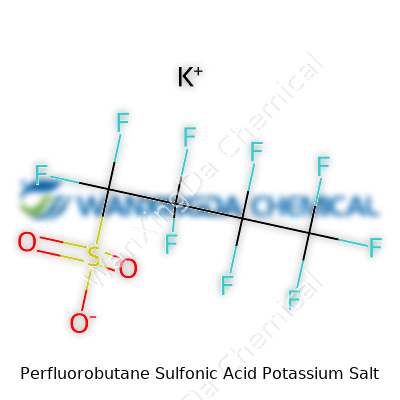
Perfluorobutane Sulfonic Acid Potassium Salt: Navigating Science, Safety, and Sustainability
The Long Road of Development
Perfluorobutane sulfonic acid potassium salt, known in laboratories and industrial circles by a host of aliases, didn’t spring into existence overnight. Its roots stretch back to the chemical innovations that followed the surge of fluorochemistry in the mid-20th century. Driven by a hunger for materials that could stand up to tough industrial scenarios—think harsh solvents, extreme heat, and aggressive acids—fluorinated compounds rose to prominence. Scientists figured out that adding a sulfonic acid group to a perfluorobutane backbone could create a molecule that strongly resists breaking down. The result? A substance that doesn’t flinch in applications that make other compounds wilt. Over the decades, as regulations tightened on older perfluorinated chemicals thanks to emerging toxicity studies, research landed on shorter chain alternatives, including this particular salt. Governments, researchers, and industry players soon found themselves in the thick of a debate about balancing utility and safety.
Product in a Nutshell
Potassium perfluorobutanesulfonate looks like a white or off-white powder, stable at room conditions, and highly soluble in water. It delivers on two fronts: acting as a robust surfactant and standing up to chemical degradation. These features make it a go-to for specialty applications, especially where other surfactants fail. The molecule’s structure—four completely fluorinated carbon atoms attached to a sulfonate group—sheds light on its strengths and the stubbornness that has led to environmental questions. Resisting breakdown feels like a benefit for industrial use, yet the trait haunts researchers tracking its footprint in water and soil.
Properties That Shape Its Role
The physical and chemical properties shape where this compound shows up. The strong C-F bonds—among the tightest in all organic chemistry—grant remarkable stability. As someone who spent years in a lab handling fluorinated agents, I remember these compounds laughing off heat and acid baths that would obliterate most organics. High solubility in water makes it travel easily through processing pipelines and, less helpfully, into natural waterways. The molecule doesn’t just float around untouched; it can interact with surfaces, proteins, and even cell membranes thanks to its ionic sulfonate group. I recall early discussions in our department about how this ionic end made certain PFAS detergents the backbone of tough cleaning fluids and electrochemical baths, yet also contributed to headaches for environmental chemists tracking persistence.
Technical Specs and Labels
Regulators force a close look at composition and purity partly because PFAS compounds—of which this potassium salt is part—carry a reputation for unwanted health and environmental effects. Specifications include content ratios, impurity thresholds, and physical state, often controlled tightly because even small traces can make a difference between safe use and regulatory nightmares. Labels go beyond chemical names, warning users about persistence and steps to avoid dust generation or environmental release. For those in research and industry, this is no minor detail; a missing warning or a misjudged level of purity morphs a chemical from tool to liability overnight.
How Preparation Shapes Outcomes
Making perfluorobutane sulfonic acid potassium salt usually means starting from perfluorobutanesulfonyl fluoride. This raw material reacts under carefully managed steps to form the acid, which then meets potassium compounds to create the final salt. Each batch needs close monitoring—residue from incomplete reactions, leftover fluoride, or cross-contamination draws the attention of both researchers and safety officers. In my experience, labs invest as much energy purifying and characterizing as synthesizing, trying to head off regulatory and safety complaints before they start. The tracks left by each batch last far beyond the laboratory or factory, a burden that folks don’t always consider while measuring out grams of white powder.
Chemical Reactions and Tweaks
Chemists can modify this molecule to change its behavior—adding different cations, tweaking the perfluorinated chain, or attaching new groups to tailor solubility or reactivity. Many modifications chase the hope of keeping the best traits—resistance, surfactancy—while dialing down environmental risks or biological activity. From where I stand, these efforts echo the story of many chemicals before it: the cycle of adoption, scrutiny, and then revision based on new evidence. Discussions at conferences or in industrial panels often circle back to the same challenge—every tweak to improve breakdown or lower toxicity risks killing the very performance that made the compound so useful in the first place.
