Nonafluorobutane-1-Sulfonic Acid: Moving Past the Shadows of Persistent Chemicals
Tracing the Roots of a Fluorine Giant
Nonafluorobutane-1-sulfonic acid (abbreviated as NFBSA for those who work around heavy molecules all day) came about in chemistry circles after the world noticed the bone-deep problems with older PFAS compounds—think PFOS and PFOA. Teflon and Scotchgard once starred in ads and household hacks, then landed in lawsuits and water testing reports. After decades of ramping up perfluorinated chemistry, folks began seeing these "forever chemicals" in everything from polar bear livers to local tap water. That fear opened the door for alternatives with shorter carbon chains—NFBSA among them—hoping fewer links would mean less sticking around in bodies and ecosystems. I remember seasoned lab techs grumbling over new paperwork for every new PFAS but agreeing the old stuff had run its course for a reason.
The Makeup and Cut of This Compound
Lay out a bottle of NFBSA, and chemists see a clear, sometimes slightly yellow, liquid—distinctive with its long perfluorinated tail and a sulfonic acid head that packs a punch. It doesn’t catch fire easily, it shrugs off water and oil, and it brings both acid strength and resilience. Most PFAS keep their cool in nasty environments, and NFBSA is no exception. Boiling it takes real heat, its density shows up heavier than plain water, and it dissolves best in solvents that play nice with fluorine. Manufacturers stamp precise technical specifications on these drums—purity expectations push past 98 percent, tight controls on residual metals, and limits on water because moisture carries risks both for stability and downstream use.
How It Shows Up and Why It Is Labeled Like It Is
Product names for NFBSA pop up in scholarly papers, regulatory filings, and sometimes on auction listings for industrial chemicals. Synonyms track the IUPAC way—perfluorobutane sulfonic acid, or just nonafluorobutanesulfonic acid. Labels pull no punches about danger icons—corrosive marks for strong acids and often a warning about environmental fate. It is never a mystery why regulators and shippers standardize this language.
The Science of Synthesis and What Goes Into Making It
To get NFBSA, industry commonly fires up an electrochemical fluorination process, where organic precursors bathe in liquid hydrogen fluoride under some serious voltage. The original carbon backbone survives, but hydrogens bow out in favor of fluorine. Sometimes, direct sulfonation of previously fluorinated butanes comes into play. From my own forays into PFAS bench work, I remember the constant hum of fume hood fans, double-glove layers, and glassware that survived strong, nasty reagents. End products face rigorous purification, and by-products end up tracked or sometimes burned at deep-well incinerators.
Chemical Reactivity and Tweaks on the Basic Skeleton
Talking about sulfonic acid functionality means most reactions focus on turning that -SO3H group into something new—salts, esters, or activators for specialized catalysts. The perfluorinated carbon backbone rarely plays along with reaction schemes—it practically ignores most chemicals that would chew up everyday hydrocarbons. This tough, inert backbone delivers the property balance companies seek: chemical stability and surface activity. Most labs use NFBSA when they want the performance edge without reactivity surprises.
Daily Life in Handling: Keeping Operations Safe
Everyone who works with PFAS, including NFBSA, learns to respect their hazards early. Corrosive to skin, hazardous if inhaled or swallowed, and persistent in the environment—the acid asks for closed systems, acid-resistant containers, and strict waste collection. In offices and plants, checklists swarm every transfer or mixing job, and wastewater goes through treatment, never down drains. Even the air in the lab gets scrubbed. After seeing young researchers underestimate how quickly acid vapors can bite, I can't stress enough the routine importance of splash goggles, glove choice, and constant ventilation checks.
Where the Material Ends Up: Applications Amazon-Wide
Industry loves PFAS for their tailormade grip on dirt, water, and oil, so NFBSA finds its way into products like stain-resistant coatings, waterproof textiles, and specialized surfactants. It's played a part in fire-fighting foams and even semiconductor manufacturing where only the toughest, purest acids survive. Environmentalists watch the runoff closely, but customers keep asking for long-lasting, easy-clean performance. In nearly every application, the features are hard to replicate with other chemicals—stain repellency lasts through washes, and in electronics, tiny defects drop when cleaners work at nanoscale.
