Methyl Triflate: An Editorial Commentary on a Powerful Chemical Tool
Historical Development
Chemistry tends to find its champions through unlikely candidates, and methyl triflate surely counts among them. Decades ago, scientists needed a way to transfer a methyl group with real efficiency. Early methods often left much to be desired: side reactions, poor yields, messy results. Then came methyl triflate. This reagent didn’t appear overnight; it owes its existence to a period when organofluorine chemistry pushed boundaries far beyond conventional thinking. Researchers struggled for years to harness the reactivity of triflic acid, and clever minds saw an opportunity. The methyl ester—the so-called “methyl triflate”—appeared in labs through persistent experimentation with triflic anhydride and methylating agents. By the late 20th century, its role became established in both academic and industrial settings. Any chemist flipping through journals from the 1970s on will spot its mark where stubborn methylations found their breakthrough.
Product Overview
Looking at methyl triflate, it’s hard not to think of how much power such a small molecule holds. It acts as a methylating agent—a substance that drops a methyl group onto a target molecule, often with remarkable speed and precision. These traits make it popular with synthetic chemists, who want reagents that don’t dither or produce unnecessary byproducts. Its reputation stems from consistent performance, even on substrates considered difficult to methylate by other means. Methyl triflate isn’t some quirky specialty; it has become a staple in the toolkit for building pharmaceuticals, agrochemicals, and functional organic materials.
Physical & Chemical Properties
Methyl triflate stands out thanks to its volatile, colorless liquid form. It gives off a sharp, lingering odor, which signals both potency and danger. At room temperature, it remains stable enough for use, but don’t let that fool you—once it meets moisture or reactive compounds, it gets aggressive fast. Its high electrophilicity means it wants to latch onto nucleophiles, making short work of most bases and some that typically resist methylation. It dissolves well in common organic solvents like dichloromethane, diethyl ether, and acetonitrile. People who spend their days in labs appreciate that it doesn’t boil until well above the ambient room, but its volatility always demands respect.
Technical Specifications & Labeling
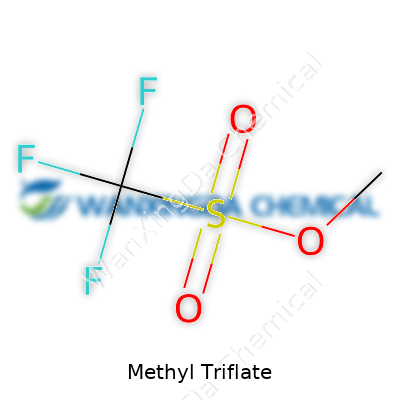
Chemists demand clarity when handling strong reagents like methyl triflate. The liquid gets sold in tightly sealed containers, usually with warnings about its acute toxicity and strong reactivity. Labels generally include basic identifying info: systematic name (methyl trifluoromethanesulfonate), chemical formula (CF3SO3CH3), and hazard symbols. There’s no need for fancy documentation to realize it’s not for casual handling—the acrid smell and advice to use well-ventilated fume hoods tell the story. Most specifications, like purity or moisture content, come from rigorous quality control, as impurities matter when a single contaminant can ruin carefully planned syntheses.
Preparation Method
The way to make methyl triflate calls for careful hands and accurate timing. Labs often prepare it by reacting methyl alcohol with triflic anhydride under anhydrous conditions, sometimes adding a base like pyridine to soak up stray acid. Temperature control stays critical—too warm, and side products creep in; too cold, and reactions crawl along. Industrial settings scale up by fine-tuning these same principles, using better containment and automation. What stands out here isn’t just the chemistry—it’s the sheer attention to operational discipline. This isn’t a product to make without deep chemical training and proper facilities.
