Methanesulfonic Anhydride: Chemistry, Context, and Considerations
Tracing Its Path: How Methanesulfonic Anhydride Stepped Onto the Scene
Tucked away in the stories of industrial progress, methanesulfonic anhydride occupies a spot that speaks to a rare blend of necessity and discovery. Chemists began piecing together its structure and role once methanesulfonic acid’s reactivity made it clear that higher-efficiency sulfonylation tools had room in the lab. Syntheses pointed out gaps—places where strong yet manageable dehydrating agents helped open new transformations. Decades of sulfur chemistry pushed researchers to find alternatives to overused anhydrides like acetic anhydride, in part because the growing focus on selectivity and safety called for a reexamination of the old toolset. Here, methanesulfonic anhydride carved out its territory, winning attention for sharp reactivity and easier handling compared to some legacy chemicals.
A Closer Look: What This Chemical Brings to the Table
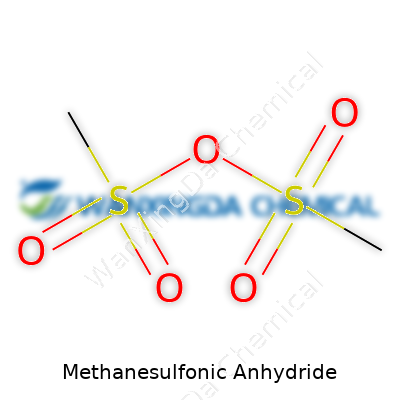
One thing crystalizes fast: methanesulfonic anhydride doesn’t mess around. The molecule, made up of two methanesulfonyl groups joined by an oxygen atom, carries a formula C2H6O5S2. Its physical appearance brings to mind colorless liquids, sometimes manifesting as faintly viscous. A boiling point north of 150 °C means it tolerates moderate lab heat, sidestepping hazards of low-boiling, hard-to-contain substances. Folks in the field notice that it tends to give off a pungent, often irritating odor, another sign to handle with awareness. It dissolves decently in most organic solvents, lining up with its preference for working alongside familiar processes in organic labs. The aggressive sulfonylating power stands out in a world crowded with milder options.
Understanding Technical Specifications and Labeling
Getting the details right on bottles and boxes means more than just ticking compliance boxes. Clear labeling reflects decades of mishaps and lessons learned. Labs routinely track purity upwards of 98%, since low-level contaminants influence sensitive syntheses. Moisture plays villain—a trace amount can ignite decomposition or cut yield in water-sensitive steps. Containers usually call for cool, dry storage, away from strong bases or oxidizers. As chemists, many of us know the difference between success and disaster can turn on a misplaced decimal or a lid left loose, so labeling—whether GC data, storage instructions, or expiry dates—forms part of the safety net in crowded shelves.
Making It: Preparation in Practice
Methanesulfonic anhydride isn’t simply plucked off a shelf. The most direct preparative route relies on dehydrating methanesulfonic acid, sometimes through treatment with phosphorus pentoxide. This step strips away water with characteristic vigor, sending up enough heat to command respect for both operator and apparatus. In bigger batches, alternative dehydrating agents sometimes enter to tweak efficiency or mitigate cost, but the same fundamentals hold true across the board: keep moisture at bay, keep cleanup thorough, and keep monitoring throughout. Too casual a touch can generate impurities or, worse, runaway reactions that toss aside safety and waste resources. Every veteran chemist can recall at least one close call stemming from dehydration steps gone sideways.
Chemical Reactions and Modification: Where It Shines (and Where It Bites)
Methanesulfonic anhydride isn’t a wallflower once in the flask. Go-to uses center on sulfonylation—adding protective groups or activating alcohols, amines, and other nucleophilic sites for further reactions. Compared to thionyl chloride or acetic anhydride, it strolls in with selectivity but packs enough punch to leave a mark on sluggish substrates. It gives chemists a way to install methanesulfonyl (mesylate) groups, which commonly end up as leaving groups in substitution reactions or protection steps. There’s a bittersweet edge, though: careless mixing with nucleophiles leads to messes, multiple byproducts, or even gas evolution, so process control always looms large. Researchers continue exploring tweaks—dialing in purity, tailoring reaction parameters—to pull new tricks out of an already-bold molecule.
