannosyl trifluoromethanesulfonate, depending on whether an author wants to highlight the sugar or the leaving group. Synonyms pile up with minor spelling shifts or the occasional reference to the exact stereochemistry, but in the end, the labs chasing the same chemistry know what’s in the bottle in front of them, regardless of nomenclature.
Staying Safe and Playing Clean: Safety and Standards in the Lab
Using mannose triflate signals a jump into chemistry that demands respect. The triflate group stands out not just for reactivity, but also for the hazards tied to it. Spills eat at metals, residues can contaminate bench tops, and inhalation risks rise when the powder goes airborne. Responsible labs train researchers to avoid water, vent well, and suit up in gloves, goggles, and sometimes even face shields. Regulatory frameworks urge up-to-date hazard labeling, waste handling, and written protocols. Rather than stifle creativity, these standards have helped labs avoid disaster, letting chemists focus on results instead of clean-up or injury.
Application: Beyond the Label, Into the Field
The impact of mannose triflate becomes obvious looking across research papers in fields as diverse as antiviral drug discovery, glycomics, and the slow churn of vaccine development. Its ability to produce rare glycosides, enable selective derivatization, or help build libraries of sugar analogues turns it from chemical oddity to mainstay. In the hands of synthetic chemists, new carbohydrate-based drugs take shape faster. Biologists find themselves able to probe cell signaling, pathogen binding, or enzyme selectivity at a level of detail the older chemical toolbox never allowed. Every success points back to those bottlenecks broken open by a single reactive sugar.
The Research Frontier: Current Focus and Roadblocks
Current research on mannose triflate digs into making its handling less finicky, broadening the range of sugars available for reaction, and linking its chemistry to automated synthesis. Alongside that, questions remain about scaling up for pharmaceutical production, managing by-products, and ensuring each transformation yields exactly what’s expected, especially in complex biological settings. Certain mana-analogues produced from mannose triflate have moved toward clinical curiosity, though only a handful cross the threshold into approved therapy or diagnostic reagents. The tension between promise and risk keeps the field lively.
Toxicity and Health Impacts
For all its lab utility, mannose triflate brings toxicological questions like any reactive intermediate. Experimental data points to irritation on contact and potential issues for respiratory health in confined or poorly ventilated spaces. The triflate leaving group, derived from strong acids like triflic acid, adds to the caution—accidental exposure can damage skin and mucous membranes, and long-term effects have not been mapped as thoroughly as with older, more common reagents. Responsible use means limiting exposure, applying good engineering controls, and never letting convenience trump risk assessment.
Looking Forward: Where Mannose Triflate Might Lead
Looking years ahead, chemists see a future where mannose triflate’s reactivity opens new avenues in automated carbohydrate synthesis, making glycan arrays and complex oligosaccharides more accessible for biological screening. The need for better antivirals and vaccines gives this chemistry a shot at direct societal impact, focusing creative minds on routes to tailor-make previously unreachable glycosides. Alongside innovation, demands will keep rising for safer, less wasteful synthesis and more environmentally friendly alternatives—areas where new research has a chance to reshape even familiar components. The journey from a hard-earned powder on the lab shelf to a catalyst for medical breakthrough or chemical shift rests not on luck, but on the hands and minds willing to rethink, reformulate, and refine.
Digging into the World of Mannose Triflate
Any scientist who has worked in a carbohydrate chemistry lab knows that some molecules transform what’s possible. Mannose triflate, a derivative of the simple sugar mannose, stands out in research circles. Instead of seeing its name pop up in mainstream headlines, you usually find it in the methods sections of complicated papers or whispered about in corridors of academic chemistry buildings. Yet, its presence makes a real difference in how scientists build sugars that feed directly into vaccine design, drug development, and glycoprotein synthesis.
The Backbone of Advanced Sugar Chemistry
Glycosylation shapes life’s building blocks, from human biology through to industrial enzymes. I remember in graduate school, my research group juggled dozens of chemicals, but everything slowed to a crawl when we ran out of mannose triflate. The reason? It's a lynchpin for creating activated sugar donors. Chemists attach the triflate group to the mannose molecule, which then turns a sluggish sugar into a reactive intermediate. This “activated” form can connect with other molecules much more smoothly than the bare sugar. Suddenly, the clunky process of building elaborate sugar chains for antibody research or antiviral studies becomes much more efficient and controlled.
Impact on Medical and Biological Research
Strong progress in vaccines and novel drugs often follows from clever sugar chemistry. Mannose triflate speeds up and improves the design of complex carbohydrates. One impressive example sits in the field of HIV vaccine research. Scientists are keen to synthesize oligosaccharides that resemble those found on the HIV envelope. These sugar structures challenge synthetic chemists—without the right “building blocks,” making enough for testing can take months or years. Mannose triflate streamlines these syntheses, cutting time and often saving research budgets.
