Perfluorobenzene: Turning Chemistry’s Corners Into New Possibilities
History in the Making
Long before clean rooms and precision lasers, chemists started tinkering with benzene rings in search of materials with less reactivity and more staying power. That curiosity brought perfluorobenzene to the lab bench, more than a hundred years ago. Early reports showed a shift in scientists’ mindset: exchanging familiar hydrogen atoms for fluorine on benzene’s hexagon opened new chapters in organic chemistry. Up until then, nobody could quite predict how profoundly that simple switch would break conventions. Shoring up the ring with hard-to-budge fluorines meant the resulting molecule shrugged off acids and bases, heat, and even ozone. Labs raced ahead to further unravel this resilient molecule, and new uses kept popping up, especially when modern analytical instruments like NMR and mass spectrometry needed chemical benchmarks that wouldn’t be chewed up by ordinary solvents.
The Skinny on Structure
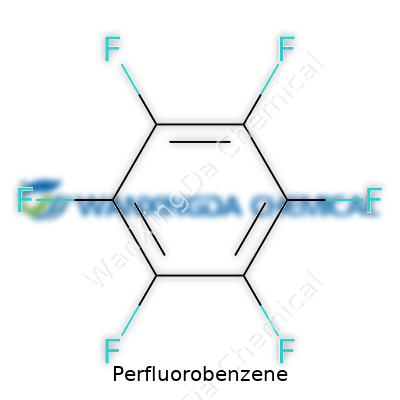
If you want to picture perfluorobenzene in your head, just imagine a benzene ring decked out with six fluorine atoms. This small tweak brings a sea change in its chemistry compared to plain benzene. It pours out as a colorless, clear liquid—a bit heavier than water and hardly interested in mixing with it. The sharp aroma chimes in as a warning: this isn’t something you want to spill. Unlike its hydrocarbon cousin, perfluorobenzene doesn’t catch fire easily and barely reacts under the sun or with most chemicals tossed its way. It boils at a modest 80 degrees Celsius, which means it can hang around as a liquid under typical lab conditions, but it evaporates readily with some warmth.
Crunching Numbers: Technical and Label Information
On paper, perfluorobenzene comes stamped with the name hexafluorobenzene and wears molecular formula C6F6 like a badge. The structure is so rigid that most chemical exchanges bounce off of it. Density hovers around 1.6 g/cm³, which means you’ll notice its heft if you hold a vial. For shipping and labeling, it goes by several monikers across the world. Containers must warn of inhalation dangers and skin contact risks, often showing hazard symbols that prompt careful handling. The CAS number—392-56-3—helps distinguish it from its less-fluorinated cousins, which comes in handy since many fluorinated aromatics fill chemical catalogues.
Making Perfluorobenzene
People sometimes think complex molecules only arise from obscure, expensive processes. Perfluorobenzene’s story shows how industry and academia team up to tackle this hurdle. Lab pioneers first tried direct fluorination of benzene, only to discover that was a recipe for runaway reactions and sticky tars. Safer and more reliable techniques emerged from the patient work of organic chemists. One common route now involves the Fowler process, where metal fluorides slowly transfer fluorine atoms under controlled conditions, letting technicians rein in the reaction and keep unwanted side products low. No giant reactors or space-age equipment necessary; just a mix of discipline, thick-walled vessels, and a keen eye for containment, since fluorine gas likes to play rough if left unsupervised.
Chemical Adventures: Reactions and Modifications
Those six fluorine atoms make a circle that throws up chemical barricades almost everywhere you look, and that stubbornness means perfluorobenzene won’t mingle with acids, bases, or oxidizers. Standard attempts to tweak the molecule—swapping out a fluorine for something else—require high pressure, high temperatures, and custom-built catalysts. The reactivity of the ring edges falls far below most aromatics, which reduces risks in some applications but hampers efforts to graft on functional groups for more exotic uses. Some specialized reactions, especially involving nucleophiles, have made headway by playing with harsh conditions to pry open the ring’s defences, but these methods remain a tough sell at large scale due to complexity and cost.
A Menu of Names
Chemists and manufacturers alike know perfluorobenzene under a handful of aliases: hexafluorobenzene, benzenehexafluoride, and sometimes the short form HFB. No matter the title, it circles back to the same structure, though keeping tabs on synonyms is more than trivia—it avoids costly mix-ups in supply chains and lab inventories. In journals and patent filings, a slip in name-checking wastes time and resources, so researchers and safety officers parse every label and shipment for accuracy.