Chemical Reactions & Modifications
Methyl triflate earns its keep through versatility in methylation reactions. It reacts with a wide range of nucleophiles—amines, phenols, even some anions that balk at milder reagents. Researchers use it to install methyl groups onto DNA bases, tricky coordination complexes, and tough-to-activate heterocycles. Sometimes, the goal involves more than just adding a methyl group. There’s a growing playbook for using it to activate electrophiles for further transformation, or as a leaving group for post-methylation modifications. Each time it reacts, it transforms a reluctant substrate into a more active intermediate, often opening new pathways that milder agents just can’t provide.
Synonyms & Product Names
Methyl triflate travels under many aliases. Some know it as methyl trifluoromethanesulfonate, others prefer the shorthand MeOTf. The IUPAC name sometimes crops up (trifluoromethanesulfonic acid methyl ester), especially in regulatory or patent documents. Regardless of the label, everyone recognizes the signature: a trifluoromethanesulfonate backbone with a methyl group hanging off. In practice, the short and sweet “MeOTf” does most of the day-to-day lifting, as chemists swap stories and procedures across bench tops and conference tables.
Safety & Operational Standards
Safety comes first, every time. Methyl triflate won’t let anyone get away with complacency. It attacks skin and eyes, damages lungs when inhaled, and can knock you flat with its vapor alone. Labs keep it in vented storage, handle it with gloves and goggles, and restrict use to those who understand its risks. Some forget just how rapidly it hydrolyzes in moist air, releasing acid fumes that make quick work of unprotected tissue. These facts show why it’s so vital to put process and training ahead of everything else—no shortcuts, no excuses. Emergency eyewash stations, spill kits, and thorough documentation all belong close by whenever this reagent comes out. Only experienced chemists handle it, and even they follow strict protocols.
Application Area
Anyone working in synthetic chemistry likely knows methyl triflate by reputation, if not from direct experience. Its strongest role appears in pharmaceutical research, where the stakes for clean, high-yield reactions run high. Medicinal chemists reach for it while building complex molecules—especially those with nitrogen or oxygen that need selective methylation. It doesn’t stop at drugs, either. Agrochemical developers tap into its power to build new pesticides, while material scientists use it to modify polymers or create electronic components. It even sneaks its way into the lab work behind advanced battery materials. What ties this range together is a drive for efficiency: when a methyl group needs to go somewhere fast and clean, methyl triflate often proves the tool for the job.
Research & Development
R&D teams have plenty of reasons to keep studying methyl triflate. Some look for safer or more sustainable alternatives, aiming to match its strength without its hazards. Others refine the reaction conditions, searching for ways to cut down on waste, reduce energy input, or improve selectivity. Researchers explore catalysts and additives that might tame its harshness, opening up target molecules once considered off-limits. Intellectual property teams keep an eye on novel uses and new process patents, reflecting a landscape where small changes in methodology can mean big competitive advantages. From my own work, I’ve seen how even incremental improvements in methylation chemistry can unlock huge downstream benefits—opening doors to new drugs, better coatings, or greener processes.
Toxicity Research
Not all power comes without cost, and methyl triflate pushes chemists to face its dark side directly. Inhalation causes immediate harm, and accidental splashes can cause severe burns. Toxicologists and lab managers keep it on the roster of chemicals that demand constant vigilance. Scientists in occupational health and regulatory bodies use animal studies and cellular assays to build a clearer picture of what long-term exposure might bring. They focus on markers of respiratory and dermal toxicity, weighing risks not only to lab workers but to the environment in case of spills or improper disposal. Each career spent around methyl triflate is a reminder of the balance between innovation and protection: we get value from the reagent, but only by staying fully aware of its hazards and never letting routine breed carelessness.
Future Prospects
The story of methyl triflate doesn’t stop at its current place in the synthetic toolkit. Chemists always seek cleaner, safer, and more sustainable processes. Green chemistry movements have spurred interest in reagents that do more with less waste or that break down into innocuous byproducts. While methyl triflate won’t disappear anytime soon, its role might shift as researchers develop better ways to achieve tough methylations—perhaps using biocatalysts or tunable reagents that cut down on toxicity. Startups and academics alike keep searching for ways to recycle reagents or create more benign equivalents, sometimes by looking to nature for inspiration. Anyone working on the next generation of pharmaceutical pipelines or high-value materials watches this space closely, knowing the lessons learned with methyl triflate will shape choices for decades to come.
