Methyl Difluoroacetate: Tracing Its Journey, Importance, and Future in Chemistry
Historical Development
Curiosity often shapes the path of chemical discovery, and methyl difluoroacetate offers a clear example. Early synthetic chemists, chasing the promise of new reactivity patterns in the mid-20th century, started exploring small organofluorine compounds. The emergence of techniques for selective fluorination was a game-changer. Methyl difluoroacetate didn’t just pop into labs one day; it followed a wave of rising demand for more precise building blocks in medicinal and agrochemical research. Laboratories in Europe and North America tried to harness the unique role of fluorine in fine-tuning chemical properties. This compound became part of a family of reagents that allowed for careful introduction of difluoromethyl or difluoromethylene groups into molecules, a smart move to influence bioactivity and stability. The evolution of its use mirrors broader shifts in synthetic strategy—only as reliable fluorination grew feasible did methyl difluoroacetate become accessible beyond specialist facilities.
Product Overview
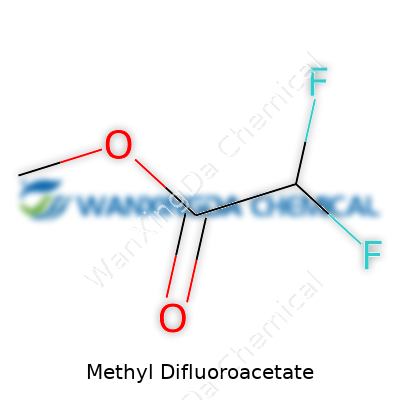
Among small esters, methyl difluoroacetate stands out. Its role isn’t just about being a chemical curiosity; it serves a valuable function both as an intermediate and as a raw material for more complex synthesis. Chemists lean on its active difluoromethyl group to influence how molecules behave in pharmaceutical research. Structurally, it brings together a methyl ester moiety with the power of two highly electronegative fluorine atoms on a central acetyl group, creating a profile that draws both researchers and industrial chemists for its reactivity. Its adoption grew rapidly in the late twentieth century as demand for fluoroorganics took off—anything from advanced drugs to new agrochemicals began to require such intermediates.
Physical & Chemical Properties
Methyl difluoroacetate presents an interesting mix of features: a clear, often colorless liquid at room temperature, its moderate volatility means it isn’t the easiest to handle but isn’t especially challenging either. Boiling points tend to hover in the range typical of simple methyl esters, though the presence of two fluorine atoms bumps up both its chemical stability and its resistance to biological breakdown. The distinctive odor, sharp and chemical, often alerts to its presence before measurements confirm it. Its high electronegativity, owing to the fluorines, dramatically alter neighboring reactivity—making the acetyl site far more prone to selectivity in reactions, a blessing and a curse depending on the synthetic aim. The compound dissolves well in polar organic solvents, which makes it approachable for many lab techniques.
Technical Specifications & Labeling
Industry has had to adopt clear, no-nonsense standards for handling and labeling such chemicals. Chemists pay attention to purity, water content, and absence of close isomers that might confuse analysis. Labeling regulations ask for unmistakable hazard identifications, warnings about eye and respiratory irritation, and handling protocols designed to protect against accidental skin contact or inhalation. Prepping reliable batches depends on quality reagents and well-maintained equipment; anything less, and researchers learn quickly that unexpected contaminants can throw synthetic plans off track. Documentation tends to spell out boiling range, storage temperatures, and clear guidance on container compatibility because long-term storage finds methyl difluoroacetate sensitive to basic conditions and aggressive nucleophiles.
Preparation Method
Modern labs use several routes to prepare methyl difluoroacetate, each with distinct trade-offs. The most common route involves the esterification of difluoroacetic acid with methanol, usually in the presence of an acid catalyst like sulfuric acid. Fluorination of methyl acetate using specialized reagents—such as elemental fluorine or more selective fluorinating agents—appeared in literature as clever chemists worked around harsh or unpredictable conditions. Practical synthesis involves careful control of temperature and reaction times because over-fluorination leads to nasty byproducts, and yields suffer when side reactions aren’t tightly managed. Advances in reactor design, improved separation techniques, and a better understanding of fluorine chemistry allowed for safer and more scalable preparation in the past two decades.