Sifting Through the Names
Products of chemical innovation carry a confusing load of names, acronyms, and numbers. In the case of perfluorobutane sulfonic acid potassium salt, synonyms and product names often fill regulatory filings and academic papers. This naming maze frustrates practitioners and researchers trying to connect toxicity data with market products or shipment manifests. In my own work with chemical supply chains, tracking a substance required cross-checking what felt like a half-dozen trade names and regulatory classifications just to make sense of a shipment.
Staying Safe and Working Clean
Operational standards have caught up quickly, with handling procedures grounded in lessons from industries that saw the cost of ignoring PFAS problems. These steps include robust glove and mask use, extensive containment, and rigorous ventilation—standard fare now in both lab and industrial settings. Disposal and spill response now lean heavily on captured experience from chemical spills that fouled local streams or threatened worker health. Safety data sheets and workplace training hammer home the basics, since accidental exposure or environmental release lingers in the background of every job involving PFAS. I’ve seen risk assessments push projects to install new scrubbers or re-engineer waste treatment—all downstream of these potent salts.
Why the Industry Leans In
The edge that perfluorobutane sulfonic acid potassium salt offers in application areas keeps bringing new users to the table. Semiconductor makers value the chemical because it holds up in harsh processing and doesn’t foul circuits. Firefighting foam producers once relied on longer-chain PFAS for unbeatable performance and now look to shorter chains like this one as possible replacements. Even electroplating and specialty coating industries turn to these salts when old-school options fail. These industries don’t cling to the chemical out of nostalgia—they face relentless pressure for better purity, reliability, or environmental performance. Many have already faced scrutiny for historic PFAS use; driving change in these spaces comes less from government mandates than the real risk of consumer backlash or market loss.
Pushing the Science Forward
Research and development efforts home in on both improving the performance and reducing the risks. For years, basic research dug into degradation pathways, environmental transport, and health impacts, building a slow but steady understanding of what makes these molecules persistent and, at times, troublesome. Today’s research isn’t just about clever new varieties; whole labs and corporate research arms focus on detection, removal, and safer alternatives. Conferences buzz with ideas for capture resins, biodegradable surfactants, and even microbes that might one day munch away at PFAS, hoping to blunt their stubborn persistence. The science has shifted from simply celebrating breakthroughs to questioning how those same breakthroughs ripple into the real world—an evolution that matters as public trust and scrutiny ratchet higher.
What We Know About Toxicity
Toxicity drives much of the controversy. While early generations of PFAS set off alarms—bioaccumulation and links to cancer, immune issues, and developmental harms—the shorter chain salts like perfluorobutane sulfonic acid potassium salt prompt questions about whether shorter always means safer. Some studies see quicker elimination from the body and lower potential for building up, yet doubts linger about chronic effects at low doses or effects in vulnerable populations. The EPA and other regulators have demanded fresh studies, but for now, a sense of uncertainty reigns in the scientific community. Colleagues in toxicology remind me that even a faint shadow of toxicity sticks to commercial chemicals far longer than favorable technical assessments. For real risk reduction, transparency, disclosure of all study data, and independent oversight will matter more than promotional reassurances.
What’s Next?
The future for perfluorobutane sulfonic acid potassium salt looks unsettled. Knowledge grows by the week—and so does public awareness. Regulatory bodies have started aiming at entire classes of fluorinated chemicals, not just single compounds. Industry groups stress a shift to “safer chemistries,” yet the technical trade-offs sting for processes that can’t find drop-in replacements. In the coming years, progress will rest on three things: investment in non-fluorinated alternatives, ramping up proven capture and disposal methods for PFAS in the waste stream, and keeping accountability high—so that industries continue sharing research, risks, and results with regulators and the wider public. My own hope? The chemical community leans not just on technical fixes, but on ethical ones too—valuing public health and environmental stewardship as much as the next performance leap.
A Chemical That Sticks Around
Perfluorobutane sulfonic acid potassium salt, known by the shorthand PFBS K-salt, pops up in all sorts of industrial applications. From my experience covering environmental stories, I’ve noticed the conversation around PFBS ramp up over the past few years. The reason has less to do with its usefulness in manufacturing and more to do with the attention paid to chemicals that fall into the “forever chemicals” category. These are substances that hang around in the environment for a long time without breaking down.
