Methyl Chlorodifluoroacetate: Past, Present, and Future in Chemistry
Tracing the Roots
Methyl chlorodifluoroacetate first caught the attention of chemists in the mid-20th century, arriving on the scene as research in organofluorine chemistry picked up steam. Looking back, it’s clear how this compound’s unique blend of properties took on greater importance as industry and academia both searched for building blocks with distinct reactivity. Historical records in old scientific journals point to its early use, especially as synthetic chemists discovered its potential in generating fluoroalkylated molecules. This niche pursuit paved the way for innovations in agrochemicals and pharmaceuticals, where the demand for specialized fluorinated agents kept rising. Over the years, the development of methyl chlorodifluoroacetate paralleled the growth of fluorine chemistry—a collaborative journey between laboratory curiosity and industrial need.
What Sets It Apart
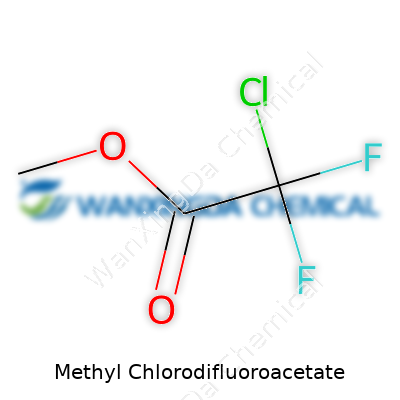
Methyl chlorodifluoroacetate stands out thanks to its specific blend of elements: chlorine, fluorine, and a methyl ester group. Each of these atoms brings something distinct to the table. The two fluorine atoms, for instance, lend the molecule a level of chemical stability and influence its behavior in both biological and synthetic settings. That stability isn’t just a talking point in chemistry lectures; it’s something that dictates how researchers design their experiments and how manufacturers evaluate the risks and rewards of integrating new chemicals into their processes. The ester group, on the other hand, opens up routes for transformation through familiar chemical reactions, such as hydrolysis and transesterification, making this molecule remarkably versatile.
Getting a Closer Look: Physical and Chemical Traits
In the lab, methyl chlorodifluoroacetate presents as a colorless to pale liquid with a notable, sharp odor. You’ll find it boils at a temperature much higher than many of its non-fluorinated cousins. That’s not just trivia; it directly affects how the compound gets stored and handled. Its moderate polarity reflects its mixed composition of fluorine and chlorine. A reasonable vapor pressure adds another layer to its handling risks, demanding careful control during syntheses. Through experience, I’ve seen that its reactivity stands out when compared to basic methyl esters, mostly thanks to the electron-withdrawing activity of the fluorine and chlorine atoms. These properties don’t just define its laboratory identity —they guide its storage protocols and its reputation among chemists.
Precision in Labeling and Technical Details
With a molecular formula of C3H3ClF2O2 and a molar mass in the low hundreds of grams per mole, methyl chlorodifluoroacetate calls for accurate representation on labels. Each batch comes with a purity claim, usually expressed as a percentage, that influences every downstream application. Labs checking for compliance rely on these numbers, especially since even small impurities can impact outcomes in sensitive syntheses. Beyond the basic numbers, labeling often includes hazard warnings relating to its reactivity, volatility, and potential health impacts—critical information for every chemist and technician who interacts with the material. While working with this compound during a project on selective fluorination, I learned firsthand that missing or ambiguous details on a reagent bottle can grind a project to a halt.
Crafting the Compound: Preparation in Practice
Producing methyl chlorodifluoroacetate typically involves the esterification of chlorodifluoroacetic acid with methanol, using a standard acid-catalyzed approach. The presence of strong acids and the volatility of starting materials mean the whole process needs careful ventilation and temperature controls. Yields vary based on reaction conditions, and small shifts in moisture levels can sabotage a batch. Some manufacturers opt for more exotic techniques under anhydrous conditions to push reaction rates, but regardless of scale, quality control remains a defining factor. Alternative routes involve the halogen exchange of methyl difluoroacetate, but these require even closer attention to hazards because of the release of noxious byproducts. Each method reflects the broader challenge chemists face when wrangling with highly reactive and sometimes unforgiving molecules.
Exploring the Chemistry—Reactions and Modifications
Methyl chlorodifluoroacetate displays a reactive profile shaped by its alpha-chloro and alpha,alpha-difluoro groups. In my hands-on experience, nucleophilic substitutions at the alpha position can generate a wide variety of more complex derivatives. Skilled chemists can exploit this for introducing new groups—transformations that often feed directly into medicinal and agrochemical development. The ester group also offers classic hydrolysis or reduction pathways, which expand its versatility. Unique reactions emerge from the interaction between fluorine's electronegativity and the reactivity of the chlorine atom, bringing synthetic possibilities that few other reagents provide. I’ve seen research teams push for milder, more selective methods to make the most of this molecule while controlling potential hazards.
