Methyl Perfluoroisobutyl Ether: More Than Just a Tongue-Twister
Looking Back at Its Historical Roots
Methyl perfluoroisobutyl ether didn’t just suddenly show up in a lab vacuum nor slide into the market overnight. Its backstory traces the surging interest in fluorinated compounds that took off in the middle of the twentieth century. Folks wanted materials that could take heat, resist solvents, and brush off chemical attacks—traits that let industries dream up all sorts of new products. The demand for stable, inert substances led to the birth of all sorts of perfluorinated ethers as companies searched for specialty solvents and heat transfer agents. People sometimes overlook the steady march of chemistry, but this family of ethers has sat quietly in the toolkits of scientists and engineers for decades now, mostly serving behind the scenes in advanced manufacturing, chemical synthesis, and electronics.
Overview and Identity: What Are We Really Talking About?
Here’s where most people check out, because the name alone rattles off the tongue like a list of side effects. Methyl perfluoroisobutyl ether belongs to the perfluorinated ether family, with carbon-fluorine bonds that pack some of the highest radiochemical stability around. Trade journals and catalogs toss out alternative names, from 'Perfluoro(1-methylethoxy)methane' to product codes, but the bottom line stays the same: you get an ether molecule, fully fluorinated, with one methyl group sticking out. Many who spend years around chemical sourcing see synonyms and registry numbers piled up in spreadsheets; at the end of the day, the performance on the shop floor or in the reactor is what really matters.
Physical and Chemical Character: Built Tough, Stays Cool
Let’s talk nuts and bolts. Methyl perfluoroisobutyl ether stands out by refusing to play along with water or typical mineral oils. Its low boiling point often lands it a job where temperature control matters, and its density feels just right for certain specialized applications. Strongest of all, those carbon-fluorine bonds make the molecule about as resistant to chemical corruption as you can get, outlasting acids, bases, and oxidizers that would turn lesser organic solvents to mush. This chemical backbone explains why you won’t catch it breaking down or reacting unless you really force the issue—this stuff isn’t here for everyday parties, but it sticks around when heat and chems get rough.
Technical Stamps and Preparation Pathways
On the technical side, clear labeling and purity ratings have grown up as environmental and safety demands increased. The real challenge doesn’t come from dreaming up another ether—it comes from making it clean enough for demanding work. Preparation usually follows fluorination routes, starting with perfluorinated alcohols, ethers, or olefins, guided by catalysts that handle the violent reactions of elemental fluorine. There’s no faking purity here. The finished product often has to show off extremely low water content, a minimum of organic and ionic traces, and meet specs for everything from refractive index to boiling range. Field experience and conversations with quality assurance folks make it clear that real-world success hangs on manufacturer reliability—nobody wants last-minute surprises when deadlines loom.
Reactions, Tinkering, and Real-World Mods
This compound resists traditional chemical tinkering, but under enough pressure or the right catalyst, certain modifications are possible. In specialized fields, researchers have tried to graft related structures onto fluorinated ethers or break apart new derivatives for even cleaner or more robust performance, especially in electronics and semiconductor production. Most folks in the field won’t mess with these reactions without good reason since the risk and cost can skyrocket fast. Still, chemists love a good challenge, and there’s always some new angle driving the next round of research funding.
Safety Expectations and Operational Habits
Safety should always shape how we interact with methyl perfluoroisobutyl ether, not only because of its chemical pedigree but also thanks to lingering questions about possible health risks. Years ago, some assumed all perfluorinated materials stood outside concern, but emerging research has since upended that comfort zone, especially after evidence started mounting about related compounds sticking around in ecosystems and even human bodies. Daily experience tells me that proper ventilation, appropriate gloves, and storage away from open flames or incompatible materials show respect for what this ether can—and can’t—do. Regulations continue evolving, so companies now invest real money into both worker monitoring and environmental compliance, with training sessions and signage that go well beyond bare-minimum requirements.
Applications: Where the Work Really Happens
In the wild, methyl perfluoroisobutyl ether finds its way into electronics cleaning, specialty cooling fluids, precision engineering, and tough solvent mixes. Engineers rely on its heat resistance when designing systems that can’t tolerate common oils. Cleanroom environments value its near-total inertness. These uses don’t necessarily turn heads, but without compounds like this, a lot of industries would lose their edge or struggle with slower, messier processes. Some in R&D argue for new roles, like improved battery electrolytes or medical device coatings. The blockages here aren’t just technical—they’re economic and regulatory, as companies juggle performance, cost, and risk in a world that keeps demanding safer, greener, and more reliable technologies.
