Methyl Trifluoroacetate: A Closer Look at Its Role in Modern Chemistry
Historical Development
Interest in organofluorine chemistry often traces back to the search for new building blocks in pharmaceuticals and agrochemical research. Through the mid-twentieth century, chemists pushed hard to develop unique molecules that could change how drugs, polymers, and specialist reagents were made. The story of methyl trifluoroacetate isn’t from just one discovery. It’s a product of synthetic ambition—a compound brought to light once fluorinated chemicals found their way into labs with new tools and more reliable supplies of fluorine-containing feedstocks. As fluorinated organics gained fame for their chemical toughness and distinct electronic influence, methyl trifluoroacetate proved its worth as a key intermediate. Looking back, research papers from the 1970s and 1980s show a growing curiosity about adding trifluoromethyl groups to molecules in order to push up metabolic stability and lipophilicity.
Product Overview
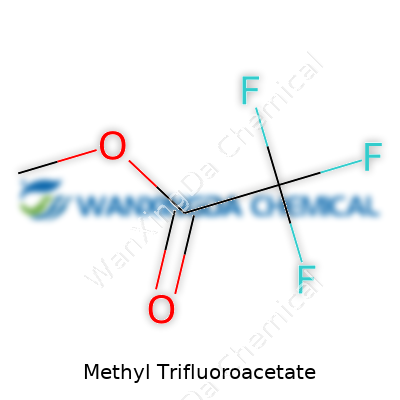
In practical terms, methyl trifluoroacetate stands out as a simple ester where a methyl group connects to a trifluoroacetyl backbone. Chemists see value in this molecule for both the fluoroalkyl group and the high reactivity of the ester function. Its small size helps it slot into complex syntheses, enabling both large-scale industrial work and detailed academic studies. Anyone working in synthesis or analysis likely knows its sharp, distinctive odor and rapid evaporation. The chemical’s clear, colorless appearance belies the punch it brings to a diverse range of routes and applications.
Physical & Chemical Properties
Methyl trifluoroacetate exists as a volatile liquid, boiling at a relatively moderate temperature for a fluorinated ester. Its volatility comes from the methyl ester, while the electron-withdrawing trifluoromethyl group blocks easy attack from nucleophiles. Water doesn’t mix with it well, but the compound dissolves smoothly in organic solvents like diethyl ether or dichloromethane―those mainstays of the lab. You get a strong hint of its acid chloride origin when you feel how moisture reacts with it, sometimes releasing the signature smell of trifluoroacetic acid. Its refractive index and density fall right in line with other light, halogen-filled esters. In NMR, the CF₃ shows up downfield, a dead giveaway to its identity.
Technical Specifications & Labeling
Standard labeling calls out methyl trifluoroacetate’s need for careful handling. Bottles carry warnings for respiratory irritation and the risk of skin and eye exposure. Because high purity matters most in synthesis, suppliers typically offer assay values above 98 percent. Impurities—including hydrolyzed acid or methyl alcohol—can mess with sensitive reactions, so chemists check each lot before scaling up. Transport rules classify it as a hazardous material due to volatility and health risks, and storage usually means keeping it sealed tight under inert gas at cool temperatures.
Preparation Method
While a few routes exist for making methyl trifluoroacetate, most begin with trifluoroacetic acid. One common lab method uses acid chloride formation with thionyl chloride or oxalyl chloride, then reacts that intermediate with methanol to build the ester. On the industrial side, methylation might use dimethyl sulfate or methyl iodide in the presence of a base, following careful quenching to control side products. Purification involves distillation at reduced pressure due to the compound’s heat sensitivity and to keep degradation low. No one gets away with sloppy technique at this stage, as trace acid or water kill yield and foul up further transformations.
