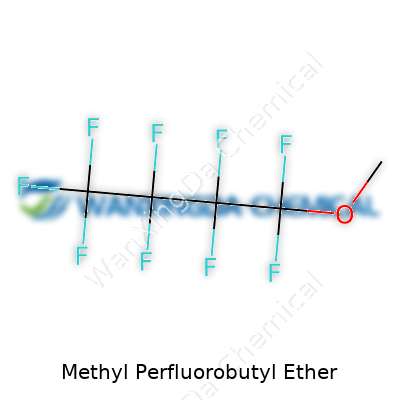
Methyl Perfluorobutyl Ether: A Deep Dive into Science, Industry, and Society
The Story Behind Methyl Perfluorobutyl Ether
Methyl perfluorobutyl ether isn't a newcomer. Decades ago, chemists searching for new solvents stumbled on the unusual qualities of perfluorinated compounds. Traditional ethers already served labs well, but adding perfluorocarbon chains rewrote the rulebook. Big chemical players started refining methyl perfluorobutyl ether for specialty tasks in the 1990s, seeing potential in electronics cleaning and heat transfer. Across my own experience working with technical fluids, seeing the transition from simple hydrocarbon solvents to smarter, more targeted chemicals, tells a story about progress—and about the headaches that tag along.
Getting to Know the Substance
With a structure built around a methyl group linked to a chain armored with fluorine atoms, methyl perfluorobutyl ether stands out. It's a clear, colorless liquid, lurks at a relatively low boiling point for a fluorinated fluid, packs serious chemical stability, and resists most acids, alkalis, and oxidizers. The stuff barely mixes with water or alcohols, and practically laughs off biological degradation. Its faint odor, reminiscent of other fluorochemicals, rarely matters except in poorly ventilated spaces. Tackling heat or fire, it won’t support combustion, which saves some sleep for engineers and plant managers.
Technical Details and Labeling Realities
When talking technical specifications, you glance at purity, molecular weight, volatility, refractive index, and residual metal content. I remember opening bottles in the lab and scouring the label for water content, because even trace moisture messes up sensitive electronics. As a hazardous chemical, it travels with detailed labeling, addressing inhalation, skin contact, and the environmental curse fluorinated chemicals carry. Governments ask for standardized safety symbols, and chemical codes like CAS 163702-08-7 stay attached to every container—regulators want no confusion.
How It’s Made and How It Changes
Synthesizing methyl perfluorobutyl ether doesn't rely on backyard chemistry. Large-scale production depends on high-pressure equipment and skilled operators, tapping methyl or perfluorinated alcohol sources, then swapping oxygen and carbon atoms with great care. Fluorination reactions demand patience and deep pockets; this isn’t a cheap process, and the feedstocks come with their own baggage. Chemical tweaks can nudge reactivity or change volatility, but once built, the molecule stands stubbornly resistant to breakdown. This resilience brings upsides and real baggage, because you can’t just rinse it down the drain and hope for the best.
Names and Faces: Synonyms of Methyl Perfluorobutyl Ether
Industry knows this molecule under several headings. Some folks speak of it as methyl nonafluorobutyl ether, others abbreviate to MPFBE or use naming conventions tracing the number of fluorine atoms. Chemistry forums toss around obscure trade names, though on the job most people ask for the perfluorinated methyl ether and the packaging does the rest.
Keeping People and Places Safe
Handling fluorinated solvents demands respect. Disposable gloves, proper ventilation, and well-calibrated detectors are the reality for operators. Health authorities keep close eyes on exposure guidelines, since animal studies point to risks with repeated inhalation or skin contact. Long molecules with lots of fluorine tend to accumulate in places that make ecologists and epidemiologists uneasy. Robbing the substance of chance to escape means good containment, smart engineering controls, and up-to-date employee training. In my career, watching a single small spill lock down a facility for hours left an impression: these substances go everywhere if uncorked.
Where It Shows Up: Real-World Applications
Electronics manufacturing counts on methyl perfluorobutyl ether for cleaning delicate circuit boards. Its stability and non-conductive nature end up crucial for rinsing away tiny contaminants from finished devices. Medical industries see value too, especially where inert heat transfer makes a difference. Sometimes used as a carrier fluid or to flush tightly toleranced parts, the ether brings benefits that old-school chlorinated solvents never matched. Research labs put the material to work as a unique solvent for HPLC separations. Unlike older generations of halogenated fluids, this one runs cleaner, evaporates well in controlled settings, and doesn’t corrode sensitive machinery.