What Scientists Push Toward: New Methods and Hopeful Solutions
Research teams now target every angle: alternative synthesis to drop hazardous byproducts, excellent separation methods for cleanup, and even molecular tweaks that can speed up natural breakdown. Major journals brim with papers chasing “shorter chain” PFAS designs, but breaking the carbon-fluorine bond efficiently outside a multi-million-dollar incinerator still puzzles many. Companies in the EU and US look for greener fluorination technologies, but for every success, a fresh layer of environmental risk assessment appears. Conversations about NFBSA rarely leave out this push-and-pull—drive for performance, concern for future generations, and an industry learning to reinvent itself under scrutiny.
Toxicity and Health: What’s at Stake and What the Evidence Shows
Early years saw PFAS research cruise under the radar. As water tests improved, researchers started seeing widespread low-level exposure. NFBSA, as a shorter-chain alternative, has drawn attention; some toxicology reports point to reduced bioaccumulation compared to older siblings, but “less bad” doesn’t mean harmless. Animal studies outline risks to liver health, and environmental persistence still makes clean-up a headache. Once these chemicals enter a watershed, pulling them out of soils and surface water marries science and headaches. I’ve seen emerging research tying certain PFAS to cholesterol effects and immune shifts, so conservative public health voices want to crimp emissions at the source rather than wait for long-term population studies. No silver bullet in sight; what matters most lands on minimizing exposure and listening when scientists voice early warnings.
Scanning the Horizon: Life Beyond “Forever”
Talking about the future of NFBSA and its class, the line stays blurred between innovation and caution. Efforts aim for molecular designs that break down in nature—not hang around for generations—while tightening rules around what can go down the drain or open incinerators. Legal and regulatory struggles now force producers to weigh liability and sustainability alongside profit and performance. Big brands face consumer demand for safer products, so new surface-active agents must prove themselves under tougher testing. That means real sustainability strategies, investment in water purification research, and hard looks at the true cost of high-tech convenience. Next chapters in PFAS chemistry won’t repeat the naive optimism of past decades; instead, they’ll need joint effort—industry labs, regulatory watchdogs, and researchers all pulling against the long shadows cast by these persistent molecules.
Inside the Chemistry That Shapes Our World
Step into any lab handling modern industrial processes, and you might run into chemicals like Nonafluorobutane-1-sulfonic acid. It’s a mouthful, but this compound turns up in surprising places. You probably haven’t seen it on store shelves, yet its effects ripple through electronics production, specialty coatings, and even firefighting foam. Most folks never think twice about how semiconductors and smart devices keep working. Without this sulfonic acid, manufacturing precision shrinks, and advanced tech just doesn’t stay on track.
The Hidden Engine Behind High-Performance Products
Back in my early career days as a research assistant, I watched circuit board prototypes sputter out—leaving engineers frustrated. Later, I learned that engineers choose substances like nonafluorobutane-1-sulfonic acid because of their stability and reliable surfactant power. In layman’s terms, it helps certain materials spread smoothly or allows etching and cleaning on a microscopic scale. Think fingerprint-sized chips that hold gigabytes of your photos or control electric cars. These acids help make those devices robust, durable, and free of microscopic grime.
But it’s not only the tech sector. In specialty coatings, this sulfonic acid helps water-resistant sprays cling to surfaces. You can picture it working in everything from stain-proof upholstery to oil-repellent clothing. If you’ve ever spilled a drink on your sofa and watched it bead up instead of soaking in, you owe a nod to fluorinated chemicals.
The Cloud Over “Forever Chemicals”
There’s no sugarcoating it—fluorinated compounds build up in nature and in our bodies. Researchers and regulators call these compounds “forever chemicals” because they don’t break down easily. Studies show links between some per- and polyfluoroalkyl substances (PFAS) and health risks, including cancer and hormone disruption. Nonafluorobutane-1-sulfonic acid belongs to this broader family.
Watching neighbors rally against water contamination shaped my thinking about chemical safety. It’s easy to get swept up in the excitement of new tech, but cleaning up polluted groundwater is slow and expensive. My community faced a real scare a few years ago, and folks still talk about what leaked into local wells decades ago. The push for alternatives grows because no one wants that risk hanging over their heads.
Balancing Needs and Navigating the Future
There’s value in recognizing achievements—modern medicine, clean energy, and advanced electronics all rely on these chemicals. Still, positive change happens through innovation. Some companies already test greener surfactants or improve recycling after industrial use. Others tighten emission standards or upgrade containment measures on factory floors. Regulatory agencies worldwide dig into research, setting new limits for PFAS in drinking water and soil.