Chemical Reactions & Modifications
Chemists value methyl trifluoroacetate for how it handles – both the toughness from the trifluoromethyl group and the reactivity from the ester linkage. Its big draw comes in acting as a trifluoroacetylating agent. The molecule slots into reactions that need selective acylation, such as protecting amines or alcohols with a trifluoroacetyl group. That protection resists strong alkali and most oxidants, but comes off with milder bases or nucleophiles. If you’re making α-trifluoromethyl ketones, esters, or even pharmaceuticals, this reagent allows for careful addition of the CF₃CO- unit. Some folks use it as a starting point for making trifluoroacetic acid or more elaborate derivatives with extra functional handles. The electronic pull of CF₃ also gives it role in developing new catalysts and ligands, especially where electron-deficient behavior shifts reactivity in transition metal complexes.
Synonyms & Product Names
Anyone digging through catalogs will find methyl trifluoroacetate listed under several similar names: methyl trifluoroacetate, trifluoroacetic acid methyl ester, and methyl ester of trifluoroacetic acid. Older literature or foreign-language papers may use obscure variants, but those three cover most references in major databases. Suppliers stick to standardized names for regulatory compliance, and cross-referencing across CAS numbers avoids confusion during ordering.
Safety & Operational Standards
Working with methyl trifluoroacetate means treating it with real respect. I remember watching colleagues use full-face respirators when a fume hood failed during a scale-up—a few milliliters out, and the sharp vapors irritate noses and lungs fast. Direct skin contact burns and doesn’t wash off easily, thanks to the compound’s rapid absorption and resistance to mild soap. Labs keep spill kits handy, with inert absorbents instead of water, since hydrolysis makes an acidic mess. Industrial plants rely on closed systems and trained handling protocols. Employees go through annual safety drills, reinforcing eyewash station use and correct glove disposal. Regulatory agencies rate this chemical as hazardous, punching up the need for consistent ventilation, gloves, goggles, and written procedures even for small jobs.
Application Area
The most active use for methyl trifluoroacetate pops up in organic synthesis, both in academic research and commercial development. People making new pharmaceuticals lean on the trifluoroacetyl group as a protecting group for nitrogen or oxygen atoms. That blocking lets them power through multi-step syntheses without unwanted side reactions from free amines or alcohols, then pull off the group at the right step. Agrochemical researchers use methyl trifluoroacetate when they need to toughen up molecules for stability in harsh field conditions. Polymers and advanced materials that need lightness, chemical resistance, and electrical insulation often tap trifluoromethyl motifs, and this ester serves as a cheap feedstock for adding those branches. Elsewhere, analytical chemists turn to methyl trifluoroacetate to derivatize organic acids or alcohols, helping boost volatility and separate tricky compounds by gas chromatography or mass spectrometry.
Research & Development
Each year, journal articles reveal new angles for methyl trifluoroacetate in developing building blocks, drug candidates, and electronic materials. Some teams focus on green chemistry—finding ways to recover and recycle the ester or cut down genotoxic waste. Others fine-tune reactions with transition metals, hoping that the trifluoromethyl group will tune catalyst selectivity. Process chemistry groups keep working on safer, higher-yielding routes for esterification, aiming at lower temperatures, faster rates, or easier purification. For a while, anyone trying to miniaturize reaction setups has liked methyl trifluoroacetate because it responds well to microfluidic control, shedding new light on process optimizations. Based on the rich flow of patents, major chemical companies see a bright future in specialty fluorine containing compounds, and methyl trifluoroacetate runs as a key intermediate in several growing drug and materials pipelines.
Toxicity Research
Researchers keep a close watch on the toxicity profile of methyl trifluoroacetate, particularly because of its penetration through skin and mucosa. Studies show acute exposure swells up respiratory passages and damages lung tissue, especially in higher concentrations. Animal models point toward rapid absorption and conversion to trifluoroacetic acid, which carries its own risks of metabolic disturbance. Chronic exposure remains under investigation, with scientists probing for organ damage or subtle neurological effects. In regulatory reviews, chemists and health physicists debate acceptable thresholds, often erring on the side of extreme caution. Even trace levels in the environment prompt scrutiny, given the stability and bioaccumulation tendency of perfluorinated products. Workplaces insist on regular air and surface monitoring, while landfill and incineration treatments follow strict protocols to cut down environmental leaks.