What’s in a Name? Synonyms and Labeling Challenges
Depending on the catalog and the region, methyl chlorodifluoroacetate might appear under names like methyl 2-chloro-2,2-difluoroacetate or just CF2ClCOOCH3. Each synonym reflects a slightly different approach to nomenclature, but the underlying compound remains the same. Navigating these aliases can create confusion, especially in global research settings where shipments cross borders. Mislabeling isn’t just an inconvenience; accidents from mistaken identity cost time and resources. Standardized naming conventions strongly influence both safety and research productivity.
Keeping People Safe: Standards and Best Practices
Methyl chlorodifluoroacetate poses challenges rooted in its volatility, acute toxicity, and potential to generate hazardous gases under fire or decomposition. In the lab, full PPE—nitrile gloves, splash-resistant goggles, and lab coats—stay non-negotiable. Proper handling means working in well-ventilated fume hoods, with immediate access to spill kits and neutralizing agents. Local regulations require detailed documentation of storage conditions, emphasizing cool, dry environments and tight-sealing containers. Training for staff ensures everyone knows the risks of inhalation or skin exposure. I recall a case where inadequate ventilation during scale-up forced us to halt work mid-run, reinforcing how easy it is to underestimate volatility and irritation potential. Advances in engineering controls and real-time monitoring systems offer much-needed reassurance, yet manual diligence remains as crucial as regulatory compliance.
Where It Makes a Difference: Applications in Research and Industry
Methyl chlorodifluoroacetate finds its home primarily in pharmaceutical research, agrochemical synthesis, and as an intermediate for specialty fluorinated compounds. Medicinal chemists value it for making molecules with enhanced metabolic stability, properties that influence drug behavior in the body. In agriculture, certain crop protection agents draw their effectiveness from this class of compounds. I’ve seen the compound serve as a valuable synthetic handle, leading to analogs with activities that standard chemistry struggles to match. Beyond these fields, academic labs use it as a test case to understand the effects of strongly electron-withdrawing groups on carbon skeletons. Its influence goes beyond sheer volume; even small-scale use can shape the direction of new molecules and treatment paradigms.
Pushing the Envelope: Research, Development, and Toxicity Studies
Active research keeps pushing new ways to use methyl chlorodifluoroacetate more safely and efficiently. Studies increasingly focus on reducing both the environmental impact and toxicological risks of fluorinated intermediates. Data from recent animal studies indicate moderate acute toxicity, with particular concern for respiratory and gastrointestinal responses. Chronic exposure data remain limited, which justifies ongoing study before any broader regulatory decisions land. Leading journals report a steady stream of new catalytic methods and reaction mechanisms designed to tame the molecule’s reactivity. These aren’t academic exercises—labs balance discovery with responsibility, realizing how persistent organic pollutants can affect water supplies and air quality. The chemistry community responds by designing smarter syntheses and developing rapid detection methods for leaks and spills.
Looking Ahead: Future Prospects
Over the next decade, methyl chlorodifluoroacetate looks set to remain relevant, especially as demand for fluorinated materials grows in medicine and technology. New techniques relying on sustainable chemistry may carve out safer, cleaner ways to produce and use the compound. As regulatory landscapes tighten, companies and labs willing to innovate will stay ahead—pursuing greener reagents, more selective reactions, and robust waste treatment. The combination of nuanced chemistry, strict safety protocols, and strategic research sets the stage for the next wave of breakthroughs, where methyl chlorodifluoroacetate stands not as a relic, but as a stepping stone toward smarter, more responsible science.
Methyl chlorodifluoroacetate doesn’t show up in dinner table conversations, but people who care about the chemicals shaping modern life should pay some attention to it. In my experience working with science writers and talking to researchers in the field, this colorless liquid holds a solid spot in the world of chemical manufacturing, especially in specialized industrial labs. The substance pops up most often as a building block for other molecules. If you’ve ever wondered how new materials, certain pharmaceuticals, or crop protection solutions get made, it takes many small, reactive chemicals like this one to create them.