Tough conversations lie ahead. The answer won’t come from banning every compound outright or ignoring risks for the sake of convenience. Transparent reporting, strict safety measures, and support for “cleaner chemistry” research give families and workers a shot at both safety and progress. Every time we choose how much risk to accept in pursuit of modern living, we shape the world our kids inherit.
Why Everyday Work Habits Matter More Than Labels
Most people outside a chemistry lab have probably never heard of Nonafluorobutane-1-sulfonic acid. Those who have, know it’s a real player among strong acids in industry and research. There are safer substances out there, but when work calls for this one—often for its stability, resistance to breakdown, or unique chemical behavior—the usual gloved-handwave doesn’t cut it. Nobody can skate by on vague warnings or “handle with care” written on a bottle. This is one of those compounds that asks for real-world respect.
Gloves, Goggles, Ventilation—Not Optional
I don’t just toss these three words around lightly. My own years around strong acids have shown me the value of proper gear. Gloves that stand up to acids—usually nitrile or butyl rubber—go on first. Lab coats keep splashes off clothes, but goggles or a proper face shield guard against that one-in-a-thousand splash or mist in the eyes, which is all it takes to end a career, or worse. You can’t fake this step by pulling your shirt over your mouth. The acid’s mists and vapors drift invisibly. A fume hood isn’t some luxury; it removes chances of breathing in nastiness you might not even notice. If you wrinkle your nose, you’re already late.
Never Work Alone, Never Guess
Comfort in the lab adds up to sloppiness fast. I spent one summer at a facility that hammered home the “buddy rule”—nobody poured, weighed, or transferred risky chemicals alone. Mistakes, spills, and sudden exposures happen even to pros. Having someone there means a quick rinse or call for help, rather than a bad statistic. Guesswork also slips into routine: “Maybe this container is clean enough.” Or, “That looks diluted already.” Those shortcuts have real consequences. Knowledge and double-checking pay off more than any trophy.
Accidents Mean Quick, Not Quiet
Nobody wants to be the person who trips an eye wash or emergency shower. Get exposed, and your only job is flushing and telling someone. I’ve seen embarrassment stall people just long enough for injuries to get worse. Acid burns don’t negotiate. The stories that stick in my mind all involve people who moved fast, stayed loud, and got help before a minor tingle turned into hospital time.
Clean Workspaces, Smart Storage
Organized labs don’t just look nice—they limit accidents. Spills on a cluttered bench, or open containers next to unrelated chemicals, open up doors to reactions you may not expect. Proper containers beat reusing an old bottle. Tightly screwed caps avoid the morning surprise of a pungent cloud when you walk in early. I once found out, painfully, that storing even small acid samples outside of proper secondary containment can turn a minor drip into a much bigger mess. That’s a cheap lesson compared to what could have gone wrong.
Respect, Not Fear
No chemical deserves panic, but every strong acid—including Nonafluorobutane-1-sulfonic acid—earns basic respect. That means reviewing safety data before starting, using gear that really blocks splashes or vapors, and keeping distractions out of the workspace. Training hands-on, not just signing a checklist, sets apart teams that go years accident-free. Labs that rarely practice spills or review gear wind up learning those lessons the hard way.
The Bottom Line: Safety Is a Habit
Nobody working with this acid should treat safety steps as extra chores. They’re the difference between a productive afternoon and a life-changing mistake. Simple, clear habits—right gloves, goggles, teamwork, organization, and fast response—take most of the surprise out of tough chemicals. No chemical, no matter the label or pedigree, becomes less dangerous with shortcuts.
The Chemistry Behind Nonafluorobutane-1-Sulfonic Acid
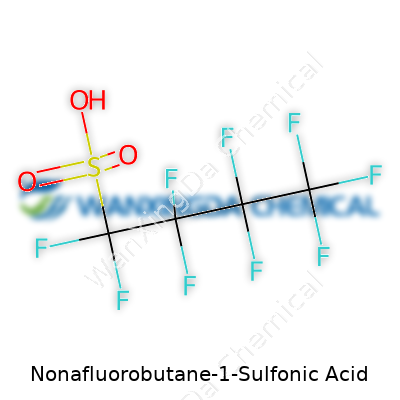
Nonafluorobutane-1-sulfonic acid has always captured the attention of scientists and regulators thanks to its unique structure and persistence in the environment. Its chemical formula—C4F9SO3H—tells a story. Breaking this down, the ‘nonafluoro’ prefix means nine of the hydrogens, normally on a butane backbone, get swapped out for fluorine atoms. This makes the molecule stubbornly resistant to getting broken down, whether by light, heat, or living things. Add a sulfonic acid group at one end, and you get a chemical that loves both water and oil. The molecular weight sits at 300.13 g/mol, and this number becomes a reference point for everything from tracking contamination to planning rare cleanup projects.
