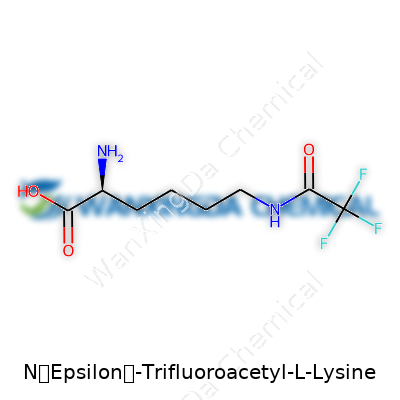
N-(Epsilon)-Trifluoroacetyl-L-Lysine: A Closer Look at This Niche Amino Acid Derivative
Historical Development
Science keeps pushing the boundaries, always searching for new ways to create useful molecules. Among these, N-(Epsilon)-Trifluoroacetyl-L-Lysine stands out in the world of chemical biology and protein engineering. A few decades ago, only a handful of chemists experimented with modifications on proteinogenic amino acids. The drive to unlock new chemistries inside living cells gave birth to modifications such as the trifluoroacetyl group, making this lysine derivative more than just a lab curiosity. Each tweak, each new variant, opens up a fresh avenue for biological study or synthetic challenge. Over time, this compound earned respect as chemists and biochemists harnessed its properties to explore protein modifications, structure–activity relationships, and new analytical tools to measure biological processes with greater precision.
Product Overview
N-(Epsilon)-Trifluoroacetyl-L-Lysine falls within a group of modified amino acids used to probe and influence protein function. The trifluoroacetyl group attached to the lysine side chain introduces unique electronic and steric effects that standard lysine cannot provide. In my own work, selecting the right derivative is like choosing the correct wrench from a well-stocked toolbox, and this particular molecule functions as more than just a building block—it’s like a switch that can change the behavior of larger biological molecules around it. When incorporated into peptides or proteins, it can block natural lysine reactivity, mimic post-translational modifications, or give researchers a distinct handle to study how small changes in structure lead to big shifts in activity.
Physical & Chemical Properties
A compound’s structure shapes how it behaves. With fluorine’s electronegativity, the trifluoroacetyl cap brings stability, resist oxidation, and not easily hydrolyzed under biological conditions. The lysine backbone remains intact, but the addition shifts the overall polarity and opens doors for chemical labeling or selective recognition. While most standard amino acids dissolve effortlessly in water, this derivative stands out due to the altered solubility profile imparted by the bulky and electronegative trifluoromethyl group. It's solid under standard lab conditions, and the distinct chemical shift from fluorine gives clear signals in NMR spectrometry, which has helped countless researchers track and confirm peptide modifications during synthesis.
Technical Specifications & Labeling
Once upon a time, working with rare amino acid derivatives meant accepting a certain level of ambiguity about purity and labeling. These days, the standards have tightened up considerably, driven by the demands of pharmaceutical research and academic rigor. Companies make an effort to provide specifications, including optical purity and minimal racemization, indicated clearly on each vial. From my own experience, reliable purity saves months of troubleshooting, removing doubts about whether a failed reaction comes down to poorly characterized material or a deeper problem with the chemistry itself. Even in small-batch synthesis, clear labeling about origin, storage temperature, and concentration serves as a lifeline for reproducibility.
Preparation Method
Creating N-(Epsilon)-Trifluoroacetyl-L-Lysine doesn't take magic, but it does call for precise chemistry. Synthesis starts by protecting other reactive groups on the lysine before introducing the trifluoroacetyl group selectively to the epsilon amino group. Standard coupling agents or acylation protocols handle the job, and after purification, most routes leave the main backbone untouched. Solid-phase peptide synthesis benefits from these protected lysine derivatives, because selective deprotection unveils the trifluoroacetyl pattern only when and where it’s needed. During my graduate work, running these syntheses made me deeply aware of how efficient protection–deprotection cycles carve the path to successful large-molecule assembly in either academic or industry labs.