Staying Safe in the Lab and Factory
Handling perfluorobenzene challenges newcomers and veterans alike. The lure of a stable molecule can lull operators into skipping protection, but the vapors sneak past standard gloves and fatigue the respiratory system if given half a chance. Regulatory bodies flag it for its environmental stability; it sticks around in soil and water, stacking up over time, raising concerns about persistence and possible long-term effects. Strict protocols call for chemical fume hoods, nitrile gloves, tight-sealing goggles, and strict tracking of every drop. Spills are hard to mop up and rarely degrade, so training drills focus on containment and scrap disposal rather than old-school detox. In high-throughput labs, risk assessments don’t just gather dust—they evolve with fresh data and changing workplace habits.
Where Work Gets Done: Uses and Industries
In my own experience, few chemicals move as quietly between fields as perfluorobenzene. Analytical chemistry gives it pride of place as a reference standard in NMR and mass spectrometry labs, since its signals ring out clearly without overlap from hydrogen. Materials scientists prize its resilience as a building block or test medium where solvents break down or cause trouble. It opens up non-stick coatings and high-performance insulating fluids that rival Teflon, and electronics makers toy with it as a solvent for specialized applications. Even as synthetic intermediates, perfluorobenzene helps shape next-generation polymers, bringing durability to fiber optics, membranes, and specialty plastics that stand up to harsh environments. Research circles buzz about fine-tuning its use as a contrast agent in advanced medical imaging or building fluorinated frameworks for capturing greenhouse gases.
Chasing New Questions: Research and Development
As the market for high-tech materials grows, chemists keep hunting for tweaks to the original perfluorobenzene recipe. R&D groups focus on leveraging its stability to solve practical problems—making batteries that last longer, or prepping polymers that hold shape and structure under punishing heat and chemical attack. Publications over the last decade show a spike in exploration of functionalizing the fluorinated ring, seeking that sweet spot where performance and processability line up. Nanotechnology labs eye the molecule’s planar structure for assembling new thin films, where every atom counts, and green chemistry teams eye the stubborn ring for inspiration but worry about end-of-life disposal. Trade-offs between innovation and environmental cost spark debates at every conference, nudging the field in directions nobody predicted twenty years back.
The Hard Truth: Toxicity and Environment
Toxicologists have not found clear links between perfluorobenzene and acute health risks for humans at low exposures, but the devil remains in the details and the data gaps. Tests in laboratory animals point to low acute toxicity by mouth or skin contact, but the vapor phase can irritate the lungs and eyes. Chronic exposure studies take years to yield answers, so policy makers err on the side of caution, restricting volumes especially near water sources. The molecule’s reputation for refusing to break down draws attention; slow degradation in the environment means every gram produced keeps tallying up. Some regulators group it with so-called “forever chemicals,” turning the spotlight to both safe use and end-of-life strategies.
Looking Ahead: Potential and Responsibility
New markets demand perfluorobenzene in tighter purities and cleverer blends, especially as batteries, electronics, and medical devices keep ratcheting up their standards. There’s no easy fix for balancing performance with environmental stewardship, but a few promising trends stand out from the noise. Some startups develop recycling techniques that reclaim the molecule from used electronics, treating waste not as a headache but as a resource. Synthesizing perfluorobenzene from renewable starting materials sits just over the horizon, promising to cut the carbon footprint of production if the process can scale. In classrooms, teachers use its resistance to most reactions as a foil to inspire debate about chemical design and sustainability. Without open discussion between lab, factory, and community, mismatches in use and disposal threaten to overshadow the technical achievements. As we push materials to last longer and work harder, chemists have to own the conversation about what comes next—on the workbench, in the field, and in the places we all call home.
Where Perfluorobenzene Shows Up in the Real World
Perfluorobenzene hides behind a name that looks like a headache from high school chemistry, but it shows up in surprising ways. This compound shows up most often in labs and in certain industries where precision trumps ordinary choices. In my years covering science news, I’ve seen perfluorobenzene become a workhorse whenever folks need a liquid that won’t budge in the face of harsh chemicals or high heat. Its powers stem from the way its carbon and fluorine atoms link up, making the whole thing stubbornly non-reactive. That stubbornness makes it valuable in places where even sturdy solvents cave under pressure.