The Everyday Uses—And the Risks
PFBS K-salt helps products repel water and oil. It supports firefighting foams, makes textiles stain resistant, and helps electronics stay dust-free. Its chemical makeup brings a level of reliability and consistency that’s tough to substitute. Walking through any supermarket or hardware aisle, you see the end results of these technologies—stain-resistant carpets, water-repellent shoes, grease-resistant food wrappers. Manufacturers keep reaching for PFBS K-salt because it gets the job done inexpensively and on a mass scale.
On the health side, studies have drawn links between longer chain relatives—like the notorious PFOA and PFOS—and some negative effects like cancer, developmental delays, and immune system quirks. PFBS, as a shorter chain alternative, entered the scene as an option with less tendency to build up in the body. Scientists note that it doesn’t stay in human blood as long as the older chemicals, which seemed like a win in terms of trade-offs. Still, researchers have found PFBS in water sources and even in wildlife, which signals it’s moving through ecosystems in ways that weren’t expected just a decade ago.
Balancing Progress and Responsibility
Companies once leaned heavily on perfluorinated compounds for a sense of certainty in how their products performed, especially in the firefighting world and in semiconductor production. Tightening regulations and mounting pressure from consumers have forced businesses to look at alternatives. PFBS seemed, for a time, like a safer bet. Yet, the sheer persistence of these substances and their ability to travel far from where they started makes me uneasy. The pattern has been to swap out one persistent chemical for another, banking on slight changes in biology to curb the risk. So far, that approach hasn’t closed the door on environmental or health concerns.
Moving Past “Forever” Chemicals
The solution lies in a mindset shift. Instead of defaulting to chemicals that bring hard-to-remove effects, a combination of innovation, transparency, and stronger safety tests could point the way forward. Investing in research for safer substitutes pays off in more ways than just good optics. Down the road, stepped-up oversight combined with full ingredient disclosure will protect the communities closest to factories and waste sites. Without outside pressure, big organizations don’t always act fast enough on their own. Stricter regulations on water testing and discharge from facilities would help keep PFBS from quietly piling up in rivers and taps.
The past few years taught me that relying on “shorter chain, maybe safer” is a risky gamble when it comes to public health. A smarter bet—no matter how complicated it feels at first—asks more of chemical manufacturers from the start, rather than waiting for a new problem to show up where people live and work.
What We Know About This Substance
Perfluorobutane sulfonic acid potassium salt lands under the family of PFAS—per- and polyfluoroalkyl substances. Many people recognize “PFAS” because words like “forever chemicals” show up in the news, and for good reason. These chemicals don’t break down in the environment, or in our bodies, for a really long time. That means if a spill happens, or it gets into drinking water, it doesn’t disappear.
Researchers and governments raise eyebrows about several PFAS because of links to health problems. I remember reading about blood tests that found PFAS in just about everyone. The scary part comes from animal studies, showing issues like immune system effects, thyroid disruption, and links to cancer. The body of evidence typically piles up around some of the better-known PFAS, like PFOA and PFOS. Perfluorobutane sulfonic acid potassium salt isn’t as famous, but it shares many chemical traits with those compounds.
Current Views On Health Hazards
PFAS, including perfluorobutane sulfonic acid potassium salt, stick around in water, soil, food, and human bodies. Researchers and public health officials keep asking: if they look similar, do they act similar? Right now, available studies suggest the potassium salt version resists breaking down as well as its cousins. A handful of animal experiments show the potential for liver and blood changes, raising suspicion about long-term low-level exposure.
Detailed answers from human data still lag behind. Large health studies focus more on PFOA and PFOS, so the full extent of risk from perfluorobutane sulfonic acid potassium salt hasn’t grabbed as many headlines. But history with chemicals often repeats itself. Often, regulators and doctors realize risks only after years—sometimes decades—of people living with low-level exposure.
Why This Subject Matters
Few folks know the names of industrial chemicals traveling through groundwater or coming out of manufacturing sites, but their effects turn personal pretty quickly. PFAS pollution made headlines across the world—communities from Michigan to Europe dealing with contaminated water, lawsuits, and calls for banning certain substances.