Multiple Names, One Molecule
Chemistry loves nicknames, often based on old habits or legacy patent filings. Methanesulfonic anhydride answers to a few: mesyl anhydride, MSAA, and, for the more formal crowd, disulfuric dimethane. Each name carries echoes of how and where chemists first started using it, bridging languages and formulas on labels across the world. This quilt of synonyms gives search engines and suppliers a hard time, but those who make a living routing complex orders or tracing literature love the redundancy.
Safety and Getting the Job Done Right
Walking into a lab, the first whiff of methanesulfonic anhydride sounds an alert. It bites the nose, chases people to fume hoods, and demands gloves and goggles at a minimum. The reactivity that makes it a favorite for activating steps doubles as a reminder—it burns skin, stings airways, and corrodes surfaces if allowed to linger. Best practice calls for airtight containers, eye wash stations, and a well-rehearsed plan for spills. Emergency procedures in many labs continue to stress the very basics: neutralize with care, segregate waste, never mix with water without a clear mind about exothermic reaction risk. In my experience, new students pick up respect for methanesulfonic anhydride fast, sometimes after a single singed glove or smoldering paper towel.
Why Use It: Application Areas Spanning Multiple Industries
What draws so many chemists and manufacturers to this anhydride isn’t just its ferocity but the specificity it offers in modern synthetic work. In pharmaceuticals, it gives access to high-purity intermediates, especially where gentle conditions keep delicate molecules happy. Electronics manufacturers call on it for sulfonating agents in specialty polymers or electronics-grade cleaning solutions. Fine chemicals and agrochemical industries branch out into large-scale syntheses, building off the high yield and reproducibility methanesulfonic anhydride can offer when process control doesn’t slip. Each group prizes the clean conversions, but in the back rooms, purchasing managers and safety officers carry a slightly different tune—they watch price volatility and hazard profiles with a weather eye, never forgetting the broader calculus of risk, reward, and regulation.
Where Curiosity Hits: Research and Development Efforts
Investigators continually push the envelope, asking hard questions about whether old methods remain best or if catalyst tweaks, green chemistry approaches, or continuous-flow techniques could boost both output and sustainability. Journals over the last decade began filling up with multi-step syntheses relying on methanesulfonic anhydride for a single, high-value modification. Enthusiasm runs highest in fields looking to build molecular complexity without dragging along old baggage like heavy-metal residues or environmentally persistent byproducts. Some labs chase process intensification; others want safer derivatives or milder alternatives. The climate for innovation stays lively, spurred by regulation, profit pressure, and a dogged curiosity to find out what else sulfonylating agents can accomplish in the hands of a determined team.
Peeling Back Layers: Toxicity and What It Means to Workers
Toxicological research into methanesulfonic anhydride reads like a cautionary tale. Acute exposure brings caustic reactions—skin burns, eye damage, respiratory irritation. Chronic effects remain an area where data drags behind usage, so prudent managers stress conservative limits on exposure. Animal studies highlight the strong corrosivity, but systemic toxicity ranks lower compared to some other industrial anhydrides, provided rigorous containment and ventilation cut off routes of accidental contact. The story repeats itself in many specialty chemicals: urgency to tap into unique reactivity comes face-to-face with responsibility to safeguard workers and the environment. Where gaps in data appear, precaution grabs the wheel, and regulatory agencies begin nudging for reporting, monitoring, and—where needed—substitution.
Eyes on Tomorrow: What the Future Holds
Chemicals don’t sit still. Future prospects for methanesulfonic anhydride hinge on cleaner syntheses, smarter containment, and a hunger for more sustainable, less hazardous processes across sectors. As green chemistry principles spread, researchers hunt for drop-in replacements or adjacent molecules with similar punch but lower environmental and health risks. Others focus on retooling production—drawing on renewable feedstocks or recycling process waste. Global supply chain changes and pressure from consumer advocacy groups have already begun tilting the balance toward transparency and traceability. No matter what, the years ahead promise both innovation and scrutiny, pulling together industry veterans, regulatory bodies, academic labs, and end users into ongoing conversations about balancing progress with protection.
A Hidden Player in Modern Chemistry
Most people probably haven’t heard of methanesulfonic anhydride, and I get it — it doesn’t carry a flashy name. For chemists, this stuff isn’t background noise. Methanesulfonic anhydride does the heavy lifting behind the scenes, powering reactions that touch everything from medicine to materials science.
Where It Matters Most
Step into the world of labs and you see how often things like methanesulfonic anhydride show up. I learned this the hard way during my grad school years, trying to synthesize new molecules for cancer research. You need chemicals that offer precise control. Methanesulfonic anhydride gives chemists that control. It works as an activator, helping transform ordinary groups on molecules into forms that react the way you need.