The Backbone of Fluorochemical Synthesis
Methyl chlorodifluoroacetate supports the production of fluorinated compounds. Chemists favor fluorine because it can make things stronger, safer, longer-lasting, or better at sliding through challenging environments. In real terms, this molecule acts like a handy puzzle piece for constructing much more complex chemicals. Drug makers tap into it when they want certain parts of a medicine to resist breaking down inside the body. Agrochemical innovators blend it into their projects to fine-tune performance out in the field. Even makers of specialty plastics look to chemistry like this when aiming for the right mix of flexibility and durability.
There’s a reason for the surge in all things fluorinated. Think about anti-stick cookware, high-performance sports gear, or medical imaging technology. The durability and versatility of these materials come back to strong carbon-fluorine bonds, and methyl chlorodifluoroacetate offers a relatively straightforward way to introduce those bonds into new compounds. Data from specialized chemical markets show a steady need for this molecule, especially in regions with advanced pharmaceutical and electronics industries.
Safety, Environment, and Responsibility
Handling methyl chlorodifluoroacetate takes respect. Talking to chemists, I hear stories about the strict protocols in place for working with any chlorinated or fluorinated compounds. Exposure risks make careful controls essential. The substance brings risks for both those who handle it and the environment. Its volatility and reactivity demand gloves, proper air systems, and storage rules.
Reviews from government agencies, such as the Environmental Protection Agency, point out that these compounds, if left unchecked, have the potential to cause harm if improperly released. Small mistakes multiply in large-scale processes, so companies invest a fair amount into safety equipment and continuous monitoring. People often ask, “Why not just avoid such tricky chemicals?” The reality is, without ingredients like this, progress in important sectors would grind to a halt.
Ethics and the Road Ahead
With growing scrutiny over persistent chemicals, there’s pressure on manufacturers to make sure every gram counts, wasted as little as possible, and recovered whenever feasible. I’ve seen examples where forward-looking labs tweak processes to capture and reuse leftover materials, cut down on waste, and improve worker safety. Collaboration across industry and science is driving smarter chemistry — methods that make the most of reactive molecules like methyl chlorodifluoroacetate without letting hazards spill beyond the lab. That’s a lesson worth remembering for any technology, not just this one.
Pay Attention to the Real Risks
Working with chemicals in any lab demands respect, but methyl chlorodifluoroacetate deserves a healthy dose of caution. As someone who’s spent plenty of time around unpredictable solvents and reactive compounds, I can tell you this: it only takes one moment of carelessness to change everything. A splash, a spill, an accidental whiff—each can have you dialing for help.
Real Hazards, Not Abstract Warnings
This chemical isn’t a mild irritant. Vapors can annoy your lungs, and the liquid burns skin on contact. Getting this stuff in your eyes? That leads to some real, lasting trouble. Accidental releases can contaminate workspaces for days—air, benches, gloves, and doorknobs. I once watched a careless spill on a balance trickle and etch a permanent reminder onto the bench top. Trust me, that wasn’t a lesson anyone wanted to repeat.
Personal Protection Isn’t Optional
No smart researcher skips personal protective equipment. Genuine safety starts with thick nitrile gloves, full goggles, and a chemical-resistant lab coat. If you take shortcuts, expect consequences. These aren’t empty rules meant to slow work down—it’s common sense collected from years of others’ mistakes. When you know gloves break down fast under some chemicals, you double-up. If standard safety glasses leave exposed skin, full face shields finish the job. Less coverage means more risk.
Ventilation Isn’t Just a Box to Check
Every chemistry worker knows the value of proper ventilation. Fume hoods aren’t suggestions. People sometimes prop up the sash to grab a flask or save time, thinking the risk is smaller than the inconvenience. Real world experience shows otherwise—fume hood airflows fail, vapors leak, and before you know it, you’re coughing, eyes burning. Any chemical this volatile deserves full airflow containment. The slight hassle beats inhaling dangerous fumes every time.
Keep Clean, Think Ahead
Good habits in the lab always pay back. Cleaning benches and equipment after work, keeping extra gloves on hand, and labeling containers keeps disasters small. I saw an unmarked beaker get punted across a bench because someone assumed it was water. It wasn’t. A small accident got larger before anyone realized the mistake. Marking, storing, and segregating reactives does far more to prevent emergencies than long technical documents.
Have a Plan—Before Trouble Shows Up
Every lab regular remembers their first emergency. Whether it’s a chemical splashed onto a wrist, or fumes blowing from a flask, the difference between panic and calm lies in muscle memory. Know the location of eyewash stations. Practice using safety showers. Have proper waste containers nearby. Team up with coworkers—two sets of hands and eyes work better than one. You’ll act faster together, and mistakes can’t hide as easily. Relying on luck or improvisation isn’t a plan—it’s a gamble.