The health value in steering clear of PFAS overuse seems clear. Studies led the European Food Safety Authority and the US Environmental Protection Agency to warn against high levels of these chemicals, even at trace amounts. My experience as a journalist following contamination stories taught me that trust breaks down once pollution stories break. People want safe water, clean food, and honest answers about what’s in the environment. That basic need links everyone—whether in rural America or industrial Europe.
What Makes Sense Going Forward
Facing a chemical with unclear health data, the common lesson is caution, not panic. Industries must shift away from persistent chemicals. Politicians need to set real limits, not just recommendations. Companies should identify safer alternatives, and scientists must keep pushing for better answers. People at home can’t test their own water for every PFAS, so they rely on stronger laws and clear labelling about contamination.
Perfluorobutane sulfonic acid potassium salt deserves scrutiny. It belongs to a chemical class with a troubling track record. Until governments catch up, avoiding unnecessary PFAS exposure is the safest bet. Community voices, consumer groups, and clear science can all help keep decision makers honest. Every parent, worker, and neighbor deserves water that won’t quietly carry a lifetime’s worth of chemicals.
Why Knowing the Formula Matters
Most folks glance at chemical names and their eyes glaze over. If you’re working in manufacturing or chemistry, though, the details behind something like Perfluorobutane Sulfonic Acid Potassium Salt—known in the scientific world by its formula, C4F9SO3K—carry some real weight. This compound has turned up in industrial and environmental discussions, not only because of what it does, but because of where it ends up.
What That Formula Tells You
Looking at C4F9SO3K, you spot a backbone loaded with fluorine and a sulfonic group tied to potassium. Chemists know this kind of structure delivers serious stability; fluorine grabs on and won’t let go. That means products using this salt—think cleaners, firefighting foams, coatings—usually stick around. Too well, actually.
I remember seeing how stubborn these compounds can be during some fieldwork near an old training site. Decades after the last foams got hosed onto the concrete, traces still turned up in groundwater tests. Once these perfluorinated compounds break loose in the environment, they don’t break down like most organics do. The formula looks tidy on a blackboard, but it raises big flags outdoors.
Why Regulators and Communities Watch Closely
Scientists and health officials have placed the broader group—PFAS, or per- and polyfluoroalkyl substances—under the spotlight because of studies linking long-term exposure to health issues. Potassium salts like this one have shown up in soil and water in places far from the factories that made them. That gets people worried for good reason.
The challenge comes in cleaning up or even tracking perfluorobutane sulfonic acid potassium salt. The stable bonds that chemists prize for industrial use also resist breakdown by natural forces. Activated carbon and advanced filtration help a bit, but no silver bullet has emerged. Tackling this is neither cheap nor quick.
Industry’s Place and Possible Paths Forward
Companies haven’t ignored the mounting evidence. Some have started shifting away from these PFAS chemicals, not only due to regulations but because customers and citizens want safer, cleaner options. There’s room for better transparency in what goes into products, how chemicals get managed, and what gets released. Peer-reviewed research keeps uncovering ways to treat and contain these compounds, but at the end of the day, basic process changes sometimes make the biggest dent.
I’ve seen firsthand what it’s like to explain a complex formula to a community worried about what’s in their water. The public deserves straight answers about what’s present and what’s being done to protect their wells and streams. Scientists, companies, and regulators all share a role here. The formula for perfluorobutane sulfonic acid potassium salt—C4F9SO3K—might seem academic, but the ripples from the way these substances behave reach far beyond the pages of a chemistry textbook.
Most folks don’t walk into a supply room and see a bottle labeled Perfluorobutane Sulfonic Acid Potassium Salt. For chemists and environmental professionals, though, handling this compound becomes a regular part of the job. It’s not just another chemical to put on a shelf—this one comes with some serious responsibility.
Why Storage Protocols Aren’t Just Red Tape
When I started handling PFAS chemicals in the late 2000s, companies were waking up to the idea that sloppy storage could mess with your results and, much worse, endanger your team. Perfluorobutane Sulfonic Acid Potassium Salt falls into the PFAS category—a class of chemicals built to last, both in products and unfortunately, in the environment. These substances don’t break down easily, which makes them useful and problematic in equal measure.