Chemical Reactions & Modifications
Anyone working with methyl difluoroacetate soon discovers the power of that difluoromethyl group—in nucleophilic substitution, it’s much less reactive than non-fluorinated esters, but, under the right conditions, electrophilic partners can exploit the electron-withdrawing effect of the fluorines. This enables efficient introduction of difluoromethyl moieties onto new carbon frameworks, key for pharmaceutical and agrochemical work. Reduction reactions—like those with lithium aluminum hydride—lead to difluoroethanol derivatives, while other transformations take advantage of the acetate’s ester function, making hydrolysis and transesterification both viable and manageable. The chemistry is not only interesting on paper; it signals broader potential for launching new classes of biologically relevant molecules if clever modifications are applied.
Synonyms & Product Names
As with many chemicals, methyl difluoroacetate appears under various names depending on context—industry sources might list it as methyl 2,2-difluoroacetate or simply as ‘MFA’. Chemical catalogs from Europe sometimes flip around the nomenclature, emphasizing either the methyl ester or the difluoroacetate backbone. Old research papers occasionally refer to related fluoroacetate esters, but current usage favors the clear IUPAC name to avoid confusion in regulatory circles and databases.
Safety & Operational Standards
Safety isn’t an afterthought with methyl difluoroacetate. Its volatility means spills and evaporation pose a daily risk, and experience teaches that gloves, goggles, and good ventilation can’t be skipped. The compound doesn’t have the acute toxicity of some organofluorines—with proper handling, dangers stay manageable—but users still face strong irritant potential if vapor or liquid makes contact with skin or eyes. I once saw a flammable liquid spill escalate into a fire risk when lab partners were slow to identify vapor buildup near hot plates; it reinforced the importance of safe work practices and regular air monitoring. Laboratory protocols frequently call for strict documentation, clearly labeled secondary containment, and well-rehearsed emergency responses. Containers suited for esters and resistant to acidic or basic degradation extend shelf life and lower risk. These aren’t just bureaucratic hurdles—they come from hard-won lessons in chemical safety.
Application Area
Medicinal chemistry uses methyl difluoroacetate as a building block for designing molecules with enhanced metabolic stability. Fluorine substitution often leads to slower drug degradation in humans, which can improve bioavailability and dosing schedules. Agrochemical developers mix it into schemes for crafting pesticides and herbicides with longer environmental lifetimes or altered reactivity profiles. It also finds use in materials science, where researchers engineer surface treatments and polymer modifications to resist solvents or to adjust wettability. In each of these domains, its reliable behavior and distinct chemical profile make it a favored starting material for inventive work. Real-world experience shows the impact—better drugs, sturdier coatings, and more resilient agrochemicals trace back to this simple molecule.
Research & Development
Research with methyl difluoroacetate keeps adapting to changing needs. As demand for fluorinated pharmaceuticals jumped in the last decade, researchers at academic and corporate labs put more effort into discovering routes that reduce hazardous byproducts and improve selectivity. Innovations in catalysis, especially with transition metals, gave rise to more controlled difluoromethylations that use less toxic reagents. The push for green chemistry also drives efforts to recycle solvents or replace persistent fluorinated waste with more biodegradable side streams. Ongoing studies probe how changing the ester group alters drug metabolism or environmental fate, guiding smarter design of new molecules. Insights gained with methyl difluoroacetate feed into both basic science and the next generation of applied solutions.