Challenges: Safety and Environmental Questions
With all the promise around mannose triflate, chemists keep an eye on safety, both in the lab and for the environment. Triflates in general can raise red flags for handling and disposal. They create reactive byproducts, and anyone who mixes large batches quickly learns to treat them with respect. Research institutions train staff and strictly manage waste streams for chemicals like these. Pushing the industry towards greener chemistry runs through conversations at every level, especially as triflate chemistry catches on in more labs. Some researchers experiment with alternative leaving groups or milder reagents, hunting for options that balance performance and sustainability.
Where to Go from Here
The promise of mannose triflate lies not only in what it accomplishes today but also in how it pushes the whole field forward. More labs with better access to standardized, pure reagents can participate in the global effort to innovate. Sharing know-how improves reliability and speeds up those tiny sugar transformations that power drug research. Scientists working with this molecule look out for better recycling processes and safer alternatives, all while celebrating the gains in yield, purity, and time.
Mannose triflate doesn't grab public attention like a wonder drug or a therapy breakthrough, but the behind-the-scenes stories reveal where the next advances could come from. Tight collaborations between chemists, pharma researchers, and regulatory experts unlock the full value of these building blocks. As the world expects faster progress in immunotherapies, vaccines, and diagnostics, the supporting actors like mannose triflate grow in importance. Each leap forward in sugar chemistry echoes through the halls of scientific discovery, helping us move one step closer to more effective medicine and fundamental understanding.
Science Beyond the Lab Bench
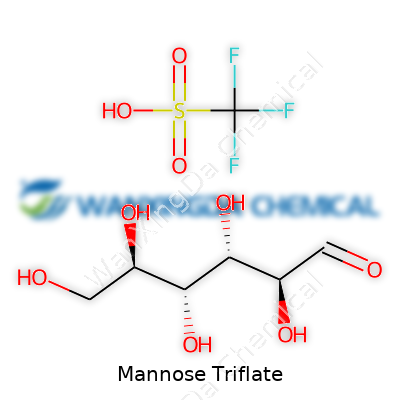
Mannose triflate might sound like a compound tucked away in a chemistry textbook, but it tells a bigger story about how molecules become building blocks for life-changing products. I remember sifting through small vials in the lab back in college, looking for just the right chemical to move a project forward. Pieces like mannose triflate often end up enabling bioengineering, medical treatments, and even green energy tech. Yet details like its formula—C6H11FO9S—can get buried under jargon. It’s worth pausing to untangle why this small bit of information matters.
A Closer Look at Mannose Triflate’s Formula
C6H11FO9S doesn’t just tell you what atoms are present; it unlocks paths for synthetic chemists. The “triflate” attached to mannose here stands for trifluoromethanesulfonate, though in mannose triflate, the structure presents as a single triflate substitution. That means swapping a simple hydroxyl group for a more reactive site—making mannose more than just a sugar. For folks trying to modify natural sugars, this triflate acts like a glass cutter for molecules—offering a neat, precise entry for further transformations.
From a practical side, the ability to functionalize sugars quickly impacts not just theoretical chemistry but also real tools for drug design. Many antibiotics, antivirals, and even some cancer drugs borrow from the “glycosylation” patterns found in nature. Creating these patterns in a factory setting relies on tight control over how new parts are added or removed. Mannose triflate’s formula C6H11FO9S gives scientists a shorthand to plan and predict such modifications.
Trusting Chemistry in the Age of Complexity
Back during my graduate studies, I noticed how skeptical people can get when science moves out of academic journals and into wider use—sometimes rightly so. Misstatements about basic facts, even something as simple as a molecular formula, can erode trust. That’s where provenence matters. The chemical formula of mannose triflate, supported by peer-reviewed literature, carries weight in both academic and industrial projects. Nobody wants a recall or dangerous misstep based on a careless mix-up of molecules.
Bridging Knowledge Gaps
If you ask a working chemist or a biotech engineer, the formula C6H11FO9S is just the start. The real challenge comes from scale-up: how to take a few milligrams of mannose triflate and make hundreds of grams safely and sustainably. Chemical manufacturing has improved, with better ventilation, waste treatment, and worker protection. Some green chemistry labs are even exploring ways to minimize harsh reagents when making sugar derivatives like mannose triflate.
Academic outreach can help, too. When science classes bridge the gap by showing students why a molecular formula carries real-world impact, it sticks better than dry memorization. I saw this firsthand when undergraduates synthesized sugar analogs and saw their hard work translate to useful tools for researchers studying rare diseases.