A Strong Tool in the Chemist’s Kit
Methyl triflate doesn’t come up in everyday conversation. Most people outside the chemistry world haven’t heard of it, yet its influence stretches across pharmaceuticals, advanced materials, and the labs where new compounds are born. Popular among synthetic chemists, methyl triflate is known as a powerful methylating agent—meaning it helps tack a methyl group onto other molecules. That process might sound technical, but it has real-world consequences for the products coming out of chemical factories, research institutes, and even some medical labs.
Why It Matters in Synthesis
Adding a methyl group can change the whole behavior of a molecule, from its speed of action in the body to its stability on a shelf. Methyl triflate’s charm comes from its reactivity—it works quickly, usually at milder conditions than some older methylating compounds like methyl iodide or dimethyl sulfate, which are themselves toxic and tough to work with. This ability to get the job done faster and sometimes with fewer byproducts has pushed methyl triflate onto benches across research labs looking for cleaner processes.
A chemist I met during a graduate industrial placement once explained it like tuning up a recipe. “Sometimes, a methyl group is the difference between a failed trial and a miracle medicine,” he said. Turning an inert substance into something more active or absorbable can sometimes hinge on this kind of step. Methyl triflate, with its strong electronegative triflate partner, delivers methyl groups in a way that other reagents just can’t match for certain tricky compounds.
Impact on Pharmaceuticals and Materials Science
Drug discovery depends on the clever use of building blocks. Researchers often face the challenge of methylating sensitive compounds, especially when other methyl sources would lead to a toxic mess or damage the molecule. Methyl triflate steps in as a cleaner, more precise tool. Some antiviral, anticancer, and cardiovascular drugs have roots connected to processes where methyl triflate plays a crucial role. Similarly, in materials research, particularly in the creation of organic electronic materials, slight changes in structure often depend on reliable, selective methylation steps.
Risks that Shouldn’t Be Ignored
There’s another side of the story, and it isn’t pretty. Methyl triflate is highly toxic, a strong alkylating agent that can cause severe burns, and brings significant environmental concerns if spilled. It isn’t something anyone wants in their skin, their lungs, or near groundwater. Even small releases must be contained, and special training comes with using it safely. These risks have led some users to favor alternatives, but in niche cases where nothing else works, methyl triflate still finds its place.
Methyl triflate can’t be the answer to every chemical question. Safer alternatives, changing regulations, and the push for greener chemistry have sparked new research into gentler methylating methods. Still, for particularly tough transformations, especially in complex molecule synthesis, methyl triflate continues to prove its worth.
Moving Toward Safer Lab Practices
The dangers shouldn’t overshadow the role methyl triflate has played, but they force everyone handling it to stay updated on best safety practices and proper waste disposal methods. The next generation of chemists will likely see more stringent rules. Labs can invest in better ventilation, secondary containment, and personal protective equipment. Some universities have started emphasizing green chemistry principles, nudging young chemists to choose the least hazardous reagent whenever possible.
Watching that shift happen up close, through my years in academic and industrial labs, has given me respect for how one powerful tool can push a field forward, while also raising high-stakes questions about safety and sustainability. Methyl triflate carries that tension—a reminder that progress in chemistry often comes hand in hand with responsibility.
Thinking Twice About Methyl Triflate in the Lab
Methyl triflate might sound like just another specialty chemical, but this stuff grabs your attention for a reason. Chemists often rely on it to "methylate" things—they attach a methyl group to molecules, sometimes speeding up reactions that otherwise would crawl. It’s a powerful tool on the bench, no question. That power comes with a catch, though, and in this case, the catch is real risk.