In pharmaceuticals, nobody wants to gamble on a reaction that gives you a messy yield. This chemical helps clean up the process, making selective transformations more predictable. The more reliable your toolbox, the faster you can design safer drugs. For me, the lesson wasn’t just about pushing science forward — it was about saving time, money, and headaches.
Moving Past the Lab Bench
Outside university labs, methanesulfonic anhydride touches a lot of industries indirectly. Making advanced plastics, coatings, and specialty chemicals all rely on it at some point in their development. That matters anywhere you see high-performance materials — think electronics or medical devices. If you want quality and durability, the right chemistry has to happen early in the process. That’s where this compound steps in.
Folks often forget how many steps it takes to get a new product on the shelf. Every one of those steps has its own risks and rewards. Small mistakes can mean big costs, both for companies and — if we’re talking medicine — for public health. Safe and consistent reagents like methanesulfonic anhydride bring some much-needed certainty.
Safety and Environmental Responsibility
Every time a new compound becomes popular in industry, questions about safety and environmental impact deserve more attention. Methanesulfonic anhydride isn’t a grocery store product; it can be hazardous without the right know-how. Over the years, I’ve seen lab accidents that happened because someone underestimated a chemical’s risk. Training, proper storage, and clear labeling make a world of difference.
It’s important to talk about how the manufacturing and disposal of chemicals shape the world outside the lab. Organizations now expect transparency about environmental footprints. Methanesulfonic anhydride, like many other specialty reagents, faces pressure to live up to stricter standards. Companies move toward using greener solvents, recycling spent materials, and investing in safer infrastructure. The work doesn’t end with invention — it continues with stewardship.
Paths Forward
Chemistry never stands still. New ideas and smarter manufacturing offer chances to improve not just efficiency, but also safety and sustainability. I look at methanesulfonic anhydride as a reminder that behind every finished product, there’s a line of unsung ingredients making it all possible. By staying informed and advocating for responsible practices, we make sure that progress leaves the world better off — both inside the lab and far beyond it.
What Is Methanesulfonic Anhydride?
Everyone who has spent time in a chemistry lab comes across formulas that look similar until a closer look reveals unique differences. Methanesulfonic anhydride, with its chemical formula (CH3SO2)2O, sits in that tricky space — not as famous as its cousin methanesulfonic acid, but no less important. Two methanesulfonyl groups linked through an oxygen atom give it this simple, yet powerful recipe: C2H6O5S2.
Why Should We Care?
Working with anhydrides opens up a door in synthetic chemistry. Methanesulfonic anhydride’s role goes way beyond black-and-white formulas. In organic chemistry classes, I saw peers react with hesitation around the word 'anhydride' because it signals reactivity and, often, some caution. This specific compound jumps into action as a dehydrating agent. Laboratories use it to activate alcohols, making them more reactive — critical when you’re making sulfonyl esters or setting up a protecting group on an alcohol.
The need for efficient, selective reagents drives up demand, especially when only a specific functional group should react. Sulfonyl anhydrides like this one help streamline reactions. Research communities rely on reproducibility and predictability, and knowing how to use a clean, reliable reagent matters enormously. When chemists get the outcome they expect, time and precious resources stay preserved.
Health and Environmental Considerations
This compound does raise eyebrows from a safety perspective. Methanesulfonic anhydride doesn’t have a pungent, familiar odor like acetic anhydride, but it reacts quickly in the presence of water or alcohols. Untrained hands can trigger fumes or burns. In real life, even a single slip — maybe a loose glove, maybe a forgotten fume hood — can turn a routine reaction into a minor emergency.
On the environmental front, spills and leaks ask for swift attention. Many accidental releases start with inadequate containment. I recall labs where routine checks caught a minor leak fast — no one likes paperwork, but catching problems early beats long cleanups every time. Neutralizing agents like sodium bicarbonate keep things under control, if they’re kept on hand and lab workers know their stuff.
Building a Responsible Use Culture
Solid fundamentals in chemical training make a difference each day. Experienced chemists who share firsthand stories of both close calls and successes help newer members recognize why lab protocols evolve. Over time, casual shortcuts get weeded out, and the focus turns toward clear processes. Proper goggles, secondary containment, and thorough training might seem redundant until the first unexpected splash or reaction cloud appears.