R&D and the Promise of New Discoveries
Folks tend to think chemical research crawls along, but the pace of discovery around perfluorinated ethers proves otherwise. Research groups in Asia, Europe, and North America chase after new synthesis methods that cut out hazardous reagents, use less energy, or yield improved physical properties tailored to emerging tech. Discussions at conferences get heated, especially over sustainability and toxicity, and journals regularly spotlight advances in purification, testing, and theoretical modeling. Funders often apply pressure for short-term results, but the biggest shifts come from collaborations that cross borders and industries. I’ve seen teams break through obstacles only after chemists, engineers, toxicologists, and supply chain experts hash things out together.
What Science Knows (and Still Debates) About Toxicity
Toxicity research keeps moving the goalposts. Decades ago, companies and labs worked under the assumption that compounds like methyl perfluoroisobutyl ether caused little harm, at least compared to the notorious PFOS and PFOA contaminants. New analytic techniques, stricter regulations, and better epidemiological studies have pushed the conversation forward, though. Scientists now study bioaccumulation, long-term organ impacts, and breakdown pathways with a sharper eye and more skepticism. In my own circle, a growing wariness surrounds deploying perfluorinated ethers in disposable applications, especially with mounting scrutiny from both public health advocates and regulatory agencies. Transparency and open publication of negative results matter as much as the positive, since nobody wins when risks get swept under the rug.
Looking Ahead: What Tomorrow Might Bring
The future for methyl perfluoroisobutyl ether depends on how we balance performance, safety, and sustainability. Newer applications grow out of real industrial problems—think advanced sensors, semiconductors, or medical gear—where nothing else quite measures up to its chemical tenacity. Still, the push for greener chemistries keeps growing as more people demand eco-friendlier alternatives and tighter regulations. Innovation probably won’t scrap perfluorinated ethers entirely, but tomorrow’s breakthroughs could limit their environmental footprint or open doors for recycling and recovery. Big strides will depend as much on political decisions and global supply chain shifts as on the slow grind of lab research. Looking around, I see a future where companies, governments, and scientists all pull in the same direction, but nobody should expect the road to smooth out on its own.
Methyl perfluoroisobutyl ether tends to slip past most headlines, but it’s found its way into some important corners of technology and industry. I hadn’t thought much about specialty solvents or dielectrics until I started talking to folks who work in electronics manufacturing and precision cleaning. They mentioned Methyl perfluoroisobutyl ether as this go-to chemical for tricky jobs. That sent me looking for why it gets that vote of confidence.
Not Your Average Solvent
The story starts with its chemical backbone. Fluorinated compounds pull off tasks other chemicals can’t match. Methyl perfluoroisobutyl ether resists breaking down when things get hot, made it onto the roster for cooling advanced electronics. High-performance computers and power electronics run into serious heat problems, and this stuff helps keep them running without frying their insides.
In industrial cleaning, the solvent eats tough oils and other residues from tiny components, without corroding fragile parts or leaving a mess behind. Cleanroom experts told me there are few options that stay stable, dry fast, and aren’t a pain to recover or recycle. Apparently, switching from chlorinated cleaners to these highly engineered ethers makes a huge difference not just for safety, but for precision too.
The Green Question
I’ve seen a lot of debate among chemists and environmental scientists. Methyl perfluoroisobutyl ether has a reputation for being persistent in the environment. Reports warn that it’s not something nature breaks down easily. That persistence does create a real challenge. We all saw how fluorinated chemicals—sometimes called PFAS—have been linked to water pollution and health worries.
In the past, people reached for whatever worked fastest or cheapest, often overlooking what could happen down the road. Now, stricter rules and more public awareness mean every use of fluorinated chemicals faces a microscope. A cleaner or coolant might work wonders for industry, but longer-term safety and cleanup can’t be pushed off. I’ve seen growing calls for tighter controls over who handles these substances and stronger incentives for safer disposal or alternatives.
Seeking Balance Between Performance and Responsibility
Like a lot of things in tech, it’s a balancing act. Demands for thinner, faster, and more efficient electronic devices have pushed manufacturers onto new ground. Chemistry is asked to keep up, but engineers and policy makers can’t afford to play catch-up with safety and the environment. Solvents like Methyl perfluoroisobutyl ether have made things possible, whether that’s keeping new chips from burning out or cleaning surgical tools before they reach hospitals.