Why Scientists Reach for Perfluorobenzene
Solvents set the stage for many breakthroughs, and in recent research, perfluorobenzene steps in when traditional options fall short. For NMR spectroscopy, a method that relies on pure, reliable solvents to analyze the structure of molecules, this one comes up often. It stays put, not interfering with sensitive measurements. I spoke with a chemist at a university who described it as a “background player” — you barely notice it because it causes so few problems. Yet, its lack of interference lets scientists push ahead with complex studies.
Industrial Uses and What That Means for Everyday Life
Beyond the lab bench, perfluorobenzene also finds a home in electronics. Think of the delicate wiring and circuit boards inside your phone or laptop. Cleaning solutions can corrode sensitive materials, so manufacturers lean on chemicals like perfluorobenzene to do the job without sparking new problems. Its resistance to heat makes it useful in the manufacture of heat-transfer fluids. Some companies even use it in processes that demand chemicals that won’t react with almost anything else around. That versatility explains why, even with a high price tag, demand keeps steady in niche but vital sectors.
The Environmental Catch
People who read about fluorinated chemicals often wonder about environmental risk — and they’re right to raise the issue. Perfluorobenzene belongs to a wider family called PFAS, which have sparked plenty of concern. These chemicals don’t break down quickly in the environment, earning them the ‘forever chemicals’ nickname. Reports have shown traces in drinking water, wildlife, and soil, setting off alarms about long-term health risks. So while perfluorobenzene may be invaluable for some uses, its resilience also means it lingers unwanted where it escapes containment.
Moving Toward Safer Solutions
For people working with this chemical, understanding its behavior and taking care to contain spills or leaks stays critical. Some labs have adopted closed-system approaches so that perfluorobenzene never leaves the equipment. Others invest in responsible disposal, sending waste for high-temperature incineration rather than letting it slip into waterways. There’s also a growing push in research circles to find safer alternatives — solvents that don’t hang around so long after their job is done. Technology usually moves on when enough smart minds focus on a problem, but until then, anyone using perfluorobenzene faces a responsibility not to pass today’s convenience on as tomorrow’s trouble.
What Is Perfluorobenzene?
Walk past a chemistry lab and chances are you won't see a bottle labeled “Perfluorobenzene” on every shelf. This clear, colorless liquid belongs to a class of chemicals known as perfluorocarbons, which are known for their stability and resistance to breaking down in nature. Chemists value these materials for projects requiring extremes. From specialty solvents in electronics to exotic uses in the lab, perfluorobenzene marks its territory out of reach from the average home or workplace. But the subject keeps coming up, usually in the shadow of health worries tied to chemicals that hang around too long.
Is It Dangerous to People?
Look at research on perfluorobenzene and the health story gets muddy. No mountain of public health data describes what happens if people breathe it or get it on their skin regularly. That doesn’t mean we should wave away any risk. In my own years around labs, the less you know about a chemical, the more careful people get. It’s hard to ignore how other perfluorinated compounds—like PFOA or PFOS—have built ugly reputations for environmental stubbornness and concerning health effects. Tests show those substances can end up in blood, water, and wildlife, and some connect their presence to hormone issues, cancer, and immune trouble.
Perfluorobenzene breaks down slowly—if at all—outdoors. What we know about these strong carbon-fluorine bonds tells a plain truth: what doesn’t fade away tends to pile up, first in soil and water, then in the bodies of animals and people. It’s the chemical’s invisibility that makes me wary. You won’t know you’re exposed until years have passed, but by then, consequences may linger. Regulators haven’t come out with any strict rules for this substance yet. The Environmental Protection Agency has focused most of its efforts on more notorious “forever chemicals.”
Why Care About It?
Many brush off niche lab chemicals as irrelevant outside research circles. I once believed that, too—until legacy toxins like PCBs and DDT surfaced in food, rivers, and breast milk decades after first use. By that point, damage cost billions and cleanup jobs seemed endless. Ignoring a possible health risk delays the hard work of understanding it. If perfluorobenzene gets out of labs and into the larger world, whether from spills or industrial releases, tracking and controlling it will become a bigger deal and far more expensive to handle after the fact.
Solutions Start with Caution and Knowledge
There’s room to get things right before problems begin. Researchers and those using perfluorobenzene need clear training. Simple steps matter: use closed systems, work in ventilated hoods, and keep waste out of public drains. Regular checks on air and water around production sites detect changes early and stop small spills from turning into community headaches. Mandatory disclosure of lab chemical inventories could help people spot risks and move toward safer alternatives.