Environmental and Human Consequences
A lot of folks talk about chemicals like nonafluorobutane-1-sulfonic acid as if they’re just clouds in a lab, but the truth settles into soils and lakes. The same ruggedness that makes these compounds useful in manufacturing—resisting breakdown, sliding off surfaces—increases their risks when they get loose in nature. Reports from countries like the US and Germany keep confirming detections in water supplies, far from their places of manufacture. People living near industrial sites sometimes wonder if their well water carries these ‘forever chemicals,’ and not without reason. The resilience of these molecules means they can build up, not only in water but in fish and other wildlife.
Facing the Challenges
I remember hearing from local farmers who noticed changes in the nearby river. Fish populations were shrinking, and the frogs—once everywhere—seemed rare. While many factors can drive those trends, rising perfluorinated compound levels draw the public’s eye for a simple reason: solutions feel hard to come by. Water treatment plants weren’t designed for molecules as stubborn as C4F9SO3H. Activated carbon and advanced filtration work for some compounds, but long-chain variants like this one often slip through.
Accountability and Solutions
Tackling this issue isn’t about panic. It comes down to honest conversations between manufacturers, communities, and policymakers. Industry groups argue that these chemicals fill important gaps in technology—like helping semiconductors stay clean or firefighting foams snuff out stubborn blazes. People in affected regions, though, push for stronger oversight and clear disclosure of contamination. The call for alternatives rings out, especially as more toxicity studies point to health effects from chronic exposures.
Some researchers are experimenting with new breakdown techniques. Ultra-short wavelength UV, greener chemical degradation, and even bacterial digestion are all on the table. The solutions may not appear overnight, but progress depends on a clear-eyed approach to what these chemicals are doing to our food, water, and health.
Lessons Learned from an Intractable Molecule
Chemicals like nonafluorobutane-1-sulfonic acid offer a powerful reminder: just because we can build something tough, doesn’t mean we can always put that genie back in the bottle. Striking a balance between technical needs and environmental responsibility no longer seems optional; it’s fast becoming a central part of public health. Staying informed and demanding visibility into both the use and release of these compounds shapes the path forward. Communities and scientists both look for ways to cut exposure and hold each other accountable, knowing full well that some molecules don’t just ‘go away’ in a season or a decade.
A Chemical Worth Respecting
Anyone who’s spent long hours in a chemical lab will tell you there’s no cutting corners with substances like Nonafluorobutane-1-sulfonic acid. This compound offers plenty in industrial chemistry, especially for high-demand processes. Its stability and performance also make it hard to ignore in modern manufacturing. Still, what makes it valuable also makes it risky without smart storage.
Hands-On Reality: It's Not About Fear, It's About Control
I’ve had to clear up spills of strong acids on more than one occasion. One lesson always sticks—the best place for corrosive, fluorinated acids is behind closed, labeled doors. This acid expects respect, not because it’s exotic, but since it eats through surfaces and bites skin fast. Flesh, plastics, or even costly lab counters end up damaged before you realize a mistake has happened.
Locked cabinets built from corrosion-resistant materials set the baseline. Forget cheap shelving. Metal parts rust, plastics can crack. Go for polypropylene, or similar resistant options. Every decent chemical supplier invests in cabinets with chemical seals and venting. This doesn’t just keep fumes contained, it reduces the risk of aggressive vapor damaging surrounding materials. In one of my old labs, careless storage cost us thousands in equipment replacements after vapors leaked into other storage.
No Shortcuts With Labeling or Organization
A system works best when everyone can see what sits inside a bottle or drum from a distance. Running markers on generic containers has no place near Nonafluorobutane-1-sulfonic acid. Keep it in original packaging or use containers approved for strong acids. Slap a big, clear label on it, naming both the substance and hazard class. In my experience, the fastest way to a preventable accident comes from a missing or smudged label.