Future Prospects
Looking forward, methyl trifluoroacetate stands to benefit from attention in pharmaceuticals, electronics, and sustainable chemistry. On the pharma side, new drugs depend on fine-tuned fluorine chemistry to clear clinical hurdles around metabolism and selectivity. Trends point to more trifluoromethyl groups in both traditional small-molecule drugs and rising peptide or oligonucleotide classes—leaning on methyl trifluoroacetate for intermediate steps. The continuing search for organic semiconductors and light, tough polymer films keeps it in demand for material science. As governments and scientists push to cut the environmental impact of fluorinated chemicals, research may steer into better recycling and recovery methods for methyl trifluoroacetate and its derivatives. The balance between high-performance chemistry and responsible stewardship shapes the conversations among researchers, regulators, and end-users, with methyl trifluoroacetate right in the spotlight as an example of both the promise and the challenge of modern organofluorine technology.
More Than Just Chemistry Jargon
Methyl trifluoroacetate sounds like another mouthful from a university text, but it shows up in a lot of places behind the scenes. In my own work in academic labs, and later in the pharmaceutical industry, this small molecule comes up more often than most people would expect. Its main claim to fame comes from how handy it proves as a reagent in organic synthesis. Folks who spend time in chemical research rarely overlook its usefulness for introducing trifluoromethyl groups into molecules, and this has a direct link to new medicine development and material science.
Real Uses, Real Impact
Lab workers and industrial chemists often reach for methyl trifluoroacetate to build complicated molecular structures. Companies depend on it to help cobble together new drugs and advanced agricultural chemicals. Trifluoromethyl groups, put in place by reagents like methyl trifluoroacetate, can change how a drug acts in the body. These groups often boost the stability or effectiveness of medicines, and can make a difference between a molecule that helps people and one that fizzles in clinical trials.
Beyond pharmaceuticals, material scientists use this compound in research on specialized plastics and coatings. Adding fluorine atoms can shut down unwanted reactions, or help produce materials with odd properties—think Teflon-type nonstick qualities or insulating foams. In the world of electronics, it sometimes helps create unique polymers used in circuit board coatings, a process I got to see up close in a manufacturing setting.
Health, Safety, and the Human Factor
This compound doesn’t show up on supermarket shelves, for good reasons. Methyl trifluoroacetate can be hazardous, so only folks with proper training and protective gear should handle it. Schools and chemical companies take this seriously. Years ago, I saw more than one lab accident avoided thanks to clear labeling and strong safety culture. Mistakes around these chemicals mean real consequences, so investing in good lab training matters just as much as fancy equipment. The focus should land not only on what new science enables but also how to keep vulnerable workers safe.
On a larger scale, environmental impact arises as a concern. Fluorinated compounds often stick around in the ecosystem and resist breaking down. The issue of so-called “forever chemicals” comes from decades of unchecked use. Regulators keep a close watch now, and scientists look for greener alternatives. Any industry using methyl trifluoroacetate must weigh the convenience of its chemistry against the costs to the planet. This push for responsibility lines up with society’s rising standards for science that puts health and the environment first.
Smarter Solutions and Responsibility
Labs can make smarter choices about how they use and dispose of methyl trifluoroacetate. Green chemistry offers cleaner pathways, limits waste, and cuts the risks of exposure. Engineers design better containment and handling systems, lowering accidental spills or leaks. Transparency about sourcing, use, and disposal, as well as supporting research into alternatives, keeps the conversation moving forward.
At the end of the day, compounds like methyl trifluoroacetate show how modern science depends on small pieces working together. The science world keeps looking for safer, newer ways to do its job, and that search includes making sure chemical tools work not just for innovation, but also for the people behind the bench and the community outside lab walls.