Toxicity Research
Tox studies rarely offer easy answers, especially with organofluorines. Chronic exposure data shows relatively mild toxicity compared to older, unregulated fluorinated compounds, but vigilance remains necessary. Short-term inhalation or skin exposure tends to irritate and cause discomfort, prompting strict guidelines for lab work. Long-term environmental impact remains an open question because fluorinated breakdown products often resist biodegradation. Studies on aquatic organisms suggest low immediate lethality, but the greater concern is bioaccumulation—persistent chemicals that build up in food chains. Regulators and researchers keep revisiting these questions, updating practices to prevent environmental release and protect workers. Building safe stewardship around methyl difluoroacetate pays dividends in sustainable research culture.
Future Prospects
Looking ahead, methyl difluoroacetate’s story is far from over. Fluorine’s role in chemistry keeps expanding as scientists look for ever more sophisticated ways to tweak behavior in biologically active molecules and advanced materials. Efficient, lower-impact synthesis will probably open new doors for its use, especially as industries shift toward greener and safer processes. Researchers hope to design drugs and crop treatments that retain fluorinated benefits without posing risk of environmental persistence. Ongoing advances in catalysis, analytical monitoring, and waste treatment promise safer, more accessible paths from lab to full-scale production. As more chemical knowledge accumulates, the value of this unassuming ester as both a workhorse intermediate and a springboard for innovation keeps growing. Every new generation of chemists finds fresh challenges and hidden potential in familiar molecules like methyl difluoroacetate, making its future as exciting as its past.
Where Methyl Difluoroacetate Matters
Methyl difluoroacetate isn’t something most people talk about at the dinner table, but brushing it off as just another chemical misses the bigger story. This molecule, with its two fluorine atoms replacing two of the usual hydrogens in methyl acetate, packs a punch in the world of organic synthesis. Chemists working on cutting-edge medicines use it as a building block. I’ve come across it in laboratory settings where every component must be carefully chosen to make complex molecules for testing potential treatments or manufacturing specialty agrochemicals.
Link to Pharmaceuticals
A lot of breakthroughs in drug development depend on these sorts of unique chemicals. The presence of fluorine changes properties in ways that aren’t obvious until you get deep into the science. Putting fluorine atoms in a drug molecule can boost its ability to survive in the body or make it latch onto a target enzyme longer. This means methyl difluoroacetate, even if it never ends up on a pharmacy shelf, plays a secret but critical role in shaping tomorrow’s cures. Researchers can attach this fragment onto a molecule to try new combinations, and those experiments wouldn't be possible without chemicals like this.
Role in Agriculture and Specialty Chemicals
Across the agricultural sector, tweaking molecules sometimes creates pesticides that work with greater precision or break down more predictably in the environment. Farmers and communities benefit when these changes lead to treatments that cut down on crop loss without harming the soil or water. Chemists have shown time and again that swapping out a few atoms can transform a compound’s behavior, and methyl difluoroacetate is a go-to building block in these transformations.
Environmental and Safety Discussions
Bringing any chemical into a new product raises questions beyond the lab. I’ve noticed in my own discussions with industry scientists that making and handling fluorinated chemicals brings an extra layer of responsibility. Some fluorinated compounds stick around in the environment or show up where you don’t want them. Regulators and companies keep an eye out for these risks, demanding clear documentation and safe handling procedures. Methyl difluoroacetate seems to fly under the radar compared to high-profile pollutants, but its use still calls for good training and regular safety checks.
Seeking Better Solutions
People ask what’s next as we rely on fine chemicals in research and manufacturing. Creating greener synthesis routes or switching to biodegradable alternatives can’t always happen overnight. In university labs where researchers push boundaries, I’ve seen how guidance from environmental advocates or industry oversight can set the right direction. Some efforts look at how to recycle materials or cut down on hazardous waste, and every piece counts. Being honest about the trade-offs — innovation versus risk — is the first step toward smarter, safer solutions.
The Bottom Line
Methyl difluoroacetate may sound distant from everyday worries, but its fingerprint happens to turn up in some of the most innovative research in medicine and agriculture. The careful, science-driven use of such chemicals is essential for progress, but staying alert to the challenges keeps both people and ecosystems safer as science moves forward.