Toxicity and Exposure Concerns
Methyl triflate doesn’t take much to threaten health. It stands out for its strength as an alkylating agent: that means it reacts easily with biological molecules, including DNA. This kind of reactivity plays a big role in toxicity. If you get it on your skin, it irritates. Inhalation? That’s a whole different ballgame. Gases or vapors can hurt lung tissue and potentially cause long-term problems.
Reports from chemical suppliers and published lab safety guides make it clear—exposure can lead to burns, tissue damage, even changes at the genetic level. Many labs treat methyl triflate in the same category as other notorious alkylating agents like methyl iodide or dimethyl sulfate. These chemicals have not only caused acute poisoning but also sparked regulatory changes due to cancer risks.
Why Proper Handling Beats Luck Every Time
It’s not common to run into methyl triflate outside of highly specialized industrial or academic labs, but for those who handle it, vigilance cannot slip. I’ve seen well-designed fume hoods and strict double-gloving routines become the bare minimum standard. Colleagues share stories of close calls—minor leaks turning into emergency evacuations, skins burns from just a tiny splash, headaches from a vapor sniff after a tiny container mishap.
While scientists understand the value of methyl triflate, they live with the tension that one misstep could spell trouble. Safety training shouldn’t feel like a checkbox; it’s a necessity. Emergency showers, type-SCBA breathing protection, and regular monitoring add layers of security that sometimes still feel thin. I remember an occasion when a co-worker needed medical attention after a droplet landed unnoticed on their wrist, burning through a regular lab coat with surprising speed.
Looking for Safer Alternatives
People in research circles don’t just accept the risks—they look for workarounds. Green chemistry reminds us that if something causes harm, there’s value in seeking a substitute. Other methylating agents sometimes take methyl triflate’s place. Some researchers are developing new protocols entirely, trying to skirt away from processes that rely on such dangerous substances.
All this brings to mind the importance of not letting speed or convenience override health. Labs don’t always have the luxury of unlimited resources, so supervisors focus on both technical and personal safety measures. Informing young chemists about these dangers matters more than ever, especially as pressure to innovate grows. Knowing about methyl triflate’s risks sets the stage for broader conversations about chemical safety in science and industry.
Responsibility Beyond the Lab Bench
Informed handling and transparent communication about methyl triflate's hazards matter to everyone in the lab—not just the people directly working with it. Accurate safety data, proper equipment maintenance, and honest reporting of incidents keep coworkers out of harm’s way. I’ve seen lab communities grow stronger by sharing lessons learned from real-life mistakes with compounds like this.
Hazardous chemistry can bring impressive scientific gains. That said, respect for substances like methyl triflate means knowing not just their uses but also their dangers, and refusing to compromise on that knowledge. Taking the time to teach, to question, and to improve safety around high-risk chemicals doesn’t slow progress; it protects everyone’s right to keep making it.
Understanding the Hazards
Storing Methyl Triflate calls for more than just finding an empty shelf. This compound brings real risk: extreme reactivity, a knack for corroding metal, and potent toxicity. Every chemist has heard stories about accidents with reactive chemicals—once you’ve seen the aftermath of a spill, safe storage stops being a checklist item and becomes daily habit.
This isn’t classroom theory. Methyl Triflate will release dangerous fumes if it meets moisture, so forget about leaving it anywhere humid or within reach of water taps. Breathing in the vapors can have serious health effects. Chemical burns are real if a careless moment lets a drop touch skin.
The Right Storage Approach
Temperature makes the first big difference. I learned early on that many organic reagents fall apart or become aggressive if they’re left in the open. Methyl Triflate should be kept cold—think refrigerator or cold room, but always below room temperature. Some labs use specialized solvent refrigerators, set away from places where people spend much time.
Ventilation matters. Fume hood storage isn’t a luxury—without solid airflow, even small leaks turn into emergencies. If you can’t dedicate a ventilated cupboard or cabinet, rethink whether it’s safe to keep the material at hand. Risks climb if fumes collect undetected.