The Practical Realities in Chemical Storage
Methyl Trifluoroacetate isn’t a household name. Yet, in research labs and chemical plants, it plays a role as a specialty reagent. Sometimes, folks outside chemistry circles ask, “Why all the fuss about storing chemicals like this?” Honestly, mishandling even small volumes can wreak havoc. I’ve watched greenhorn lab techs underestimate the volatility of this clear liquid, only to see them frantically scrub spills. Over nearly two decades working in academic labs and consulting for industry, I’ve picked up a healthy respect for proper storage conditions.
Key Risks: Flammability and Instability
Most sources agree, methyl trifluoroacetate is volatile and flammable. Leaving it near ignition sources or in a sunny window can turn a mundane day into a disaster. One of the worst blunders I’ve witnessed was someone stowing it above eye level, right next to a water heater. Not only do the vapors catch, they also sneak into every crack. If the cap isn’t tight, expect a stinging, acrid smell to travel through shared workspaces. Keeping this chemical away from heat and sparks is common sense, but discipline in the lab often slips when people get busy.
This chemical also reacts with water and strong bases. Moist air seeps into badly sealed bottles. Finding the bottom of a container crudded up with decomposition, or realizing reactions have shifted yields because of poor storage, teaches you to keep it bone dry and tightly shut. Plenty of times, I’ve watched new chemists reach for a reagent only to find it’s gone off or lost potency. Losses like that add up, both in ruined reactions and wasted funding.
How the Pros Store Methyl Trifluoroacetate
Walk into any well-run chemistry lab, and you’ll see small quantities of methyl trifluoroacetate resting in flammable storage cabinets. These cabinets shield bottles from direct light and help contain vapors. Storing it cool, below room temperature if possible, slows reaction and evaporation rates. Cold rooms and refrigerators (explosion-proof, not the one in your break room fridge) keep volatility in check. Between the cool air and locked metal cabinets, risks drop.
Proper labeling and sturdy, airtight containers prevent leaks. Glass works, if the closure is secure — I’ve seen Teflon-lined caps hold up better than plastic ones. Once I watched a shipment show up in a recycled bottle with a loose lid; nobody could ignore the headache-inducing fumes. Real containers save real safety headaches.
Why Policy and Training Matter
Rules exist for reasons. Regulations about flammable material storage reflect more than paperwork; they come from a long history of accidents. Training isn’t just drudgery. People remember the why, not just the what, once they see or read about fires or ruined work. I believe every new lab member needs a hands-on walk-through with chemicals like this one, not just a signature on a form. In smaller businesses, creating that culture falls to supervisors who lead by example. Shortcuts in handling usually show up in near-misses and lost time, so constant reminders and refreshers keep everyone sharp.
Better Solutions Start Small
No magical system fixes storage woes overnight. Clear protocols, real training, and small investments in quality containers and cabinets save money and lives. I’ve worked places where people shrugged off safety for years, right up until insurance rates spiked or real injuries occurred. People only learn the value of careful chemical storage — especially for risky liquids like methyl trifluoroacetate — through practical habits. Set the example, lay out the facts, and keep everyone honest, and safer workplaces follow.
The Conversation Around Chemical Hazards
People who spend time working in a lab or managing chemicals in industry know that some substances demand respect, no matter how regular they seem. Safe use of chemical compounds stands on clear facts and lessons learned from experience—not on misplaced confidence. Methyl trifluoroacetate, for example, appears in many chemical syntheses and pharmaceutical research, but the story behind its hazards doesn’t always get enough daylight.
What We Know About Its Risks
With a molecular structure that includes a reactive methyl group and a trio of strongly electronegative fluorine atoms, methyl trifluoroacetate doesn’t belong in the household cabinet. It gives off a sharp smell and, in high concentrations, can irritate the nose, throat, and lungs. From what’s shared in safety data sheets and academic reviews, prolonged exposure, especially through inhalation or skin contact, brings a fair share of trouble.
Breathing in vapors may lead to coughing or burning sensations in the chest—not unlike what happens with other volatile organic compounds. Liquid contact can irritate the eyes or burn the skin. Here in the lab, extra care goes toward keeping it off the hands and far from the face. Gloves, eye protection, and fume hoods become the norm, not an afterthought.