Simple Chemistry, Big Impact
Methyl Difluoroacetate—sounds technical, but it’s a straightforward compound if you spend any time with chemistry. Bringing it down to earth, this molecule features a methyl group and two fluorine atoms stuck to an acetate backbone. Its chemical formula is C3H4F2O2. That arrangement shapes how the compound behaves, both in the beaker and out in the world. My own fascination with simple organic molecules started back in college, cracking open dusty textbooks and running my fingers over hand-drawn stick diagrams. Nothing makes you appreciate chemistry’s impact like seeing those formulas turn into real-world action.
Practical Uses and Day-to-Day Relevance
Methyl Difluoroacetate generally shows up where folks need a source of difluoromethyl groups—whether that’s in research labs, the pharmaceutical world, or in specialized manufacturing. Chemists value it for its ability to tweak the behavior of drug candidates or fine-tune chemical reactions. I remember seeing a research paper once that highlighted how a small change, like swapping in two fluorine atoms, could make a drug last longer in the body or become more stable in storage. Tiny molecules can steer big effects, and this compound serves as a reminder of that.
Why the Formula Matters
It’s easy to gloss over the details and just gloss over formulas like C3H4F2O2. If you’re a student slogging through organic synthesis—or a parent checking a chemical label in the garage—understanding this isn’t about showing off in trivia night. It’s about safety, accuracy, and making informed decisions. The specific hydrogen, carbon, and fluorine counts spell out how reactive the molecule can get, how it might interact in a reaction, and even hint at toxicity. Chemistry isn’t just theory; these details mean something in the real world.
Safety and Stewardship
Many people don’t realize that swapping hydrogens for fluorines does more than just alter the name. Fluorinated chemicals often resist breaking down, sticking around in nature and sometimes raising eyebrows from environmental scientists. Synthetic chemists face a responsibility to weigh these effects, not just for immediate performance but for long-term safety. Years back, I sat in on a seminar about “forever chemicals” and couldn’t help but think how every small ingredient on a chemical inventory list fits into that bigger picture. Just because a molecule like this seems niche doesn’t excuse us from considering impact.
Finding Solutions Beyond the Lab
Education stands out as one of the strongest tools for making better choices with chemicals, even less common ones like Methyl Difluoroacetate. Industry and academic professionals should champion transparency, both through labeling and accessible information about molecules used in products. Back in the day, young chemists like myself often relied on word-of-mouth or half-understood lectures. Today, nothing beats open databases and clear guidelines to help everyone—chemist or not—follow best practices.
Regulation does its part, too, prompting safer substitution when necessary and promoting research into less persistent alternatives. Searching for balance isn’t always easy, but staying alert and curious about chemical formulas keeps us all a step ahead. C3H4F2O2 may seem tiny, but understanding it places power in the hands of scientists and citizens alike, letting us shape a safer, smarter future.
A Silent Player in Chemistry—and Its Risks
Every so often, a little-known chemical pops up, and suddenly everyone wants to know if it's dangerous. Methyl difluoroacetate doesn't turn up in a Google search as often as, say, household bleach or gasoline. Still, ignoring it would be a mistake, especially since it shows up in niche laboratory settings, maybe the odd chemical synthesis for pharmaceuticals or fine chemicals.
I remember the first time I saw a flask of methyl difluoroacetate on the lab bench. No panic in the air, just the usual cautious vibe scientists get when working with chemicals where safety data feels incomplete. The reality is, not every compound used in research comes with a thick binder of health data. That’s no excuse to get sloppy.
What Science Knows — And What Still Stumps Us
Here’s the thing. Methyl difluoroacetate carries the “difluoro” label, which means it contains fluorinated groups. These don’t always make a chemical toxic, but in many cases, organofluorines behave in unpredictable ways. Think of legacy foes like Freon and certain firefighting foams—safe on paper, but hazardous for the environment and living cells after regular contact.