In many places, chemistry carries a serious responsibility. Beyond the formula, methanesulfonic anhydride reminds us that every step — from storage to scale-up — has consequences for safety and quality. Open communication lets teams address issues early and keeps a lab culture rooted in respect for both the work and the people doing it.
Moving Chemistry Forward
Better chemistry means using reliable information, smart tools, and strong habits. The chemical formula for methanesulfonic anhydride isn’t just a line in a textbook. It reflects a system where knowledge, attention to detail, and collaboration come together every day — not just to complete a reaction, but to build trust in how science gets done.
The Case for Respect in the Lab
Not every bottle on a lab shelf is equal. In my years of chemical research, I’ve learned the hard way that mishandling reagents sometimes gives you a lesson you don’t want. Methanesulfonic anhydride—MSA—looks pretty unremarkable in a glass vial. The real trouble starts with careless storage and handling. This chemical can mess up your lungs, your equipment, and even your project if you don’t use common sense and respect its properties.
Why Skimping on Storage Control Backfires
MSA reacts quickly with water. Fans of dry chemistry know even a humid lab bench can be the enemy. A genuine dry box or a dedicated desiccator keeps it all right. We kept ours in amber bottles with tightly screwed caps, sometimes double-sealed. Exposing this stuff to air never ends well. Even a quick dip in moisture can weaken a batch and raise the risk of corrosion or fuming. This is not just about protecting product quality—once anhydrides start breaking down, nasty by-products can waft into the workspace.
Personal Safety Gets Real
People sometimes treat chemical gloves and goggles as optional. MSA gives a nasty sting to the skin and can be worse on your eyes. In my lab, I saw plenty of folks grab pipettes with bare hands out of convenience, only to wind up in the sink washing off a splash. A full sleeve, serious gloves, and goggles become non-negotiable. For anyone transferring MSA from one container to another, a fume hood saves your lungs from those sharp vapors. I don’t know a single careful chemist who’s gone without one and felt safe.
Keeping the Workspace Honest
MSA erodes metals, melts plastic labware, and will chew through unlabeled containers given enough time. If it leaks, the mess attracts more moisture and starts a chain reaction. I always kept a clear log of where the chemical sat in the lab, with hazard signs front and center. No one wants to find out a bottle oozed through a shelf overnight.
Disposal and Emergency Plans
Spills aren’t a “maybe” in chemistry; they’re a “when.” Neutralizing agents sit within arm’s reach, and everyone needs to know the basics of cleanup. Flushing spills with lots of water makes things worse. Dry material and careful neutralization remain safer. I would never trust a rumor or shortcut. Consulting trusted safety data and regulatory guides makes all the difference when moments count.
Learning From Those Who Know More
The best labs swap stories as often as protocols. People who have faced up to chemical danger and walked away smarter give the most trustworthy advice. I take peer-reviewed guidance, not hearsay, and combine it with firsthand warnings. In the long run, giving MSA full respect, from the moment it enters the door to the way it exits the waste bin, means you can focus on research instead of damage control.
Smarts Before Speed
No deadline justifies shortcuts on safety. The best science speaks for itself, but only when its practitioners make it home healthy. Storing and working with MSA safely draws on experience, good habits, and solid information—steps that pay back every time you open the storage cabinet with confidence.
Looking Past the Chemical Name
Most folks haven’t heard much about methanesulfonic anhydride unless they spend time in research labs. Still, chemicals like this shape many essentials around us—drugs, new materials, advanced electronics. Plenty of promise, but you only see the flipside if you’ve ever handled it up close, suited up, and respected the sharp warning labels.
The Risks Hiding in Plain Sight
Pick up the container, and it’s clear this isn’t something to treat casually. Methanesulfonic anhydride earns its “corrosive” label for a reason. Any spill can burn right through latex gloves, leave your skin blistered, your eyes in trouble. Smelling it? Not a bright idea. Fumes sting the nose, rough up your throat, and could leave your lungs angry for hours or days. Many solvents or acids give off a crisp signal, but this compound often works quietly. That makes a moment of distraction risky.
Having worked with organosulfur chemicals, the lesson hits quickly: one misplaced beaker or carelessly sealed bottle can mean hours cleaning up and weeks before everyone relaxes. There’s always a story. Someone in my old lab dropped a tiny vial; we all scrambled for eyewash. His safety goggles bought precious seconds. Speed matters, with first aid kits and showers only steps away—it all saves skin, eyesight, and careers.