Pushing the Science: Research and Development
Chemists and engineers look for new ways to put methyl perfluorobutyl ether to use. Improving the life span of technical fluids fascinates researchers, especially as devices shrink and thermal management grows trickier. Teams at leading universities and private firms sift data from pilot-scale reactors, asking if they can nudge stability or tailor volatility for new tasks. The push for safer, more readily biodegradable fluorinated compounds hits roadblocks, but incremental progress keeps the industry on its toes. In our team’s work, even tiny changes to solvent structure sometimes opened new doors in cleaning or separations.
Toxicity: The Lingering Cloud
Nobody calls perfluorinated ethers harmless. Animal studies nail down risks that come from breathing high concentrations or chronic workplace exposure. The body, built to handle oxygen, water, proteins—not exotic fluorocarbons—often responds with confusion. Perfluorinated chains won’t break down in soil or water on any timeline we care about, and this persistence draws serious scrutiny. Traces have turned up in groundwater, wildlife, and even human blood samples. Regulatory agencies clamp down hard on waste handling, demanding incineration or high-temperature destruction, not landfill disposal. Across industries using this fluid, engineers dig for containment and recycling solutions. The alternative—rampant environmental spread—costs more in lawsuits, emergency response, and fleeing public trust than anyone cares to admit.
The Road Ahead: Looking to the Future
With each iteration, demand grows for technical fluids that blend high performance with health and environmental responsibility. As a group, perfluorinated chemicals face growing bans and restrictions across Europe and North America. Engineers work on clever recycling systems, drawing every use out of each gallon before destruction. Replacement solvents inch forward, but the chemistry world rarely lets go of workhorse molecules until absolutely necessary. Science keeps exploring bio-based and less-persistent alternatives, but thermal stability and chemical resistance prove tough benchmarks to hit. As regulatory pressure builds, big players look to tighter containment, real-time monitoring, and process redesign to cut down emissions.
Practical Solutions for a Cleaner Tomorrow
Industry’s real-world shift won’t happen just with new molecules. Smart containment, closed-loop recovery systems, hands-on worker training, and policy built on health science give the best odds of keeping technical benefits while lowering risk. My own experience tells me cutting corners always leads to headaches—whether measured in lawsuits, sick colleagues, or lost contracts. Supporting academic research into safe substitutes and stricter waste management pays off, even if only by keeping doors open in regulated markets. A future shaped by science and accountability calls not for fear, but for honest effort and stubborn resilience.
Step through a research lab or electronics production plant and you’ll encounter chemicals with names longer than most shopping lists. Methyl Perfluorobutyl Ether stands out for its mouthful of a title, but its real utility lies in the work it quietly handles behind closed doors. Over the years, I’ve watched this compound pop up in surprising places, often overlooked in favor of flashier technologies. Still, its importance shows up each time someone boots up a device or a scientist needs a clean slate for sensitive work.
Why Do Companies Use Methyl Perfluorobutyl Ether?
Methyl Perfluorobutyl Ether finds the spotlight mostly for its unique solvent powers. Electronic manufacturers trust it for precision cleaning, especially on items like circuit boards and sensitive wafers. You spill coffee on your laptop, send it in for repair, and somewhere along the line, a specialist might lean on this ether to wipe away residue without damaging intricate components.
Its chemical makeup brings strong resistance to breaking down under heat or electric charge. That spells safety for tech workers and end users both. Hospitals lean on this ether for sterilizing medical gear and prepping surfaces that see critical use. Scientists favor it for high-purity needs, where even a hint of residue can spoil months of work or botch an experiment. Its low surface tension lets it slip into tight crevices and whisk away even stubborn grime.
Impact on Health and the World
As with many strong cleaners, this compound wants respect. Industry leaders flag its possible health effects and stress wearing proper gear and using equipment that keeps fumes away from workers’ faces. Long periods of exposure raise questions about respiratory and organ health, though specifics can be hard to pin down. Companies walk a line between effective cleaning and keeping their crew from harm.