Most published toxicity reports on methyl difluoroacetate are thin. There’s barely any long-term human exposure data, mostly animal studies if anything gets referenced at all. Toxicity often depends on the family, and some methyl esters pack a punch—think methyl acetate, which acts as an irritant. Add fluorine to the mix, and now you’re in a territory where simple exposure through skin or inhalation gets unpredictable fast. A lot of chemists treat these compounds as potential moderate hazards out of caution. You won’t see it handled with bare hands, and gloves, eye protection, and fume hoods are standard.
Legitimate Concerns—And Why Carelessness Costs
Any lab worth its reputation considers accidental inhalation or spills a real threat, even if the company selling the chemical doesn’t slap a skull-and-crossbones on the label. A sharp solvent smell can signal irritation to eyes and lungs. Fluorinated substances sometimes stick around in the body, and researchers worry about bioaccumulation even with compounds that have short half-lives.
Regulatory groups like the EPA, OSHA, or the European Chemicals Agency don’t always keep up with newer or rarely used chemicals for industrial settings. Waiting for public alertness can feel like an exercise in futility. That doesn’t mean ignore established lab precautions. Spills and vapor happen; acting quickly with proper ventilation, containment, and decontamination pays off every time.
Looking for Solutions—And Responsibility
The lesson here stretches beyond just one chemical. Transparency and information sharing matter more than ever. Labs posting their own safety notes on specialized chemicals online, public safety agencies encouraging wider reporting even for low-volume solvents, and universities updating in-house hazard databases based on ongoing research—these measures cut down risk.
Nobody should shrug off exposure just because a compound remains obscure in public conversation. Taking extra care, pushing for more complete data, and respecting even the quietest of chemicals keeps everyone a little safer—both in the lab and far outside it. That’s stewardship on a real, daily level.
Why This Chemical Demands Respect
Methyl difluoroacetate looks simple on paper—easy to write off with its almost forgettable name. Yet those who have ever worked in a lab know, even plain-looking bottles can cause real trouble if not stored right. This chemical falls under the umbrella of fluorinated esters, which means it can be volatile, and it isn’t something anyone wants to spill, breathe in, or suffer contact with skin. Though you won’t find it in grocery stores or home workshops, it crops up in chemical research environments and some manufacturing spaces.
I spent a number of years in a university lab, and I’ve watched careless storage create avoidable hazards. I've also spent late nights re-labeling shelves after seeing how quickly a simple mix-up could lead to a headache or, if the substance is abused, a more dangerous mess. Over time, small risks pile up into big ones.
Choosing the Right Spot
Keeping methyl difluoroacetate out of trouble means picking a cool, dry, and well-ventilated storage area—never an office drawer or any random corner. Warmth can push pressure up inside a sealed bottle, and moisture in the air risks encouraging side reactions that can make handling more unpredictable. Strong ventilation matters in case a bottle leaks or vapors escape; nobody wants headaches, dizziness, or worse from accidental exposure.
Flammable storage cabinets with clear labeling usually do the job well. These cabinets are built to resist fire and designed to limit the spread of accidental fumes in case a leak does happen. Make sure to keep incompatible chemicals away—acidic solutions, strong oxidizers, and bases belong on separate shelves. One careless shelf-mate can trigger an unplanned chemical reaction nobody wants to deal with.
Dependable Containers and Honest Labeling
Bulky glass bottles with airtight seals tend to work best. Plastic might seem safe, but certain solvents can slowly eat through or weaken plastic over months. Always check the cap; an ill-fitting top means vapors will leak, sometimes so slowly you don’t notice until the headache starts. Spare everyone the confusion, and include the chemical name and date opened on the label. That little bit of extra work pays off fast.
Clear records aren’t bureaucracy—they’re lifelines. If you inherit storage space from someone else, assume nothing’s safe until you’ve checked the labels and inventory. A forgotten, mislabelled bottle can turn a routine clean-up into a call to hazardous materials teams.