Governments should invest in studies filling the gaps about how perfluorobenzene behaves in people and the environment. That means funding fresh toxicology work—not waiting for a crisis to spur action. Schools and training centers must prepare scientists to recognize the power and danger inside their storage cabinets. People trust researchers to handle things with care; getting ahead of the evidence on unknowns like perfluorobenzene honors that responsibility.
Digging Into the Formula: C6F6
Perfluorobenzene has a mouthful of a name, but its formula, C6F6, keeps things simple. You start with benzene, which chemists everywhere know as C6H6, then swap every hydrogen atom for a fluorine atom. That swap changes more than just the symbol on the page. This single tweak creates a compound that acts miles apart from the original. Those fluorine atoms pack real punch: they're big, they're stubborn, and they cling tightly to the carbon ring, making the molecule as stable as a fortress.
Why C6F6 Keeps Scientists Talking
I still remember the first time I learned how swapping out all those hydrogens for fluorines transforms a familiar chemical into something rare and durable. Fluorine doesn't like to let go. That's part of why perfluorobenzene resists breakdown, even under tough conditions where most organic molecules would crumble. This strength comes at a cost; it takes plenty of effort and specialized techniques to manufacture. Yet, because it doesn’t break down easily, it opens doors in chemical research, electronics, and even as a solvent for delicate reactions that can’t tolerate the usual suspects like water or alcohol.
These qualities highlight how the tiniest change at the molecular level can set off an entirely new range of possibilities. It always leaves me thinking about the impact precise chemistry can have beyond the lab. Consider that C6F6 doesn’t just serve as an inert bystander; researchers use it to explore nonstick coatings, new drug delivery routes, and materials that could change aerospace or clean technology.
Perfluorinated Compounds: Power and Responsibility
We can’t ignore the elephant in the room. Perfluorinated compounds like perfluorobenzene have a reputation, and not always a clean one. Their persistence, which chemists love, also means they don’t readily leave the environment. While perfluorobenzene itself isn’t as widely used as some others, the larger family—PFAS—has turned into a touchpoint in environmental and public health circles. These compounds have shown up everywhere from industrial wastewater to the bloodstreams of people who live nowhere near a chemical plant.
It’s sobering to realize that as much as these molecules help us, there’s always a question of what happens next. I once spoke with a chemist who called it the “forever molecule problem.” Perfluorobenzene stays put and, at large scale, could enter water or soil if waste isn’t managed. Communities have started demanding more accountability and better solutions. Some labs are now trying to cook up ways to break down perfluorinated compounds safely, something that chemists once thought impossible. These new ideas look at everything from ultra-strong ultraviolet light to biochemical catalysts borrowed from nature.
Balancing Innovation with Caution
There’s no escaping the fact that C6F6 and its siblings force us to balance innovation with responsibility. Their usefulness in the lab and industry doesn’t excuse careless disposal or ignoring long-term consequences. Until we figure out safer ways to manage them, the best move is to limit misuse, track where these compounds turn up, and invest in real solutions for cleanup.
Perfluorobenzene Storage Isn’t Just About the Science
Anyone who’s spent real time around chemistry labs knows that certain chemicals don’t just “sit” on shelves. Perfluorobenzene falls into this category. It’s a fluorinated aromatic compound, and the way people treat it sets the tone for overall lab safety and environmental responsibility. So, this isn’t just about ticking a box on a safety inspection form.
What Science Says—and What Experience Teaches
Perfluorobenzene doesn’t ignite easily compared to many other organic solvents, thanks to those rock-solid carbon-fluorine bonds. That gives a false sense of security to those unfamiliar with fluorochemicals. Just because something seems stable doesn’t mean you toss it in any old bottle and forget it in a dusty corner. From experience, cutting corners leads to leaks, equipment degradation, and miserable cleanups down the road.
Why a Tight Seal Matters
Over time, even tiny leaks build up in a storeroom. Perfluorobenzene evaporates slower than lighter solvents, but vapor exposure still accumulates in a poorly ventilated area. A proper glass container with a Teflon-lined cap keeps everything in check. Metal containers often corrode over time, even though the chemical isn’t aggressively reactive toward most substances. Moisture in the air can make things complicated as well, because contamination means ruined experiments and wasted money.
Temperature and Light—Not Just About Comfort
Fluctuating temperatures take a toll on chemical stability. In hot environments, pressure builds up in containers, making even solid caps fail. Over the years, I’ve seen storage rooms with wild temperature swings lead to cracked bottles and foul smells. So, stacking perfluorobenzene in a cool, shaded space isn’t an afterthought—it’s a lesson learned from chemical stains and wasted afternoons cleaning spills.