Chemical Reactions & Modifications
Chemists use this molecule as a tool for customizing protein surfaces or probing enzymatic specificity. The trifluoroacetyl group resists many common deacetylase enzymes, so researchers inject this version of lysine to study epigenetic regulation or visualize protein turnover in cells. With a robust chemical signature, it enables “click” chemistry or selective tagging with fluorinated agents for imaging studies. In the hands of a creative researcher, it becomes a chemical handle to create conjugates, design enzyme inhibitors, or prevent undesired reactions during multi-step peptide assembly. Years ago, tinkering with these modifications taught me that each protecting group or side-chain tweak could make or break the entire synthesis, and nowhere is that more true than with carefully crafted amino acid derivatives.
Synonyms & Product Names
Experts refer to this compound with names like N6-Trifluoroacetyl-L-lysine or ε-TFA-Lys, but regardless of which label shows up on a supplier’s catalog, the chemistry remains consistent. Some scientific papers might shorten it to Tfa-Lys, particularly in peptide sequences, while database entries include systematic IUPAC nomenclature. Keeping track of these synonyms helps avoid confusion and wasted time when sorting through the literature or ordering from vendors, especially since even a single misplaced trifluoromethyl can derail a synthesis or research project.
Safety & Operational Standards
Lab safety matters, even for materials with established use. While this derivative doesn’t carry the dramatics of toxic gases or biohazardous agents, handling any trifluorinated compounds means keeping a keen eye on ventilation and minimizing skin exposure. During chemical synthesis, splashes or accidental inhalation should never be shrugged off just because the compound looks harmless. Gloves, goggles, and good hygiene treat every material with respect, and clear documentation ensures anyone picking up the bottle gets all the facts. Rigorous safety protocols benefit not only researchers but also the wider community, helping build trust in scientific integrity and keeping accidents to a minimum.
Application Area
Researchers use N-(Epsilon)-Trifluoroacetyl-L-Lysine across multiple fields. In protein engineering, the modified lysine plays a role in exploring the boundaries of enzyme active sites or in reprogramming proteins to resist natural degradation pathways. Analytical chemists take advantage of its strong fluorine signal to validate peptide synthesis or to build imaging agents for mass spectrometry or magnetic resonance studies. The altered reactivity makes it a natural fit for studying deacetylase-resistant modifications in histone biology, opening windows into gene regulation and cellular memory. I've seen firsthand how a well-placed modified amino acid changes the outcome of binding studies, inhibitor screens, or cellular assays, providing answers that natural amino acids alone can’t deliver.
Research & Development
Science feeds on new questions, and modified amino acids bring a wealth of them. Recent years saw a sharp increase in custom peptide libraries featuring protected lysines like trifluoroacetylated variants. Some groups aim to design better therapeutics by dialing the stability or charge on key amino acids, and N-(Epsilon)-Trifluoroacetyl-L-Lysine features prominently in those efforts. Ongoing research also looks at optimizing its incorporation through novel enzymes, non-canonical genetic codes, or novel orthogonal translation systems. For those working at this interface, every successful experiment opens the door to smarter medicines or deeper insights into life’s molecular alphabet.
Toxicity Research
Any time a chemical strays from the natural menu, toxicity becomes a reasonable concern. Studies in both in vitro and in vivo models typically look for signs of cytotoxicity or unexpected metabolic breakdown products. For trifluoroacetyl groups, the main risks often stem from release of trifluoroacetic acid or metabolic trapping in cellular compartments. While reported toxicity remains limited at the concentrations used in lab studies, larger-scale applications or therapeutic contexts always call for rigorous vetting. Reviewing toxicity studies helps guide responsible use, shaping not only safe lab practices but also informing regulatory standards for wider deployment in diagnostics or drug development.