Solutions Rooted in Reality
Improving safety means sharing experiences. Newcomers benefit from honest stories about accidents, not just dry safety drills. Openly talking through near-misses encourages smarter habits. Working conditions improve when supervisors don’t rush people or ignore faulty equipment. Investment in safety equipment, up-to-date training, and fostering a workplace where refusing unsafe work is respected—these make the biggest difference.
If you run a lab or just work in one, the real takeaway is simple: Don’t get complacent around methyl chlorodifluoroacetate. The best researchers value safety as much as skill, and for good reason. No experiment is more important than your well-being.
Science Behind the Formula
Chemistry shapes our daily lives in more ways than we realize, and compounds like methyl chlorodifluoroacetate showcase just how much substance lies beneath a chemical name. At its core, methyl chlorodifluoroacetate lays claim to the formula C3H3ClF2O2. The name packs a lot: “methyl” tips off a simple carbon group, “chlorodifluoro” points to the presence of both chlorine and two fluorine atoms, and the “acetate” backbone hints at its carboxylic acid heritage. Those of us who’ve spent late nights around lab benches know the structure isn’t just jargon: one carbon chain, a central carbon linked to both a chlorine and two fluorines, finished off with an ester group. It sounds textbook, but it means this molecule packs both reactivity and stability—qualities prized by synthetic chemists and material scientists alike.
Industrial and Environmental Significance
Industries prize methyl chlorodifluoroacetate as a building block for making complex molecules. Because the two fluorines and single chlorine shift electron density, this ester acts as a fine starting point for engineers working in pharmaceuticals or developing specialty polymers. The electronegative power of those halogens causes the molecule to react in unique ways, allowing for clever chemistry that might reduce the number of reaction steps in a process. Cutting down steps doesn’t just save time in a commercial lab; it slashes waste and brings down operational costs.
These days, fluorine-containing chemicals stir debate. Some folks get uneasy about persistent fluorinated compounds, especially those that don’t break down in the environment. Methyl chlorodifluoroacetate doesn’t belong in the same league as notorious PFAS, but responsible handling should anchor any lab that works with it. That means good ventilation, well-trained staff, and mindful waste processing. Writing from a bench chemist’s perspective, proper stewardship protects everyone, from the people making the material to the communities downstream.
Innovation and Safe Use
Researchers are finding new ways to put compounds like methyl chlorodifluoroacetate to work. Medicinal chemists might use it for quick introduction of fluorine, giving new drug molecules longer shelf lives or improved absorption rates. The food industry, though less likely to deploy this exact chemical, benefits from related fluorinated esters for making packaging both safer and more durable.
Harnessing the upside while reducing any downside comes down to the choices we make in the lab and the policies we follow on a larger scale. Just as in cooking, small changes in the recipe can lead to big differences. Recycling solvents, using greener synthesis methods, and setting proper exposure limits can all make life safer for workers and the environment. The technology exists to do better. The choice always comes down to using what we already know, standing behind clear regulations, and investing in better systems.
Looking Forward
People outside of chemistry may never hear about methyl chlorodifluoroacetate, but the ingredients behind the scenes shape the products and medicines we depend on. Understanding these molecules with care for both innovation and impact lays the groundwork for a safer, smarter tomorrow.
Mistakes Stick with Chemicals Like This
As someone who has worked in industrial labs and spent more time than I care to admit double-checking chemical cabinets, I've seen what happens when people take shortcuts. Methyl Chlorodifluoroacetate is not the sort of compound that forgives carelessness. Exposure to its fumes can hit your respiratory system hard, cause skin problems, and even mess with your eyes. The last place you want surprises is in chemical storage, and that holds double for this one.
Space and Conditions Matter Much More Than Labels
Methyl Chlorodifluoroacetate belongs strictly in a cool, dry, and well-ventilated spot. Forget about shoving it near heat sources or windows. Direct sunlight ramps up the risk of decomposition, which in a worst-case scenario means toxic gases wafting through your facility. I’ve seen folks try to save space by packing things close together on a shelf, but with chemicals like this, crowding isn’t just inconvenient, it’s dangerous. Adequate spacing can stop cross-contamination or chain reactions if there’s a spill or leak.
Materials Make a Difference
Glass, certain plastics, or coated steel usually offer reliable protection for chemicals prone to reaction. Plain metal or porous containers don’t cut it here. I’ve watched a colleague once pour off a small portion into a non-standard plastic flask, and in less than a week, that flask distorted, leaking fumes into our storeroom. Mistakes like that stick in your mind for a reason. Dedicated containers, sealed tight, and regularly checked for any signs of corrosion or wear, go a long way in preventing accidents.