Fires, Explosions, and the Chain Reaction Factor
Many see the word “anhydride” and connect it to dehydration or synthesis. That technical bit also means fire and sudden reactions lurk in the background. Combine methanesulfonic anhydride with water? It releases heat, sprays acid, and fills the air with fumes. Add in some organic solvents or incompatible chemicals, and you can get run-away reactions that end in fireballs. Anyone in the lab game long enough hears these tales, and most of us double-check every label for this reason.
What Care Looks Like—Practical, Not Pretty
Handling such a compound means planning every step. It starts with protective gear—chemical splash goggles, sturdy nitrile gloves that outlast latex, and a thick apron or lab coat. Hoods aren’t for show; they trap fumes before you even notice them. Every single transfer—pour, dilution, disposal—happens inside the hood with extra absorbent pads at hand for drips.
Safety data sheets don’t live in a dusty drawer. Experienced workers read them before the first experiment. Kits for spills sit nearby, not across the building. These habits spread from skilled techs and older researchers to the new faces. Labs post bold warning signs and train new members until every routine feels almost boring. But boredom beats the panic of a spill or fire.
Why Respect Matters More than Risk
The brightest breakthroughs sometimes start with risky chemicals like methanesulfonic anhydride. Smart teams treat the danger with up-front honesty and real rules—never shortcuts or improvising with protection. Eye washes, ventilation, gloves, and simple drills all keep pain, and years of hard work, from going up in literal smoke.
No one wants another tale of a rushed job turning painful. Culture, training, and a healthy respect for what could go wrong build labs where discovery happens safely—and folks get to go home unscathed at the end of the day.
What Happens to Methanesulfonic Anhydride in Water and Other Solvents?
Ask any chemist about Methanesulfonic Anhydride, and you’ll probably get a straight-face response: it reacts in water, fast. This compound, popular among synthetic organic chemists, carries plenty of potential for transformations. But that advantage comes bundled with a challenge: its wild behavior with water and some other solvents. Over the years, working in academic and industrial labs, I’ve seen more flasks bubble and cloud suddenly than I’d like to admit. That’s what you get when you try to mix an anhydride like this with water. Instead of dissolving, it usually breaks apart, forming methanesulfonic acid almost immediately. There just isn’t much chance to use water as a solvent here.
Several factors come into play. Methanesulfonic Anhydride is made to take water apart, not blend with it. The reaction between this anhydride and water isn’t subtle. The water donates oxygen, cleaving the anhydride bond and dumping the reaction product into your flask. The process releases heat and makes experiments unpredictable. I’ve learned to steer clear of water or alcohols as solvents unless the goal centers on hydrolysis.
Organic Solvents: Where the Story Changes
Unlike with water, Methanesulfonic Anhydride plays much nicer with certain organic solvents. In dichloromethane, acetonitrile, or chloroform, it shows proper solubility, making life easier for anyone setting up a reaction. These solvents keep the compound stable long enough for it to do its job—dehydrations, activations, or functional group transformations—before breaking down. On the bench, that means a chemist can add their reagents without worrying about runaway side reactions starting before the main event.
Why focus on solvents like this? The safety and outcome of a chemical synthesis change with the solvent used. The exasperation of watching a precious batch fizz away comes from failing to match reactivity and solvent choices. In a professional setting, that can cost both time and budget. A clear understanding helps bring predictability, fewer accidents, and better reproducibility.
Why Does Solubility Matter for More than Lab Work?
Chemistry doesn’t stop at cleanliness or yield. Companies and academics alike track each step for environmental safety, worker health, and compliance. In my lab, efforts to scale up reactions once ran into roadblocks because residues broached waste handling rules. Anhydrides that react violently with water can generate heat and acids, which push up risks for anyone transporting, storing, or disposing of them. Choosing a safe solvent limits unexpected toxic or corrosive byproducts. That attention to detail can help avoid expensive cleanup efforts and keep people safe on the job.
Green chemistry also benefits from knowing where your compound will and won’t dissolve or react. Using non-water solvents often means you can recover and recycle them. The industry has begun nudging toward friendlier solvents and processes. Studies point to gains in yield, efficiency, and environmental footprint by looking at how anhydrides, like this one, interact with solvents. The goal is safer labs, milder conditions, and less environmental hassle down the line.
Making Good Choices in Chemistry
At the bench or in the plant, it pays off to use the right solvent. Knowledge about Methanesulfonic Anhydride dissolving—or breaking apart—plays straight into safer outcomes and reliable results. In the push for better, cleaner chemistry, respecting these quirks shapes everything from the size of your flask to how you handle waste.