Materials make a difference. Standard lab shelving—especially those with exposed metal fittings—won’t resist the corrosive edge of Methyl Triflate over time. Glass containers with PTFE (Teflon) lined caps earn their keep here. Polyethylene or polypropylene outer containers add a backup layer. I’ve seen what happens when a bottle cap degrades: Methyl Triflate escapes faster than most people expect.
Reducing Human Error
Teamwork keeps people safe. Labels are the first line of defense. Handwritten, fading marker notes won’t cut it. Clear labels stop people from grabbing the wrong bottle during a busy afternoon. Some labs run monthly audits on their shelves—this sounds tedious until you’ve dug through a neglected storage area.
Transport needs extra care. A colleague of mine once moved a sealed vial across campus in a regular box—bad idea. Anything this reactive deserves double containment and clearly marked transport containers. One lapse in judgment can ruin a project—or much worse.
Why Storage Choices Matter
Labs face tough pressure to work faster and cut corners. But poor storage risks more than wasted material. A single incident can shut down a research group, trigger costly cleanups, or lead to severe injuries. Researchers at several universities have faced major investigations after storage failures with less dangerous chemicals.
Safe storage for Methyl Triflate boils down to a respect for the chemical’s power and a commitment to the people in the lab. Managers shouldn’t leave anyone guessing. Regular training, real practice with spills, and honest review sessions strengthen habits. In my own experience, a few extra minutes spent checking that fridge or relabeling containers has paid off many times over.
Better Solutions for the Future
Reliable storage depends on both good habits and the right infrastructure. More labs turn to digital inventory systems, which alert staff before materials expire or containers fail. Investing in better containment tech and smart alarm sensors isn’t cheap, but the cost pales next to the price of cleanup or lost research time.
Regulations on reactive chemicals keep getting tighter, but common sense and teamwork carry just as much weight. Keeping Methyl Triflate cold, dry, locked, and well-labeled isn’t just safe—it keeps the lab running for everyone.
Methyl Triflate: One Mistake Can Change Lives
I remember the first time I stood in a chemistry lab and read the label on a small bottle of methyl triflate. The warning symbols looked intimidating — health hazard, environmental concern, tear-your-eyes-open kind of danger. Colleagues traded stories about splash incidents, hospital trips, and evaporating gloves. It didn't take a background in chemistry to figure out: methyl triflate isn’t something you ever want to touch with your bare hands or catch a whiff of.
No Room for Carelessness
This compound reacts violently with moisture and attacks living tissue on contact. Accidentally inhaling the fumes or splashing even a tiny drop can lead to lasting injury. One research assistant told me about a drop corroding through a brand-new workspace bench. Safety sheets call it a “potent methylating agent,” but that phrase doesn’t capture the panic in real accidents.
It makes sense why any lab dealing with methyl triflate brings in containment hoods, double gloves, and specialized training. Fume hoods don’t just keep the job tidy, they keep breathing zones clear. Face shields keep splashes away from eyes. Exhaust systems need regular maintenance, because a filter breakdown can endanger a whole building. Goggles and chemical suits sound like overkill until you hear someone coughing after a spill.
Disposal — The Weakest Link
I once saw a new lab tech turn to the sink after a botched experiment. The supervisor’s yell stopped him. Draining methyl triflate with water creates more danger, since it mixes with moisture to release acid and toxic gas. Flushing hazardous waste down the drain hurts more people than it protects, especially maintenance workers and water treatment staff. The story of a nearby city dealing with contaminated wastewater plants keeps resurfacing at lab conferences.
Safer Paths Exist
The right route for leftover methyl triflate starts with neutralization — treating it with a strong base in a controlled setup, often cooled with ice to slow the reaction. This isn’t work for just anyone. Trained waste handlers in specialized hazmat teams usually come in, package the neutralized residue, and send it for incineration at licensed facilities. Some countries leave the process to on-site destruction, using neutralization kits while suited up in full protective gear.