Future Prospects
Interest in N-(Epsilon)-Trifluoroacetyl-L-Lysine and related amino acid derivatives keeps rising, fueled by the hunger for smarter drugs, more detailed probes, and deeper understanding of protein function in disease. The rise of synthetic biology only adds momentum, where designers insert non-natural building blocks into living systems, effectively rewriting the rules of biology itself. If we want to tackle emerging diseases, engineer new materials, or understand life at a higher resolution, then these tools become indispensable. Investment in safer synthesis, smarter application, and ongoing dialogue about risks will steer future developments. From my vantage point, this field blends scientific creativity with technical rigor in a way that few others match, and each breakthrough with trifluoroacetylated lysine stands as another milestone in the ongoing story of molecular design.
Looking Deeper Into a Modified Amino Acid
Out of the many small molecules that find their way into research labs, N(Epsilon)-Trifluoroacetyl-L-Lysine stands out for a useful trick: it lets scientists study life’s machinery with more precision than ever before. This compound, essentially a lysine molecule shaped with a trifluoroacetyl group, might not have the widespread recognition of penicillin or aspirin, though in the world of biochemistry, its role has been growing steadily.
Unlocking Protein Mysteries
In labs around the world, researchers use N(Epsilon)-Trifluoroacetyl-L-Lysine to explore how proteins work at the chemical level. Proteins are not static objects; they often change character through modifications called acetylations. Regular lysine in a protein sometimes gets tagged with an acetyl group, and this tweak can change the entire shape or behavior of a protein. The trifluoroacetyl tag serves as a chemical tool that stands in for the usual acetyl group, but with a twist: its trifluoromethyl group makes it easier to spot using analytical methods, especially nuclear magnetic resonance (NMR) and mass spectrometry.
By swapping in trifluoroacetyl groups, scientists map out which proteins have changed and at what spot. This allows close examination of how modifications affect protein function, folding, and interactions. For diseases linked to misbehaving proteins, such as cancers or neurological disorders, even small details uncovered through research with this compound can point the way to new drug targets or biomarkers.
Building Tools for Chemical Biology
The use of N(Epsilon)-Trifluoroacetyl-L-Lysine also highlights the creativity of chemical biology. By adding this modified lysine into synthetic peptides or engineered proteins, researchers create tools that reveal hidden activities inside living cells. For example, enzymes that sniff out and remove acetyl groups—like certain deacetylases—often react differently to the trifluoroacetyl tag. This reaction tells scientists which enzymes handle which modifications and helps design drugs that block or tweak these enzymes in a precise way.
Compared with traditional biochemical approaches, using trifluoroacetylated residues can deliver sharper results. The chemical’s unique fluorine atoms do not interfere with regular cell processes, but their distinctive signal lights up in many types of imaging systems. This precision means less noise and confusion when reading results, which saves time and money while improving the reliability of discoveries in everything from drug development to fundamental protein research.
Catching the Eyes of Drug Hunters
Pharmaceutical companies have started to follow the lead set by academic researchers. Compounds similar to N(Epsilon)-Trifluoroacetyl-L-Lysine offer templates for building drugs that mimic or block protein modifications. These efforts may take years to pay off, but the seeds planted by this chemistry promise new ways to control diseases at the source, right at the level of a single amino acid.
Room for Growth—and Vigilance
Science works best when researchers keep a clean, careful approach. Using synthetic amino acids in experimental setups demands thoughtful design and solid testing. Sloppy work could lead to wrong conclusions or missed warning signs. Luckily, stricter standards and improved analytical methods keep progress steady.
For students and early-career scientists, learning about molecules like N(Epsilon)-Trifluoroacetyl-L-Lysine can open doors to fields blending chemistry, biology, and medicine. Understanding these tools sets the stage for real progress—translating bench research into tomorrow’s medicines, diagnostics, and therapies that reach outside the walls of any one lab.
Having worked in research labs for years, I’ve seen more spills, degraded samples, and ruined batches than anyone would like to admit. N(Epsilon)-Trifluoroacetyl-L-Lysine isn’t your garden-variety amino acid derivative. The delicate structure and sensitive side groups demand careful handling, not just to protect investment, but to maintain the consistency science depends on.