Moving Forward with Clarity and Purpose
Each time we highlight core details like the formula for mannose triflate, we help steer conversation toward transparency and accuracy. Knowledge about chemicals should empower more than just specialists. A clear understanding of molecules lets a wider group—from students to policymakers—ask smarter questions and seek better outcomes.
Small Oversights Carry Big Risks in the Lab
Researchers and lab techs juggle a range of chemicals every day, but not all compounds cause the same headaches. Mannose triflate, for those in organic synthesis or carbohydrate chemistry, ranks near the top for things you don’t want to store carelessly. One hard lesson learned in graduate school sticks with me: sloppy storage can turn a productive morning into hours of cleanup—and wasted money. The stakes only go up when you’re scaling work or sharing bench space.
Why Does It Matter?
Mannose triflate isn’t just any sugar derivative. Its high reactivity makes it invaluable for specific glycosylation reactions, yet that same property invites trouble. Exposing it to ambient air, humidity, or light can cut shelf life or lead to decomposition. Losing a valuable batch means wasted grant dollars and tight timelines stretched even thinner.
Chemical supply data often emphasize dry, cool, dark storage conditions for similar compounds. That advice isn’t just regulatory box-checking; it comes from real cases where hydrolysis or gradual breakdown eats away at product quality. A single careless hour on a cluttered shelf might quietly do what weeks in the freezer wouldn’t.
Best Practices from Real Lab Life
A -20°C freezer forms the gold standard for storage, especially for anything with a triflate group that’s eyeing the nearest water molecule to react with. Still, not every lab has endless freezer space. Laughable as it sounds, I’ve watched researchers stash similar compounds behind lunch boxes or in worn-out fridges. This rarely ends well.
Silica gel packets or a working desiccator pay off when humidity is high. Good lab labels—date opened and initials—aren’t just administrative tedium. It’s too easy to forget the details after a few months. A sealed glass bottle beats capless plastic for blocking moisture, and transparent bottles make for tempting shortcuts that backfire under fluorescent lighting.
Understanding What Lies Beneath
Most accidents don’t start with a dramatic spill. Problems creep up with small cracks in container lids, condensation after the bottle sits out, and quick moves to weigh out a portion without warming to room temperature properly. Many labs overlook these minor steps when under pressure to hurry experiments.
The chemistry speaks for itself: triflate esters act as strong electrophiles, and water is everywhere. Even trace moisture slowly transforms useful reagent into sludge. Discoloration, clumping, or subtle losses in performance during reactions usually trace back to a lazy afternoon at the bench. Each student group I’ve trained takes away the same message—build the routine, and you avoid repeat mistakes.
What Can We Do Better?
Labs benefit from assigning clear responsibility for chemical inventory checks. Rotating who double-checks temperatures, ensures caps are tight, and looks for early signs of trouble helps everyone. Investing in proper storage containers and stickers feels dull until replacements cost ten times more.
Universities and chemical suppliers could do more than bury the essentials in safety data sheets: real-world case stories pack a bigger punch. In some research hubs, group meetings end with a quick pointer about best practices or quick stories about times things went wrong.
Keeping Mannose Triflate Useful Longer
Experience proves that careful storage feels like extra work at first, but no one regrets the habit when experiments run smoothly. Reproducible results and safer labs go hand in hand with sensible, proven routines for tricky reagents. In the end, remembering where and how you store that tiny bottle means the difference between progress and lost days recounting mistakes.
Understanding Mannose Triflate
Chemical names can sound intimidating. Mannose triflate is one of those mouthfuls that might set off alarm bells for folks outside the lab. It belongs to a class of compounds often used in specialized organic synthesis, especially when crafting complex sugars or tweaking molecular structures for biological research. Its role might seem distant from everyday life, but its safety profile deserves a closer look.
Why Chemical Hazards Matter in the Lab
Researchers, chemists, and students encounter many reagents like mannose triflate. Labs usually deal with small quantities, but the significance of safety isn’t just about how much—it's about how those molecules behave. I remember once working on a sugar modification project, where strict safety protocols guided even the smallest transfer of chemicals. That’s because triflates as a group tend to have strong reactivity, especially with bases or moisture, leading to potentially harmful situations if handled carelessly.
Known Risks of Triflate Compounds and Mannose Triflate
Mannose triflate carries the triflyl group, linked to strong acid derivatives. Many triflates can irritate skin, eyes, and airways if mishandled. The exposure risk grows if powders become airborne or if spills occur. For mannose triflate specifically, there's no broad toxicity data published, but its similar relatives commonly warrant eye and respiratory protection. Evidence in chemical literature points to mild to moderate hazards, best comparable with other organic reagents handled every day in research.