Isolation Isn’t Just a COVID Buzzword
Strong acids, strong bases, and water-reactive or oxidizing agents have no business sharing a shelf with Methyl Chlorodifluoroacetate. Chemical compatibility charts aren’t just paperwork — they keep people safe. In shared storage spaces, maintaining sharp boundaries reduces the risk of violent reactions if two incompatible substances ever meet. Over the years, I’ve watched labs treat these rules with varying levels of respect, and the ones who don’t pay for it in lost chemical stock and, worse, lost equipment or injuries.
Labels and Real-World Awareness
Handwritten labels fade. Obscure abbreviations trip people up. Clear, permanent, precise labeling keeps everyone on the same page, including the person who isn’t familiar with your organizational quirks. This helps during emergencies or routine audits alike. In the event of a spill or exposure, there’s no time to guess what you’re dealing with.
Solutions Start with Training and Routine
The labs that avoid close calls train every new staff member to handle and store hazardous chemicals as if their health depends on it — because it does. Regular reviews of storage conditions and keeping up with inventory checklists catch small problems before they grow. National and international regulations exist for a reason. Following them doesn’t just build trust with regulatory bodies, it builds trust within your team.
Change Grows from Experience
Every incident I’ve seen involving Methyl Chlorodifluoroacetate could have been prevented by a mix of respect for the chemical and routine checks. It’s not enough to know the risks in theory — treating storage as a living responsibility creates a work environment that’s safer for everyone. In my experience, most “close calls” are a wake-up call that gets people to take the rules seriously. An unassuming bottle in a storeroom can shape the safety culture of an entire organization, for better or worse.
The Hazards Lurking Behind the Chemistry
Some chemicals pop up in conversation for their usefulness in industry or research, not because people want to chat about them at dinner. Methyl chlorodifluoroacetate falls into that bucket. On the surface, it sounds complicated, almost like something out of a college chemistry lab. Peel back the jargon, though, and you see a mix of carbon, hydrogen, chlorine, fluorine, and oxygen snugged together in a way that makes chemists pay attention.
I’ve seen how industries chase efficiency and profit, sometimes sacrificing foresight. This is a molecule that attracts interest for its reactions, but what gets mixed in almost always comes with trade-offs. The presence of both chlorine and fluorine in this compound puts it on a watch list for environmental and health groups, not just because of what it does in a lab, but what it leaves behind.
Looking for Trouble: Health and Environmental Impact
Picture yourself handling this material. Most of us won’t ever come close, but factory workers, researchers, and warehouse operators don’t have the luxury of distance. Like a lot of organic chemicals that blend halogens into the mix, methyl chlorodifluoroacetate brings risks. Inhalation or skin contact can cause irritation, and repeated exposure can turn into something much worse — respiratory issues, headaches, or more hidden long-term risks.
Stories from chemical plants stick with me. Sometimes, it only takes a cracked seal or missing glove for a trip to the emergency room. Chlorinated and fluorinated compounds punch above their weight when it comes to toxicity because our bodies neither process them easily nor toss them aside.
Step outside the factory walls, and the story keeps rolling. Chemicals with chlorine and fluorine atoms show a stubborn streak — they don’t just break down on a sunny day. In soil or water, they linger. That means downstream problems. Think bioaccumulation in fish, poison moving up food chains, water supplies catching residues that no one invited. Incidents with similar compounds have pointed to cancer links, disruption of hormone systems, and even ozone layer damage when airborne.
Don’t Just Panic, Get Proactive
Avoiding a chemical doesn’t always feel realistic. Some industrial processes rely on compounds like this for good reason — or at least, for good economics. The key is real risk management, not just paperwork that gathers dust. Engineering controls, strict ventilation, high-quality gloves, and honest worker training beat slogans every time. Regulators have laid out rules for storage and disposal for related chemicals; skirting them settles nowhere except in tomorrow’s headlines.
Communities living near manufacturing sites shouldn’t lose sleep, but they deserve transparency — real information, not just odd-smelling smoke and a siren in the night. Strong environmental monitoring, backed by public reporting, helps catch leaks before trouble builds. Labs should always keep a keen eye on alternatives with fewer halogens. Green chemistry isn’t a trend; it’s a necessity if the goal is to keep groundwater, air, and bodies clean five or fifteen years from now.
Companies with experience in chemical handling can help set the pace. Open reporting, paired with third-party audits, grows public trust. It’s not rocket science — it’s just facing up to risks instead of hiding behind legal walls. Until safer substitutes hit the mainstream, putting health and environment first never goes out of style.