It’s easy to point fingers at an industrial chemical once headlines focus on the downside. In my talks with both engineers and environmental watchdogs, though, a pattern popped up—open communication works better than just swapping blame. Some companies run closed-loop systems that trap and reuse their cleaning agents until the last drop, slashing emissions. Tech labs look for ways to blend their need for mind-blowing cooling with less risky formulations. Green chemistry teams keep experimenting, hoping for a solution tougher than the problem.
There’s no magic bullet. Maybe Methyl perfluoroisobutyl ether delivers an edge that’s tough to beat for now, but we’ve all learned not to write blank checks to chemicals, no matter how clever or effective they seem. Every field using it faces pressure to prove they’re not just buying time, but actually planning for the long term—because the bill from ignoring these problems always comes due.
Why Methyl Perfluoroisobutyl Ether Raises Eyebrows in the Lab
Methyl Perfluoroisobutyl Ether might sound like another cryptic name out of a chemical textbook, but its place in technology is anything but obscure. Folks in electronics, aerospace, and specialty labs cross paths with it when they work on cleaning, cooling, or specialty coatings. It works well, but it isn't a kitchen-counter cleaner. Anyone who’s actually spent time around this stuff knows one thing: respecting its potential risks isn't negotiable.
Ventilation Isn’t Optional—It’s Non-Negotiable
Too many labs make the mistake of relying on tiny desktop fans and half-open windows. I’ve seen the difference that proper ventilation makes: chemical fumes never stick around. Installing a real fume hood or a solid, room-sized exhaust system beats any half-measure when you’re working with volatile chemicals. Take it from technicians who’ve skipped this step and felt lightheaded or irritated in the eyes—breathing this stuff day in and day out without an exhaust will catch up to you.
PPE Is More Than a Costume
Personal Protective Equipment sits in every chemical safety talk, but dealing with this ether doesn’t give you the luxury of half-zipped coats and missing gloves. Nitrile or neoprene gloves keep the nasty solvents away from your skin. I always check the gloves for holes because the fastest way to chemical burns or persistent dryness is to get lazy with your gear. Safety goggles and a snug lab coat round off the basic defense, and for big jobs or splashes, even a face shield sees real use.
Spills Happen—Have a Routine, Don’t Wing It
No matter how careful people get, spills sneak up. The difference is in the cleanup plan. Sand, absorbent pads, and a chemical waste drum keep everything contained. I learned early not to use paper towels or your bare hands on this one. Even tiny amounts of solvent on the surface can quickly evaporate into the air. If the lab or workroom sets a habit of labeling, storing, and disposing of every bit of chemical waste, fewer panicked runs to wash stations follow.
Respect the Unknowns and Keep Up with the Science
Every year, researchers publish new findings about the health impacts of perfluorinated compounds. Long-term health risks and environmental persistence aren’t just rumor—they’re in the soil and water long after the waste leaves the lab. Anyone who cares about staying healthy doesn’t downplay these unknowns. Following guidelines from bodies like OSHA and the EPA, and keeping up with SDS updates, keeps you ahead in the safety game. If in doubt, I have always found that consulting with an industrial hygienist or environmental health officer beats guessing.
Training and Communication Save More Than Just Time
Talking through safety drills, holding refreshers, and not treating new hires like they’ll “figure it out” keeps accidents rare. I’ve seen rookie mistakes happen because someone felt embarrassed to ask for help. Open lines and a learning mindset cut through confusion and make sure everyone walks out safe and healthy.
Working with potent chemicals like Methyl Perfluoroisobutyl Ether isn’t like cooking with oil. It demands preparation, clear procedures, and real investment in protective gear. I’ve watched the best-run labs always put people ahead of shortcuts, and that’s what keeps everyone going home at the end of the day.