Accidental ingestion or persistent skin exposure raises most of the concern. Solvents often used with mannose triflate, such as dichloromethane or acetonitrile, usually call for fume hood use not only because they evaporate easily but because they can carry particles or traces of the reactive sugar onto surfaces or into lungs. It’s not just about the substance itself—it’s about the environment and habits built around it.
Regulation, Precautions, and Responsibility
Regulatory agencies like OSHA and the European Chemicals Agency regularly review chemicals like triflates for lab safety. They rely on researchers’ data, companies' submissions, and published findings. Where data gaps exist, regulations demand caution, personal protective equipment, and ventilation. Common sense, too, forms the backbone of lab safety—no eating or drinking near chemicals, gloves changed often, and spills dealt with right away.
One solution: clear, updated safety data sheets. These documents rarely turn up in classrooms, though everyone working with new reagents deserves to see them. In practice, enforcing a strict fume hood policy and proper disposal keeps risks low. Spreading this culture of shared responsibility makes science safer. It saves costs, prevents injuries, and protects careers.
Looking Forward: Handling Science With Care
Every compound brings value and risk—mannose triflate included. Calling it dangerous oversimplifies, assuming someone respects the lab’s rules. Complacency provides the real danger. With more public awareness and improved communication among researchers, safety becomes a habit rather than an afterthought. Whether a scientist or a curious observer, knowing the risks changes the way people view the wonders and hazards tucked inside those labeled bottles.
Why Purity Matters for Mannose Triflate
Mannose triflate often shows up in stories about complex carbohydrate chemistry and cutting-edge biotech research. Some see it as just another reagent, but for those working with enzymes, drug candidates, or synthesis of rare sugar derivatives, subtle flaws in purity can derail an entire project. From my own time in a university lab, a failed experiment usually traced back to some detail like this, not a grand mistake. That’s how critical the purity of such reagents can be.
Labs count on Mannose triflate to perform as expected. Impurities can trigger misleading results or introduce unexpected byproducts, especially in glycosylation reactions or analytical setups. Purity doesn’t just protect a sample; it also sets the tone for reliable, reproducible data, and smooth regulatory review. Sloppy chemistry rarely pays off, and in my experience, a few rogue impurities have made months of work obsolete. The stakes are real, especially in pharmaceutical or vaccine development, where every contaminant represents a new unknown.
Typical Purity Specifications in the Marketplace
Most suppliers provide Mannose triflate with purity levels exceeding 95%. Some top out at around 98%, and a handful manage to supply even higher grades when the application demands it. These aren’t random numbers—high-performance liquid chromatography (HPLC), nuclear magnetic resonance (NMR), or mass spectrometry usually back up the claim. The demand for this degree of purity comes from necessity. For anyone doing work in glycoscience or on biologically active structures, even a slight difference in composition could mean results that can’t be repeated by anyone else, throwing confidence out the window.
Impurities linger for many reasons: process byproducts, leftover solvents, or even breakdown products from the manufacturing step. If a batch carries any significant leftover, researchers risk seeing peaks in analytics that should not be there. Safety becomes an issue in pharmaceutical research since byproducts may pose toxicological concerns, especially as regulatory scrutiny grows. Higher-purity batches avoid these pitfalls and help make sure new findings stand the test of time and peer review.
How Users Can Manage Purity Concerns
Quality documentation really matters. Anyone sourcing Mannose triflate for meaningful work shouldn’t accept vague claims or half-hearted certificates. Suppliers able to share detailed analytical reports—ideally with chromatograms and spectral data—build trust. Many research teams set their own minimum thresholds and perform in-house testing, comparing supplier data to independent measurements. Relying solely on catalog numbers adds risk. In a lab I worked in, we standardized our verifications by batch, insisting on duplicate HPLC runs. It wasn’t overkill; it protected our results when collaborations and journal reviewers raised eyebrows.
Another piece worth attention is storage and handling. Even top-grade Mannose triflate won’t stay pure if it soaks up moisture or suffers from improper sealing. Institutions committed to quality adopt best practices in storage, using desiccators and rapid handling to keep air exposure low. Nothing replaces careful technique at the bench. Consistency in handling guards against purity drifting over time, making compliance with internal and external standards much easier for future projects.
Raising the Bar for Scientific Progress
The pressure is real—in academic labs, commercial settings, and beyond—to show results that not only break new ground but will stand up to scrutiny tomorrow. Pure Mannose triflate sits closer to the foundation of these efforts than many realize. Investing in thorough verification and demanding transparency from suppliers doesn’t just protect a single experiment. It supports stronger science, better data, and the trust that every field needs to keep moving forward.