Respect for Chemistry: Temperature and Environment
Chemists learn early that small tweaks—temperature, moisture, even stray sunlight—can spell disaster during storage. N(Epsilon)-Trifluoroacetyl-L-Lysine, with its trifluoroacetyl group, takes that lesson up a notch. Most labs store it cold, typically in refrigerators set between 2–8°C. Research shows low temperatures slow down hydrolysis and other processes, which guards against breakdown. Leave it on a benchtop and degradation starts faster than most busy researchers realize.
Dryness must remain top-of-mind. Desiccant-packed containers do more than reduce the risk of clumping; they essentially lock out the ambient humidity that can cause decomposition or promote nasty byproducts. Moisture acts as a hidden enemy, especially over longer storage periods. This is where attention to detail pays off: well-sealed containers, stored in low-humidity environments, keep quality high and variability low.
Light, Contamination, and Lab Realities
Direct sunlight, or even ambient bench lighting, can sometimes hit more reactive chemicals harder than we anticipate. Over the years, I saw predictable yellowing and breakdowns when samples sat near sunny windows. For N(Epsilon)-Trifluoroacetyl-L-Lysine, amber vials or opaque containers add another barrier. Light-protection isn’t just about avoiding color changes—it’s about making sure the structure stays true, batch after batch.
Anyone sharing bench space knows how easy cross-contamination sneaks in, especially with compounds that show up in trace analysis. Dedicated scoops and gloves help, but the biggest safeguard comes from keeping lids tightly closed and handling only in clean spaces. Each extra hand in the jar increases risks, so making storage containers easy to handle—big clear labels, sturdy screw-tops—really matters.
Why Stay Vigilant?
Precision science doesn’t leave much room for error. If a compound like N(Epsilon)-Trifluoroacetyl-L-Lysine degrades, whole protein modification experiments or drug prototype batches may end up unusable, wasting days of work. Labs striving for reproducibility can’t compromise on storage. Throw in the cost of custom-synthesized chemicals and these losses sting even more.
Clear protocols, practical labeling, and good training turn routine storage into reliability. I always made new researchers walk through storage routines, and surprise audits paid off in finding forgotten vials or improper setups. Standardizing temperatures, logging open dates, stacking with labeled orientation, and auditing stock regularly make sure you’re using the freshest materials. Real progress in chemistry builds on these habits.
What Can Improve the Status Quo?
More automation in lab inventory would help rattle the memory when supplies move toward expiration. Digital sensors tracking temperature or humidity—cheap and available now—tax less on the memory and build stronger habits. Discussions on lab safety sometimes miss these day-to-day choices, but storing specialty compounds safely ought to get as much airtime as fancy new instruments.
In the end, no safety data sheet beats the experienced eye and mindful care of the people working at the bench. Diligent habits not only save money, they build a culture of respect for materials and the discoveries they enable.
Why Purity Holds the Spotlight in Amino Acid Research
Researchers working with N(Epsilon)-Trifluoroacetyl-L-Lysine often start by asking about purity. In the world of amino acid derivatives, this question never stops being important. The detail isn’t just for the sake of formality—when someone steps into the lab and spends the day pipetting and watching reaction curves, the last thing they want is to chase ghosts caused by impurities. Each impurity can mean background noise in an assay or skewed data and confusion that nobody needs.
What the Market Offers for Purity
Most reputable chemical suppliers stock N(Epsilon)-Trifluoroacetyl-L-Lysine at a purity level above 95%. A select few offer grades boasting purities greater than 98%. Some researchers dig into certificate of analysis documents and look for HPLC or NMR readouts showing a single clean peak, evidence that the product lives up to expectations.
Genuine high-purity amino acid derivatives don’t come handed over for cheap. Purity drives up the price, not just by a little. There’s labor behind purification, and companies tap into chromatography and crystallization—methods that can stretch out timelines and increase cost but yield fewer contaminants in the end product.
Impurities, even in trace amounts, can make their presence felt where reproducibility matters. Peptide synthesis and enzyme modification are two places where leftover reactants or side-products can unravel hard work. I remember wrestling with poorly purified amino acids back in grad school, where a small contaminant would turn up during mass spec as an unexpected peak, wasting days in troubleshooting.