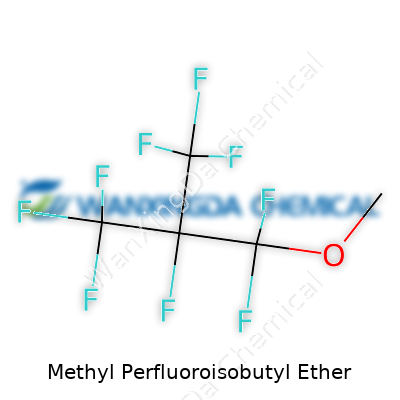
The Structure of Methyl Perfluoroisobutyl Ether
Methyl Perfluoroisobutyl Ether doesn’t come up in coffee shop talk, but its presence ripples across high-tech fields. The chemical formula, C5H3F13O, spells out its structure: a methyl group linked to a perfluoroisobutyl ether backbone. Imagine swapping out the regular hydrogens you’d expect with fluorine atoms—one of the toughest elements to dislodge. This structure gives the molecule a set of wings most compounds can’t match when it comes to sheer chemical stability and resistance to heat.
The molecule has a center marked by an ether oxygen atom, with a methyl group on one side and a perfluorinated isobutyl group on the other. Chemically, you’d see it drawn as CH3OC(CF3)2CF2CF3, a design that looks a bit odd if you’ve only seen simple alcohols or ketones before. The heavy load of fluorines means this ether shrugs off attacks from acids, bases, and everything in-between—including water and fire.
Real-World Impact
Now, why all the fuss? Compounds like Methyl Perfluoroisobutyl Ether don’t just collect dust on lab shelves. They step into roles where ordinary solvents or lubricants collapse. Fluorinated ethers crop up in specialty industries including electronics, aerospace, and medical device manufacturing. I’ve run into their uses firsthand while troubleshooting thermal runaway in complex circuit boards. A regular solvent would evaporate or break down under prolonged heat, leaving delicate parts vulnerable. Methyl Perfluoroisobutyl Ether stays put, keeping electronics dry and well-insulated even as parts sizzle into the red zone.
There’s also growing buzz in the world of battery coolants. Lithium batteries in electric vehicles and grid storage banks run hot, and traditional coolants have a nasty habit of breaking down or—even worse—catching fire. Fluorinated ethers don’t just stand up to the heat. They barely notice it. Stories from engineers stress how these chemicals make the difference between a safe high-capacity battery and a smoking heap of scrap.
Environmental Puzzle
Endurance sounds great, but it plants a thorny problem. Fluorocarbons don’t rot, dissolve, or fade in the environment. They stick around for decades, sometimes generations. Perfluorinated compounds earned the "forever chemical" nickname for a reason. Following news studies from the EPA and scientists in Europe, almost everyone in industrial chemistry knows the push to limit these chemicals, or at least replace them with safer alternatives.
As a person who follows chemical policy, seeing the industry caught between performance demands and environmental responsibility hits close to home. There’s constant pressure to find greener substitutes. Progress often creeps forward in inches, not leaps. Chemists are looking at short-chain alternatives, tweaking molecular designs so that they’re easier for microbes or sunlight to chop up. Investment in proper recycling systems for spent fluorinated ethers could keep the worst offenders out of the water and soil.
Steps Forward
If businesses and researchers keep an ear out for safer molecules—opting for drop-in substitutes, testing bio-based ethers, or engineering new polymers without the stickiness of fluorine—things could change for the better. Regulation will keep tightening. The field needs transparency, honest reporting, and clear communication with workers and communities. There’s a lot to learn from both sides: the remarkable staying power of fluorinated ethers and the equally persistent challenge of managing their environmental footprint.
Looking Safety in the Eye
Methyl Perfluoroisobutyl Ether doesn’t land in headlines very often, but those who deal with fluorinated solvents know it brings a unique blend of properties and challenges. Its chemical stability and low toxicity don't erase the real risks it brings if treated lightly. This compound doesn’t ignite easily, but its very nature means it wants a careful hand during storage and transport. Many chemical incidents I’ve come across started where someone thought “stable” meant “no sweat.” This stuff proves that assumption wrong.
Why Storage Demands Smart Choices
You won’t find large tanks lined up for this ether on a regular warehouse tour. It asks for handling that respects both people and the planet. Tight, sealed containers matter to keep vapors in and moisture out. Rust and corrosion challenge most metals, and not every container can stand up to the long-term effects of fluorinated liquids. High-density polyethylene and certain stainless steels keep up better. Sloppy sealing runs the risk of escaping fumes and spoiling product quality.
Facilities with good air flow stay safer than stuffy corners. Fume hoods, proper venting, and temperature control make up the backbone of smart storage. Anyone who’s worked a midnight shift with leaking solvent lines knows a whiff of these vapors can wreck the rest of your day. Leak-proof secondary containment prevents a small drip from turning into a big headache, especially if local rules clamp down hard on chemical spills.