Humidity and Temperature: Consistency Pays Off
Any chemist working with acids soon learns to keep an eye on heat spikes and moisture. High humidity encourages unwanted reactions or slow degradation of the container. I once watched a solid acid cake and clog a bottle for months. Choose a cool, dry room, with a reliable thermometer. Avoid placing the acid anywhere that sees temperature swings, like warehouse windows or near radiators. I always think—if I wouldn’t trust milk in that corner, I won’t trust a sensitive acid there either.
Separation Saves More Than Time
Crowded shelves with different chemicals look impressive until something leaks. Pairing acids and bases is begging for disaster. Segregate Nonafluorobutane-1-sulfonic acid from incompatible chemicals, especially strong bases, organic material, or anything reactive. One honest mistake at a shared storage site stained an entire wall and caused a scary rush for the eyewash station. It never hurts to keep an updated, easy-to-read map of chemical storage zones.
Rely on Training, Not Luck
No matter how many safety posters line the walls, they can’t substitute for experience or regular safety drills. I’ve walked new trainees through real-life spill drills, and those moments stick much longer than PowerPoint slides. Everyone on site should know where the acid sits, what to do in case of a spill, and how to use emergency supplies nearby. The difference shows when someone’s calm under pressure instead of wide-eyed.
Moving Forward With Practical Respect
Some chemicals demand airtight discipline. Nonafluorobutane-1-sulfonic acid rewards anyone who gives it the structure it deserves: solid cabinets, clear labels, the right climate, and zero tolerance for clutter. Rather than treating it like a hazard to fear, think of safe storage as building a reputation for working well, working safely, and never getting caught off-guard by a predictable mistake.
One of the “Forever Chemicals” in the Spotlight
Nonafluorobutane-1-sulfonic acid belongs to the PFAS family, a group of compounds earning the “forever chemical” label for their stubbornness in the environment. If you’ve read about water contamination controversies lately, PFAS probably rings a bell. While nonafluorobutane-1-sulfonic acid doesn’t get as much attention as its cousin PFOA, it deserves a closer look. These molecules resist breaking down, either in nature or inside our bodies. That’s not just a quirk — it signals a promise of lasting environmental trouble wherever this compound ends up.
Environmental Impact Goes Beyond the Obvious
It sticks around in water, soil, and air much longer than most industrial chemicals. After release, it moves easily through groundwater and surface water — an ability that raises the risk of drinking water contamination. Fish, wildlife, and humans all come into the picture next, since these chemicals build up in living tissue over time. In me, any new chemical that lingers and spreads this way sounds like a risk, as past experience with PCBs, DDT, and earlier PFAS lessons shows. The science says that even low-level, long-term exposure adds up, possibly causing health problems. The bottom line: a chemical that keeps moving without changing brings worry for any ecosystem it touches.
What the Research Says: Real Concerns
Lab tests and field studies have started linking PFAS in water and food to health issues like immune suppression, reproductive trouble, and possibly some cancers. The evidence is strongest for older PFAS types, but regulators and researchers warn newer ones — including nonafluorobutane-1-sulfonic acid — might pose similar risks due to their structure. Some research suggests this compound doesn’t break down much differently. EPA and European regulators both list it among substances needing monitoring or phase-out, which says a lot about the direction science is pointing.
Learning From Past Mistakes
We’ve seen the pattern before: a new, industrially useful molecule rolls out, only for society to discover years later that the cost in public health and environmental stability piles up. PFAS regulation lags behind the science, but communities and companies are starting to wake up. Some water systems have already found PFAS, including nonafluorobutane-1-sulfonic acid, in treated drinking water. Cleanup technology, such as activated carbon or reverse osmosis, helps reduce levels in water, yet these fixes rarely reach communities early enough. Many existing laws struggle to keep up with how widely PFAS travel or how easy it is for industries to switch to a new, barely-studied alternative.
Looking Toward Solutions
Transparency from manufacturers could help, since knowing where and how this chemical gets used allows communities and regulators to get ahead of contamination. Stricter discharge limits might keep more PFAS out of water in the first place. Some experts call for requiring environmental risk data before approving any new chemical of this class, not after. New technology for destroying PFAS, not just filtering or moving it, could offer hope, though practical solutions for large areas remain out of reach.
The promise of nonafluorobutane-1-sulfonic acid — stain-proof, fire-resistant, and nearly indestructible — turns into its greatest environmental danger. It enters the world easily, escapes even faster, and stays nearly forever. That’s a lesson the world is still learning, one urgent study and water test at a time.