The bigger worry, from a broader perspective, comes from the “perfluoro” part of its name. Substances in this chemical family break down slowly in nature. Once they seep into water or soil, they can hang around for generations. Scientists call these “forever chemicals” for a reason. A study published in Environmental Science & Technology traced PFAS compounds — this ether’s cousins — across rivers and groundwater worldwide. Some of the health issues linked with heavy or long-term PFAS exposure include hormone disruption and possible cancer risks.
Searching for Balanced Solutions
Switching to greener chemistry feels like slogging uphill. High-purity cleaning solvents aren’t easily swapped for water and soap. Old-school options often did more ecological or human harm than new alternatives, so staying up-to-date on research helps keep both people and the planet safer. Every time a regulation update hits, companies scramble to test out substitute formulas. The challenge: matching the same performance in cleaning without unwanted leftovers.
Regulators in the United States and Europe already moved to limit or track PFAS compounds in water and air. Deciding how far to go — without shutting down important industries — means weighing public safety against jobs and technology progress. One avenue lies in stricter recycling and capture systems inside factories. Forging public-private partnerships can guide research toward safe breakdown pathways, shrinking the long-term footprint these chemicals leave in the world.
Learning about Methyl Perfluorobutyl Ether opens a window into bigger debates about safety, responsibility, and innovation. No one wants laptops full of crud or syringes fresh from the factory with hidden germs. But hidden costs need facing, not just for now, but for future generations, too. As choices pile up, each one deserves a clear-eyed look, rooted in fact and focused on real people’s lives.
Digging Into The Facts
Methyl Perfluorobutyl Ether isn’t a compound most folks come across unless they work in a lab or a tech facility. I first bumped into its name while helping a friend clean out some dusty fume hoods at her workplace, and what struck me was how little the average person hears about these newer synthetic chemicals. Every time a compound like this pops up in industrial circles, questions start flying about its safety.
This ether acts as a specialty solvent and heat-transfer fluid. Some engineers count on its unique chemical stability and non-flammable nature. Those properties might sound reassuring, but chemistry doesn’t hand out free passes; every benefit can have a downside. My years working alongside chemists taught me that handling anything with “perfluoro” in the name calls for awareness, if not a bit of healthy skepticism.
Understanding the Health Risk
Most solvents introduce some risk. With Methyl Perfluorobutyl Ether, respiratory irritation and eye discomfort land high on health watchlists. Technicians have described the scent as sharp, and nobody wise stands over the open bottle inhaling fumes for fun. Anyone careless enough to splash the stuff on bare skin often regrets it soon after. Every chemical storage room hangs thick with stories about someone learning the hard way.
Long-term effects keep coming up in scientific reviews. Modern perfluorinated chemicals have drawn the attention of researchers for their environmental persistence. Some of them stick around in nature, circulating through water systems and wildlife for unimaginable lengths of time. Data keeps growing on perfluorinated compounds and their possible links to hormone disruption and other health issues. Regulators and health agencies still dig through the evidence, but past experience with similar chemicals makes most scientists want strict controls in place..
Environmental Impact Matters
Discussions about chemical safety too often stick to the question “Will it hurt me right now?” For years, I watched companies try to sidestep the bigger picture. Methyl Perfluorobutyl Ether, like its chemical relatives, resists breaking down in water or soil. Waste handling becomes a central concern. Legally and ethically, users must keep track of how and where the solvents get disposed. Stories from communities living near industrial plants show how hard it can be to clean up once these compounds slip through the cracks.
Steps for Safer Handling
The biggest safeguard stays the same: respect the chemical. Gloves, splash goggles, and fume hoods aren’t just for show. Every lab I ever visited hammered that lesson home. Facilities need routine checks for leaks and up-to-date safety training. Most mishaps trace back to someone skipping a step, ignoring a sign, or making quick work to save a few minutes. A culture of caution pays off.
Inventories should track how much solvent comes in and out. Waste containers should be sealed and labeled clearly. Regular reviews of how solvents are stored and handled help spot any lazy habits before they grow into hazards. Clear policies that protect workers and protect the environment build trust and help avoid ugly headlines later.
Looking Forward
Technology rarely moves backward, and the list of engineered chemicals gets longer every year. What sticks with me is how trust—between companies, workers, and surrounding communities—depends on honesty about risks and visible care for safety. Chemicals like Methyl Perfluorobutyl Ether open new possibilities but demand real responsibility from everyone in the supply chain.