Documenting every step matters. Lax recordkeeping and skirting the regulated path draws heavy penalties, for good reason. Poor disposal hasn’t stayed a “lab problem.” Illegal dumping made headlines more than once, with traces of powerful methylating agents found near old research sites decades after closure. Cleanup costs reach into the millions and cancer statistics edge up. It only takes a small slip-up to break trust with the public, especially if cleanup teams catch traces in the groundwater.
Investing in Safer Habits
I’ve worked in labs tightening up paperwork, locking chemical cabinets, and enforcing double sign-off on disposal. These policies aren’t about bureaucracy; they keep people safe. Every training drill and recorded waste shipment adds up to fewer accidents and headaches. Solid support from management helps. Some labs bring in outside contractors for disposal — a bit more expensive, but it beats risking lives for the sake of a shortcut.
At the end of the day, respecting methyl triflate isn’t just about following a rulebook. It’s about protecting co-workers, communities, and our soil and water. Handling it right doesn’t earn awards, but those quiet days without accidents remind us it’s worth every ounce of caution.
A Closer Look at the Structure
Methyl triflate doesn’t show up on store shelves or in everyday products, but anyone working in synthetic chemistry will bump into its name sooner or later. Its formula—C2H3F3O3S—may not look flashy, yet this molecule packs power thanks to its chemical structure. Picture a methyl group (CH3–) bonded to a triflate group (–OSO2CF3). The carbon, oxygen, sulfur, and fluorine atoms connect together in a way that gives this compound both high reactivity and a place in countless organic syntheses.
The backbone of methyl triflate revolves around the strength and electron-withdrawing pull of the triflate group. The triflate (trifluoromethanesulfonate) anion draws away electrons, making the methyl end act like a live wire—ready to attach itself to any nucleophile (a fancy way of saying anything with an electron-rich site). In the lab, this trait means methyl triflate can deliver that methyl group almost like a courier. Chemists reach for it when other methylating agents feel sluggish or when selectivity makes the difference between a dead end and a productive reaction.
Why Chemists Care About This Molecule
I remember my organic chemistry professor discussing the need for reliable methylation reagents. As someone who tried, and sometimes failed, to get reactions to completion, I soon learned why methyl triflate stands out. It’s actually stronger than many classic methyl donors, such as methyl iodide. When a tough aromatic ring refuses to participate, methyl triflate steps up, often reacting under milder conditions and finishing in less time. This translates to higher yields and cleaner products, qualities anyone in the lab will appreciate after dozens of purifications.
The kicker comes in pharmaceuticals and materials science. Many modern drugs rely on the precise addition of a methyl group. That single carbon atom can change how a molecule behaves in the body: shifting potency, changing selectivity, or even deciding a drug’s safety. Methyl triflate’s reputation among chemists comes from its ability to tackle tricky sites, making it a behind-the-scenes player in the development of new medicines and high-performance materials.
Working With Dangerous Power
High reactivity rarely comes free. Methyl triflate is toxic, corrosive, and volatile. For years, safety warnings piled up around its use. In my own lab days, every time it came out of the refrigerator, the entire team paid attention. Goggles, gloves, fume hood—non-negotiable. Nobody wanted a whiff of that vapor or a spill on the scalp.
Safety procedures can’t be left as an afterthought. Detailed protocols, training, and investment in protective gear stand between success and disaster. Institutions are shaping new guidelines each year based on fresh accident reports. Some chemists now explore less hazardous methylation agents or work on encapsulation and automation to limit direct handling of methyl triflate. There’s also growing interest in green chemistry—developing safer and faster methods that reduce overall exposure and waste. Investment in training and a culture that rewards safe habits matter much more than slapping a warning label on a bottle.
Closing Thoughts
Methyl triflate’s structure gives it an edge, especially in specialized branches of synthetic organic chemistry. Every time someone struggles with a stubborn methylation, this compound brings answers—but demands respect in return. Smart protocols, staff know-how, and ongoing research point the way toward safer and more productive outcomes. For chemists in the trenches, that combination of power and peril defines the story of methyl triflate.