Labeling Saves Time—and Lives
A clear label keeps coworkers honest and safe. There’s always someone new in the lab, and assuming everyone knows which bottle holds what is wishful thinking. Accidents often start with unlabeled or poorly labeled containers. Including the chemical’s name, date opened, and hazard class heads off disaster. It also makes annual inventory checks less of a headache.
Solving the Bigger Problems
Lab managers and researchers can’t ignore regulations, either. In the US, the Occupational Safety and Health Administration (OSHA) expects safe chemical storage practices for good reason. Neglect invites fines, investigations, and damage to reputations, not to mention possible harm to those nearby. Beyond personal experience, public records show that cutting corners with chemical storage leads to fires, contamination, and chronic health risks. Following guidelines for ventilation, access control, and secondary containment shields not only staff but the whole building.
Plenty of good information comes from the American Chemical Society and environmental protection agencies. Tapping into these resources means investing in prevention, which ultimately costs less than cleaning up a disaster. The story is the same across labs, universities, and industrial settings: make chemical storage a priority, keep things simple, and act before things go sideways.
Understanding What Makes Perfluorobenzene Unique
Growing up around chemistry labs sparks curiosity in the odd little quirks each compound brings to the table. Perfluorobenzene, or C6F6, once caught my eye during a university research project. This isn’t your average organic liquid—swap out hydrogen atoms in benzene for fluorine, and you get a substance with some remarkable features.
Perfluorobenzene carries a striking combination of clarity and chemical toughness. Its sweet spot as a colorless liquid sets it apart from many strongly colored or odorous chemicals in its family. The way light travels through it shows off its low refractive index, a trait that tech scientists often chase for optical uses. There’s barely a whiff of odor, which is a relief in environments cluttered with strong-smelling reagents.
Toughness in a Bottle
This compound laughs off water; you could splash it around with little effect. Perfluorobenzene shrugs off polar or ionic substances, barely dissolving in water but blending well with other fluorinated solvents. That tendency extends to its chemical stability—the carbon-fluorine bond doesn’t break easily. I still remember how frustrating it was to coax any kind of reaction out of it during synthesis labs. For folks in the chemical industry, this resistance matters. You can use it as a solvent for aggressive acids or strong bases without fretting over breakdown and contamination.
The Physical Details Matter
The liquid boils at roughly 80 degrees Celsius, so it moves between states in a temperature range that doesn’t demand much energy. This low boiling point feels surprising—especially set against other aromatic compounds, which usually take a bit more heat to evaporate. The relatively moderate density, about 1.62 grams per cubic centimeter, means it feels a little heavy for a clear liquid, sinking quickly to the bottom if mixed with lighter compounds.
Its low flammability adds another layer of safety in the lab, especially compared with many common solvents that seem to burst into flames at the slightest spark. This gives researchers some breathing room, literally, and offers a safety edge in tightly packed labs. Adding to the safety story, perfluorobenzene doesn’t corrode glassware or standard polymers, which saves budgets and keeps experiments running smoothly across long semesters.
Why These Physical Properties Stand Out
Too often, chemical properties get discussed as technical trivia. In practice, these traits push innovation in research and manufacturing. Its chemical toughness underpins reliability in electronics manufacturing. Without it, cleaning intricate parts or prepping surfaces for special coatings gets risky. The stability also means less frequent solvent replacement, which helps keep hazardous waste in check—a lesson I picked up early, corralling bottles after an undergraduate organic synthesis marathon.
On the other hand, the resilience of perfluorinated chemicals carries an environmental price. They last for decades, sometimes centuries, in nature and can travel in water systems. Balancing their utility with their persistence requires careful thought. Some progress can come from greener alternatives—shorter-lived fluorinated compounds, improved recycling protocols, or targeted degradation strategies using advanced catalysis. In research and policy, the most promising steps involve honest discussions about trade-offs.
Final Thoughts on Responsibility and Opportunity
The quirky set of physical and chemical behaviors makes perfluorobenzene irreplaceable in specific corners of industry and research. At the same time, the community owes it to future generations to keep questioning how—and when—to use it responsibly. As the push for sustainability continues in laboratories and factories, the story of this clear, tough liquid echoes something many scientists already know: Nothing in the chemical world exists in a vacuum.