How Researchers Verify Purity
Placing blind trust in a label reads risky. People in the lab community swap tips about companies with a record for reliable analysis. High-purity grades should come with full documentation. HPLC traces telling a clear story, or NMR spectra with simple, well-resolved signals, provide reassurance.
Some labs check everything in-house before a big synthesis run. A colleague used to run a quick TLC or mass spec on every batch—a routine born from experience after a few mishaps. That habit saved him from more than one botched experiment.
Striving for Better and Safer Purity Standards
Industry guidelines keep evolving as demands for reproducibility grow. Vendors adapt by standardizing quality control, sharing analysis data, and moving towards cleaner production environments. Researchers talk about transparency and documentation as much as about the product itself. The need for safer, more reliable chemical reagents has pushed the community to demand tighter controls and better traceability.
Some scientists, especially those in pharma or advanced biotech, go further. They establish their own set of requirements and push suppliers for custom purifications or third-party analysis. The investment pays off, especially if a tiny impurity could lead to months of wasted work or impact on safety assessments.
Looking to the Future
The market for N(Epsilon)-Trifluoroacetyl-L-Lysine keeps growing with advances in protein engineering and bioconjugation. Manufacturers tune their protocols to fit ever-stricter research standards. This drive means the question of purity stays relevant—innovation raises the bar, but the search for cleaner, well-characterized chemicals never truly ends.
Grasping the Basics of a Tricky Amino Acid Derivative
Lab benches around the world hold all sorts of custom-made chemicals, but N(Epsilon)-Trifluoroacetyl-L-Lysine stands out for its unique twist: a trifluoroacetyl group stuck to the epsilon amino group of lysine. Chemists build on this molecule to protect the reactive side chain in synthetic experiments, especially while piecing together peptides. A simple question comes up all the time: Does this stuff dissolve better in water, or do you need an organic solvent?
What Makes This Lysine Special
To anyone who’s spent hours troubleshooting a stubborn reaction, solubility can make or break the day. With regular lysine, water works fine because the molecule mixes easily. Add a trifluoroacetyl group—suddenly, things get touchier. That group pulls the molecule toward the organic side. Trifluoroacetyl isn’t polar enough to bond with water like plain lysine does. Its effect takes a soluble-in-water amino acid and makes it much less friendly with water.
What the Science Says
Studies and lab experience both point the same way: N(Epsilon)-Trifluoroacetyl-L-Lysine doesn’t mix well with water. Tri-fluorinated molecules repel water molecules, and this protection group adds bulk to lysine. Researchers have reported much better results dissolving these derivatives in solvents such as methanol, ethanol, or dimethylformamide (DMF). These organic liquids match up with the partially hydrophobic structure, letting the compounds slip apart and dissolve. Acetonitrile and DMSO also show good compatibility.
That said, the story doesn’t end there. Temperature, concentration, and pH shift the picture. Warm up the solvent, or change the balance of acid and base, and you might coax some trifluoroacetyl-lysine into water. For most lab-scale setups, though, standard room temperature and neutral water leave a cloudy mess. Switching over to an organic solvent usually clears things up.
Why Solubility Challenges Matter Beyond the Bench
Anyone trying to scale up a synthesis runs into a different breed of trouble. Many green chemistry efforts aim to limit hazardous organic solvents, not keep adding them. The catch with N(Epsilon)-Trifluoroacetyl-L-Lysine: you still need DMF or similar to dissolve it. In my research days, swapping triphosgene for less harmful reagents or shaking up the solvent scheme felt like walking a tightrope. Less organic solvent means a gentler process, but poor solubility stalls everything.
Looking for Solutions: What the Field Is Trying
Some researchers play with solvent mixtures. Water-miscible organics like ethanol can give you the benefit of both worlds, if you keep careful track of the ratios. Tweaking the protecting group offers another route: some chemists use groups that break down or come off more easily in water, though this can complicate the chemistry. Encapsulation by cyclodextrins or adding solubilizing tags may also help nudge such amino acid derivatives into water—at least in small experiments. For now, the typical playbook sticks with organic solvents for dissolving N(Epsilon)-Trifluoroacetyl-L-Lysine, especially when quick, reliable dissolution matters.