Training Goes Further Than Rules
Anyone with access to methyl difluoroacetate should have real training. Videos and manuals help, but nothing replaces a walk-through with someone who’s handled the stuff before. Too many incidents start with, “Nobody told me…” Hands-on walkthroughs and drills bring home how easily accidents can happen if gear or labels get ignored.
Personal protective equipment should always be within arm’s reach—lab coats and chemical splash goggles at minimum. Gloves must be chosen carefully, as not every material blocks fluorinated esters effectively. Chemical-resistant trays beneath storage shelves add insurance in case a leak does happen.
Why Vigilance Matters More Than Any Written Rule
For every accident I’ve heard about, there’s usually a story behind it. Maybe a new hire didn’t think twice, or an old timer cut a corner late after hours. Safe storage is about culture as much as cabinets. Small routines—like wiping off bottles, checking seals, or making sure somebody’s double-checked the ventilation—become the best defense against the risks that come from complacency. Responsible storage exists not to bog down research, but to make sure the work can continue safely for everyone involved.
Getting Acquainted with Methyl Difluoroacetate
Methyl Difluoroacetate isn’t on the lips of most folks unless you live and breathe chemistry. This modest compound, known by its chemical formula C3H4F2O2, often works quietly behind the scenes in labs and sometimes in industry. Looking at its physical properties gives you a sense of just how unforgiving, or workable, it can be if you ever find yourself handling it on the job.
Physical Features That Matter in the Real World
Methyl Difluoroacetate shows up as a colorless, mobile liquid. It messes with your nose immediately since it gives off a sharp, almost biting odor that tells you not to get too cozy without proper ventilation. At room temperature, it moves pretty quickly—meaning it’s got a low viscosity—so it won’t cling to surfaces or pour sluggishly.
Boiling point gives people headaches in both kitchens and chemical plants, and this compound isn’t much different. Methyl Difluoroacetate boils at about 82 to 84 degrees Celsius. That’s low enough to make you pay attention if you’re thinking about heating it or transferring it to open containers. Keeping control of heat and airflow can quickly move from good practice to critical safety. It evaporates fast too, which means simple spills don’t hang around.
Density falls below that of water, landing somewhere near 1.27 g/cm³. This detail might seem small, but it comes up every single time containers are filled or mixed. I’ve learned that these differences change how things layer out or settle—something experienced crews keep in mind to avoid mistakes when scaling up.
Why Its Solubility and Reactivity Hit Close to Home
One of the most relevant factors for folks handling chemicals day-to-day is solubility. Methyl Difluoroacetate dissolves well in organic solvents—think of the usual suspects like ether or alcohol—but shows less love for water. In my experience, this limits where and how spills can migrate, but it also means cleanup stays manageable if you catch it early. The partial water solubility can fool people into underestimating its reach, which is never a good move in a busy workspace.
Temperature plays its own dangerous games with volatility. When you heat this stuff, vapors form rapidly and hang in the air low to the ground, since they’re heavier than air. Older ventilation systems can let these vapors build up. Hearing stories from maintenance crews and watching vigilance fade after a few quiet months tells me the risk is real, even when guidelines seem boring.
Making It Safer For Everyone
People often rush to focus on lab procedures or regulatory documents. Personal experience reminds me it’s just as important to share clear, real-world lessons: avoid open flames, keep containers closed up tight, and check your PPE for signs of wear before every shift. Fact-based awareness of flammability and volatility helps people make smarter choices, so chemical burns and surprise explosions stay rare.
Training programs should walk staff through the real look, feel, and smell of these liquids. This matters because no sheet of paper will ever match the impact of someone who’s seen a near-miss. Checklists work, but stories from experienced hands add weight.
Choosing Respect Over Routine
Every small insight from handling Methyl Difluoroacetate—its mobility, its creeping vapor, its stubborn smell—adds up to a sharper, safer workplace when people learn what to expect, rather than rely on luck or outdated habits. Respecting a compound’s quirks gives everyone a stronger shot at going home with nothing more than another ordinary day behind them.