How Shipping Changes the Stakes
Moving Methyl Perfluoroisobutyl Ether from place to place brings in another layer. Freight trucks and cargo ships roll past thousands who never gave a second thought to what’s inside the drums. I’ve seen whole batches arrive unusable because of lazy labeling or battered containers. Dropping drums, ignoring temperature swings, or forgetting to check seals means gambling with both human safety and big financial loss.
Proper paperwork shapes the journey from start to finish. Regulations change fast, and authorities draw fine lines between safe delivery and a legal headache. It’s not about red tape for the sake of it; these rules come from scars left by accidents that hurt real people. Up-to-date hazard labels and well-trained logistics crews take away some of the luck and put know-how back in control.
Room for Better Practices
A few solutions could change the landscape for everyone handling this ether. Better training for warehouse staff pays off, because most mistakes grow out of rushed or careless work. Real drills and clear checklists make a bigger impact than any sign on a wall. Inspections that look past paperwork—checking actual lid seals, secondary containment, and climate controls—cut risk down to size.
Industry groups don’t always reach down to the daily shop floor, but tapping into shared experience and chemical safety data keeps everyone a little sharper. Investing in emerging tracking tools, like remote temperature sensors and tamper-evident seals, sends a signal: these containers carry more than just chemicals—they carry trust.
Neighbors, regulators, and workers all want the same result: safe storage, smooth delivery, and nothing sneaking out where it shouldn’t go. Respect for the risks and tools to address them stand out far more than empty reassurances. Lessons learned, scars earned, and a little humility push storage and transport practices forward—one barrel at a time.
Methyl Perfluoroisobutyl Ether comes up in discussions about chemical use in industry a lot more these days, mostly because of its spot in the family of PFAS chemicals—those “forever chemicals” turning up in everything from firefighting foam to nonstick cookware. Every time a new chemical enters wide use, neighbors and workers ask what happens to our water, air, and bodies. Having spent years digging into chemical safety, I’ve learned it pays to treat new compounds from the PFAS family with healthy skepticism, even the ones not yet studied to exhaustion.
Known Concerns About PFAS
PFAS chemicals stick around in the environment and tend not to break down, sometimes hanging out in soil and groundwater for generations. Methyl Perfluoroisobutyl Ether shares some of those traits. What that means, in simple terms, is our grandkids could find traces of today’s industrial mistakes in their drinking water. We’ve already seen this story unfold in towns near manufacturing plants: folks end up with elevated levels of certain PFAS in their blood, even decades after exposure. Some researchers say long-term buildup can raise the risk of thyroid disease, cancers, and reduced immune response—no surprise neighbors are worried when a new PFAS shows up on the scene.
Sparse Health Data, Familiar Red Flags
The thing about Methyl Perfluoroisobutyl Ether is that there isn’t a lot of public information on its direct effects in people. That doesn’t mean it’s harmless—just that the data hasn’t made it to doctors or public health experts. It shares chemical cousins with compounds linked to liver and kidney problems, developmental delays, and even certain types of tumors in animal studies. If a chemical behaves like others that don’t break down in the body, I take extra caution, especially when there’s a known history of trouble in the PFAS group.
Bioaccumulation and the Water Problem
One challenge with Methyl Perfluoroisobutyl Ether comes from bioaccumulation. Fish in rivers downstream from plants pick up these chemicals, bringing them right into food supplies. Even folks miles away from any plant could end up consuming tiny doses through water or food. Some cities now regularly test water for select PFAS, but not every village or rural town has the funds or tech for detailed screening. This uneven coverage raises questions about environmental justice. Industries often land near working-class neighborhoods, but the risks roll out far wider.
Toward Safer Practices and Policy
Real progress against PFAS pollution comes from a few angles. Companies can switch to less persistent alternatives or look at closed-loop production to keep chemicals inside the plant. Lawmakers need to back tighter reporting and cleanup standards for PFAS emissions—making sure the polluters pay for contamination, not the families living nearby. Public health offices should call for independent, published data on methyl perfluoroisobutyl ether and its effects so that communities and healthcare workers know what to look for.
Until then, families deserve transparency. People ought to know what’s used in local factories and how their drinking water is being protected. This isn’t about demonizing chemicals; it’s about balancing innovation with real consequences for people and the world they live in. Ignoring the lessons from past PFAS blunders only sets up a repeat of old mistakes.