Peeling Back the Science
Some chemicals seem like wizards in a bottle, and Methyl Perfluorobutyl Ether is one of them. Technicians get excited when an ether like this shows up in the lab because the stuff just shrugs off water and oil. If you’ve ever spilled coffee on a treated surface and seen the beads roll off, you’ve brushed up against the everyday magic that comes from the same family as this fluorinated ether. Its fully fluorinated carbon chain gives it a special knack for stability, which is the real trick behind a lot of practical uses.
No Sweat From Heat or Cold
Heat might frazzle some liquids, but this ether won’t break a sweat. It stays stable over a broad range of temperatures, resisting boiling until things get hot enough to fry an egg several times over. That matters when you work in a research lab—or even when engineers use it in electronics—because you want something that won’t change its mind just because the air conditioning failed. Thanks to this, it often finds a home as a heat transfer fluid or a cooling agent, especially where failure is not an option.
Density and Invisibility Go Hand in Hand
Pick up a vial, and it feels heavy. The density is no joke; it sits somewhere above most common solvents, so it doesn’t float away or evaporate in a flash. Yet, it’s nearly invisible in action. No color, almost no scent. That means it doesn’t add another layer of mess to most projects and won’t send you running for fresh air. The low toxicity lets people handle it without worrying too much, but good gloves are still your friend.
Why Non-Reactivity Draws Eyes
The chemical backbone does the heavy lifting. The carbon-fluorine bonds are among the toughest in organic chemistry, and nothing short of serious, specialized conditions will break them. Most acids, bases, or even reactive metals can’t touch this molecule. That means you can use it in sensitive environments without worrying it’ll react with stuff it shouldn’t. It won’t stain, corrode, or eat away at containers, electronics, or lab glassware. For an old chemist who’s broken plenty of beakers, that kind of reliability is gold.
Environmental sticker shock
Now there’s a catch. The same features that keep Methyl Perfluorobutyl Ether from breaking down for decades make it a headache outside the lab. In the environment, it just hangs around. The same structure that resists acids and bases doesn’t do nature any favors either. Some studies have shown that, like many perfluorinated compounds, it can stick around in water and soil far beyond our lifetimes. The push for greener chemistry means the world needs to take its persistence seriously, and that weighs on anyone who cares about the next generation.
Searching for Responsible Solutions
Sustainable chemistry calls for action. Companies can design better containment and recycling programs. Chemists can work on finding substitutes with shorter environmental half-lives. Regulatory bodies could keep a closer eye on the disposal and industrial use to avoid leakages and spills. Everyone playing with powerful chemistry inside the lab—myself included—owes it to the world outside to manage these substances with respect. Just because something is invisible and stable doesn’t mean it’s harmless if it slips away from careful hands.
Lessons From Handling Chemicals
Years working around chemicals have taught me respect for both the good they can do and the harm they bring when stored carelessly. Methyl Perfluorobutyl Ether catches a lot of attention in lab circles and industrial spaces. People lean into its use because it resists heat, evades reactivity with many materials, and keeps its cool in diverse conditions. Reading between the lines, this creates a temptation: stash it wherever there’s space and call it a day. In reality, cutting corners with storage doesn’t just invite financial losses—risks reach further, putting health and safety at center stage.
Understanding What You’re Dealing With
This ether is no kitchen cleaner. It belongs to a family of highly fluorinated compounds, giving it chemical stability but also making it persistent in the environment and tricky to handle for the long haul. From experience, improper storage methods spell trouble—everything from accidental inhalation to leaky containers has real-world consequences. Preventable spills and vapors will not wait for warnings. Those who’ve experienced even a whiff of volatile chemicals know the headaches aren’t metaphoric.
Solid Storage Steps—No Room For Shortcuts
Storing volatile organic solvents means more than a locked door. The focus starts with choosing the right container. Bypass plastics that could degrade; lean instead on tightly sealed metal or glass. Exposure to air, sunlight, and heat invites chemical changes and pressure build-up inside poorly chosen vessels. In practical terms, transparent bottles under a window tend to spell disaster sooner or later. I’ve seen warping and discoloration sneak up on inattentive labs, and the cleanup isn't pretty (or cheap).