On a chemical level, methyl trifluoroacetate’s reactivity comes mostly from the trifluoromethyl group. That same feature that makes it valuable for drug and polymer synthesis also means it doesn’t always play nice with the rest of the world. If released without control, this compound can contribute to environmental problems, including formation of persistent organic pollutants. The trifluoromethyl group tends to resist breaking down, which brings us closer to that big challenge—forever chemicals in the environment.
Regulatory Gaps and Real-World Experience
Chemical regulations often lag behind science and workplace experience. In some regions methyl trifluoroacetate isn't specially regulated, which might trick people into underestimating the harm it could deliver. Yet, anyone who’s ever had to clean up after a spill or breathed in a poorly vented cloud knows it’s no ordinary reagent.
I remember an incident early in my research career, in a shared university lab, where a small spill led to eye and throat irritation for everyone at the bench. We cleaned up fast, but the lesson stuck: even a small volume turns into a big problem without the right ventilation or equipment. Seasoned chemists stress this point all the time—a chemical’s hazard is more than a number on a spreadsheet, but something you feel right away if you drop your guard.
Practical Steps Toward Safer Use
Reducing risk does not happen just by reading the labels. It means providing proper training, enforcing the use of fume hoods, and encouraging a culture of reporting incidents without fear. At workplaces and universities, finding easy access to personal protective equipment and reliable spill controls makes a noticeable difference. Disposal matters, too—nobody wants more persistent chemicals ending up in local water supplies.
Better tracking and reporting, along with clearer rules and investment in less hazardous alternatives, benefits the people handling these substances and everyone else down the line. Dialogue between researchers, safety officers, and regulators helps uncover loopholes and catches problems before they hit the news. In the end, respect and vigilance go further than any single sheet of rules.
The Formula That Matters: C3H3F3O2
Methyl trifluoroacetate isn’t just another tongue-twisting chemical name. Its formula, C3H3F3O2, packs a punch far beyond high school chemistry. In my years talking with chemists at universities and in startup labs, meaningful advances start not in the abstract, but with the humble facts, the type written on the sides of reagent bottles. C3H3F3O2 tells a story that goes straight to the molecular heart of both challenge and progress in synthesis.
How a Simple Ester Shapes Innovations
This molecule holds a trifluoromethyl group that gives it unique leverage in chemical reactions. Organic chemists seek out reagents like this when they want to add a CF3 unit, shifting physical and biological characteristics in significant ways. Products ranging from crop protection agents to advanced pharmaceuticals start with little building blocks like methyl trifluoroacetate. In the real world, that often means better outcomes for patients and farmers alike.
Seeing its effect firsthand helps me understand why trifluoromethyl esters deserve respect. They improve metabolic stability, making a drug candidate less likely to break down too quickly. For chemists chasing the next antiviral, that’s not magic. That’s the strategic use of C3H3F3O2.
Spotlight on Safety and Responsibility
Every chemist who has worked in a lab appreciates the lesson that powerful molecules come with responsibilities. Methyl trifluoroacetate isn’t something you’d want to splash around. It is volatile and reacts under the right conditions with nucleophiles or water, releasing fumes you wouldn’t want to inhale. The industry keeps tight ships: clear ventilation, proper labeling, gloves, and goggles are non-negotiable. Research facilities take these steps not because somebody once said so, but because ignoring them invites chemical burns or dangerous exposures.
Education about safety works best through showing rather than telling. In my early lab days, a colleague ignored a cracked stopper, and one whiff later, everyone in the fume hood area remembered why protocols matter. Chemists keep safety data close and rely on proven routines rather than luck.
From Raw Materials to Real-World Solutions
Demand for methyl trifluoroacetate reflects the larger shift toward fluorinated compounds across science and industry. The fluorine atoms in this molecule open doors that hydrogen or chlorine never could. Agrochemical designers reach for these compounds to build weedkillers or fungicides that break down less quickly, improving harvest yields and reducing application costs.