Expert Opinion: Navigating This in Your Work
Anyone mapping out a synthesis or shipping custom peptides needs to weigh these solubility details. While regulations and safety guidelines keep tightening the screws on solvent use, real-world chemistry demands practical, effective solutions. Sticking to proven, compatible solvents ensures the reaction goes forward and the product shows up in good yield. As science pushes for cleaner, greener methods, deep knowledge of chemical structures—and their quirks—will only become more valuable.
Accepting Responsibility with Newer Chemicals
Whenever a synthetic amino acid like N(Epsilon)-Trifluoroacetyl-L-Lysine lands on a lab workbench, the burden of responsibility goes hand in hand with the search for new findings. Even if it’s a specialty compound showing up for peptide synthesis or as a chemical building block, nobody wants to see carelessness turn a scientific tool into an environmental or personal hazard.
Personal Experience and Lab Realities
Back in graduate school, I shared a cramped lab where unwatched chemical bottles sometimes ended up stashed behind the glassware. Every chemist I know has a mental list of “those” reagents—ones requiring gloves, goggles, and a healthy dose of respect. Trifluoroacetylated lysines fit this bill, not just for their potential irritancy, but also because trails of fluorinated organics too easily cross from bench to body or waste stream. Once, a careless spill in our group led to a sticky clean-up and a loud reminder about labeling and double-gloving.
What the Literature Teaches
Studies show that handling fluorinated compounds usually asks for more than lazy lab habits. Trifluoroacetyl groups tend to increase reactivity, and any exposure, inhalation, or skin contact with their dust or solutions can invite health problems. The key is isolation—physical separation from food, skin, and unintended chemical partners.
For N(Epsilon)-Trifluoroacetyl-L-Lysine, working inside a fume hood isn’t optional; vapors and fine particulates demand robust ventilation. Wearing nitrile gloves and a fitted lab coat provides a basic level of protection, but I always keep a fresh spill kit nearby for good measure. Storing the substance features on my checklist, too. I’ve learned not to trust old cardboard or makeshift shelving. The right secondary container, clearly labeled, keeps potential accidents contained if something leaks or shatters.
The Disposal Challenge: A Real-World Tangle
Disposal remains the trickiest part. Not long ago, our lab faced delays because the campus waste contractor flagged fluorinated waste for special paperwork. N(Epsilon)-Trifluoroacetyl-L-Lysine doesn’t just go down the drain or into regular trash. According to environmental agencies, trifluoromethyl groups resist breakdown and can add to persistent pollution, a growing concern in both water treatment plants and open landfills.
The right solution means following hazardous waste rules. This usually involves labeling the waste as “fluorinated organic,” sealing it in leak-proof containers, and logging it for institutional hazardous waste pickup. Some institutions insist on segregating it from other organic waste streams because combustion or improper disposal may create toxic byproducts. In all honesty, I sometimes grumble about the paperwork, but a quick look at contaminated waterways or headlines about PFAS, and it’s clear caution beats regret.
Room for Improvement
In the academic and small industrial world, education and institutional support make the biggest difference. Training new researchers to anticipate secondary contamination—from bench tops to pipette tips—could prevent many common mishaps. I’ve seen labs create clear, step-by-step charts showing the whole life cycle of a reagent, from delivery to disposal; these visual reminders set a standard that even frazzled postdocs can follow.
One might hope for greener alternatives to these chemicals, though as of now, their chemical properties often serve a unique role in peptide research and protein modification. So while tools like N(Epsilon)-Trifluoroacetyl-L-Lysine remain essential for certain applications, safe handling and thoughtful disposal can’t become afterthoughts. The science moves faster than the safety rules sometimes, so it takes everyone in the lab, not just the safety officer, to get it right.