Proper labeling cannot be brushed aside. Even seasoned chemists have made the mistake of assuming everyone knows what’s inside each bottle. Unmarked or mislabeled bottles sow confusion in shared workspaces, sometimes leading to regrettable mix-ups. The best labs I’ve worked in treat labeling like a rule, not a guideline, boasting clear, legible, and detailed descriptions.
The best choice for storage? Cool, dry, and well-ventilated spaces. Ordinary office shelves or basements collect humidity and elevate risk. Airflow matters just as much as temperature control. I remember one summer when an overstocked, sealed closet led to dramatic pressure changes, popping lids, and one sticky mess. Dedicated chemical storage rooms with consistent ventilation system checks become more than a line item on a budget—they protect health and save money by preventing accidents.
Long-Term Considerations
The conversation around these ethers keeps growing. Some researchers raise concerns about environmental persistence and human exposure. No single storage trick erases that. The safest labs review their chemical inventory regularly and ensure old stock stays fresh or gets disposed of according to local hazardous waste rules. This attention to detail limits risks associated with accidental release or cross-contamination.
Learning From Experience—Not Regret
Every person who stores chemicals shoulders responsibility for more than their own safety. Proper storage, regular checks, and honest communication about hazards reflects a mindset that values people more than product. Methyl Perfluorobutyl Ether proves that cutting-edge compounds don’t excuse old-fashioned mistakes. Setting up safeguards and demanding respectful handling creates a culture where lessons don’t come from misfortune, but from forethought and care. That’s where true expertise grows, one mindful choice at a time.
Untangling a Complex Chemical
Methyl Perfluorobutyl Ether doesn’t roll off the tongue, but its footprint doesn’t escape notice for those paying attention to chemicals and environmental health. Industries put this compound to use for its stability and resistance. It stands tall against heat, acids, and various solvents, which makes it attractive in electronics and specialist applications. Every time I hear about its use, I think back to my days seeing labs push harder for new materials, usually in pursuit of efficiency or performance. But that reliability comes with consequences that ripple well beyond the lab or factory.
Persistence in Nature
Unlike many everyday substances, Methyl Perfluorobutyl Ether sticks around in the environment. Traditional bacteria and sunlight barely leave a scratch. It enters water, soil, and possibly even the air during manufacturing or from products that eventually break down. I recall news stories detailing rivers where fish populations struggle—customarily, such changes tie back to industrial waste, and the chemistry here shares a similar stubbornness.
Once these chemicals enter natural systems, they travel far. Scientific studies have picked up related compounds in places as remote as the Arctic, which tells us they do not respect borders. Local choices end up global concerns. What happens in an urban chemical plant might touch a distant coastline years later.
Impact on Living Beings
Perfluorinated compounds, as a family, raise red flags for how they interact with living bodies, both human and animal. The main issue is bioaccumulation: creatures take in these chemicals faster than they break them down. Watching scientists talk about this reminds me of older debates around asbestos or leaded gasoline—the symptoms only pop up after time passes. While researchers continue to sort through how much harm Methyl Perfluorobutyl Ether does, the worry focuses on immune, hormonal, and reproductive effects for people and wildlife.
Communities living near plants using or disposing of perfluorinated compounds have started raising their voices about water contamination. One thing is clear: once these chemicals enter our waters, getting them out proves technical and costly.
No Easy Solutions—But Action Matters
Sticking with the status quo brings mounting problems. Few governments regulate Methyl Perfluorobutyl Ether as strongly as related persistent organic pollutants. Industry claims limited risk if handled properly, yet evidence mounts that leaks and releases cannot always be prevented. I’ve seen innovation thrive under pressure. Bans on other persistent chemicals led companies to invent safer substitutes. Moving forward takes transparency from industry, high-quality research, and firm regulations.
Switching to greener compounds can protect water and communities. We’ve done it with other troubling chemicals—think about the reduction of ozone-depleting substances after the Montreal Protocol. Every time public pressure met science, it forced a rethink. The challenge lies in keeping both industry needs and public health in sight, making sure we protect future generations from decisions that are convenient now but damaging later.
Keeping Watch for a Safer Future
Communities deserve to know what’s in their water and soil. As regulators and advocates push for more testing and disclosure, regular people gain the tools to protect themselves. Greater investment in cleanup technologies adds another layer of defense. As with past environmental battles, public engagement proved crucial. Looking ahead, we’re all part of the solution in demanding safer practices and cracking down on the most stubborn pollutants in our world.