Drug designers lean into this field for new treatments against stubborn viruses or cancer. Methyl trifluoroacetate isn’t solving these problems alone, but it forms a reliable piece of the puzzle. Its structure allows for selective modifications, giving researchers the toolbox they need to try out new ideas on a reasonable budget.
Smarter Practices for a Safer Tomorrow
There’s a growing call for green chemistry across the globe. Labs now rethink how much of each reagent goes down the drain, how much energy gets wasted, and what the end-of-life plan looks like. Methyl trifluoroacetate, because it is a staple, stands at the center of this shift. Instead of old habits, researchers look for better containment, smarter cleanup, and creative recycling strategies.
Caring about a chemical’s formula isn’t just academic. For those who work with these building blocks, understanding C3H3F3O2 means taking every step with purpose: safer workspaces, less waste, and science that gets trusted for the right reasons.
Everyday Chemistry Demands Respect
Every so often, folks who work in labs or chemical facilities meet something that reminds them why their protective gear matters. Methyl trifluoroacetate stands out as one of those chemicals. It’s got a bite. Even people who have clocked years handling tougher acids or solvents don’t take these fluorinated chemicals lightly. Once, pulling open a container of this material, a coworker got a whiff—a sharp, stinging reminder that no protocol should be skipped, no matter how rushed a day feels.
Gloves and Goggles Are Non-Negotiables
I’ve watched seasoned chemists toss off stories about the odd acetone spill, thinking back on how unprotected skin just gets a bit dry after. Methyl trifluoroacetate is cut from a different cloth. Just a splash burns and lingers. The gloves come out before the bottle even leaves storage. Nitrile or even thicker material makes a difference—latex wilts to some solvents. Face shields, not just regular goggles, support the eyes. After seeing what a single drop can do, respect for the right gear grows, especially because many solvents ignore the “wash with water for 15 minutes” cliché. When this ester goes airborne, the lungs catch the worst of it. Fume hoods are more than furniture; they’re lifelines.
Storage: Out of Sight Yet Front of Mind
Plenty of chemicals wind up stashed on the wrong shelf, and bad things tend to happen sooner or later. Methyl trifluoroacetate belongs in a cool, ventilated spot. The tighter the seal, the less worry about fumes sneaking out and making everything smell like rotten apples meets hospital corridor. I’ve heard of people returning after the weekend, only to find stray vapors had left a residue film, even when no one touched the bottle since Friday. Neighbors matter, too. Anything reactive or with a bit of moisture can make things spiral—once water gets near methyl trifluoroacetate, you risk generating corrosive fumes like trifluoroacetic acid, an eye and lung destroyer.
What Happens If Things Go Wrong?
In most accidents, scrambling wastes time. Knowing what to do without second-guessing saves skin, eyes, and sometimes lives. If a spill happens, ventilation steps up first; getting the area clear and letting only trained people clean. No one pats themselves on the back for improvising with paper towels. Sometimes it means evacuating the area, calling in the experts, and filing a full incident report. Communication and training can be life savers.
Training: The Real Foundation
Someone joining a research team or chemical plant rarely gets an introduction to the most hazardous corners of their workplace until things get hairy. I think back to my first week working with fluorinated compounds—no amount of textbook knowledge prepared me like hands-on training did. Mandatory refreshers, not just once yearly, help keep everyone alert. At well-run facilities, sharing stories about close calls keeps the collective memory fresh, reminding new hires and old-timers alike what’s at stake.
Safer Practices, Stronger Outcomes
Handling methyl trifluoroacetate isn’t just a routine checkbox. Treating it with the seriousness it commands means learning, equipping, and storing correctly. Skipping a glove, working outside the hood, leaving a cap loose—one shortcut and trouble finds its way in fast. Chemical safety starts with hard-learned lessons and continues with stubborn attention to detail. This commitment protects not only the worker, but everyone who shares the workspace.