Health authorities like the EPA and European regulators have flagged PFAS for their persistence and potential impact on everything from wildlife to human health. That means the way this chemical sits on your shelf matters just as much as how it’s used. A botched storage setup leads to contamination scares, regulatory headaches, and sometimes huge cleanup bills. I’ve seen labs waste costly reagents—not because they used them, but because of careless capping, exposure, or leaks.
Safe Storage Isn’t Fancy—It’s Practical
After years of stepping over chemical drums and working next to battered cabinets, I’ve learned a few hard truths. Perfluorobutane Sulfonic Acid Potassium Salt sits best in a cool, dry place, far from direct sunlight. Moisture invites trouble; this salt likes to draw in water from humid air, and nobody wants a clumpy, degraded bottle on their hands.
A tightly sealed container does more than just “look” secure. It keeps out air, humidity, and stops fumes from escaping into the storage room, which could pose problems for people working nearby. Think about clear labeling too—I once grabbed what I thought was sodium chloride, only to find out it was a far nastier compound with similar crystals. Lab mistakes like these don’t just waste time—they create real hazards.
Storing any PFAS, this salt included, needs a designated area away from acids, bases, or substances that might cause a reaction. Flammable cabinets aren’t the right home for this type of compound. I’ve seen some labs keep PFAS chemicals in general storage, but that raises the risk of accidental mixing. Cross-contamination makes lab tests unreliable and can even invalidate entire research projects.
Regulation and Training Go Hand in Hand
Safe storage isn’t just about racks and bins. Without regular staff training, even the best storage plan fails. I make a point of walking new hires through chemical storage every quarter, emphasizing why PFAS need a separate space. A simple poster in the storage room listing incompatible substances and emergency procedures can make all the difference during an accident. Mining regulations and guidance from organizations like NIOSH brings an extra layer of safety—these rules are based on lessons learned the hard way, not theory.
More companies and research facilities are realizing that regular inspections help spot cracked lids, spills, or expired chemicals before they become bigger problems. Periodic checks turned up a leaky bottle in our storeroom last year—if we’d missed it, cleanup could have run into the thousands.
Room for Improvement
Many workplaces still treat chemical storage as an afterthought, only giving it attention after a scare. It shouldn’t take a near-miss to spark a change. I advocate for keeping PFAS chemicals like Perfluorobutane Sulfonic Acid Potassium Salt in their own clearly labeled cabinets, with inventory logs updated every time a bottle goes in or out. Tracking what’s in storage and who handled it last provides a real-world safety net. The science keeps evolving, but respect for proper storage remains a backbone of chemical safety in any setting.
Staying Real About Hazmat Precautions
Years ago, I watched a seasoned chemist get a painful chemical burn from a simple oversight—she picked up the wrong bottle and didn’t realize until it was too late. That image comes to mind every time I see anyone work with compounds like Perfluorobutane Sulfonic Acid Potassium Salt (PFBS K-salt). These chemicals seem as common as table salt in the industrial world, but ignoring what they can do to skin, eyes, or lungs invites trouble that lingers for months—or longer.
I’m no stranger to lab protocols, but PFBS K-salt makes me extra cautious. The compound belongs to the notorious PFAS family, known as “forever chemicals” for good reason. These substances resist breaking down, persist in the environment, and stick around in our bodies over time. Not only do they pose a risk during handling, worries extend to what happens after they leave the lab. Improper management doesn’t just affect workers—it can turn into a problem for neighbors and drinking water miles away.
Suiting Up is Non-Negotiable
The urge to skip gloves or a face shield just for a quick pour hits everyone once in a while. The truth is, there’s never a good excuse to go barehanded with PFBS K-salt. Splash goggles, nitrile gloves, and lab coats form the first line of defense. I’ve seen nitrile outperform latex in these situations, staying intact after accidental splashes. Chemical-resistant aprons and even boot covers make sense if you deal with larger batches or risk spills.
Lab ventilation pulls more weight here than just “freshening the air.” Local exhaust systems, like properly placed fume hoods, cut down on inhalation risk. Even dusty residues can pose an invisible hazard, sneaking past simple masks. Respirators specifically rated for chemical use aren’t overkill—no one goes home bragging about developing a chemical cough.
Cleaning Up—And Not Just for Appearances
Once, I watched a new technician ignore a powdery spill, thinking a little dust couldn’t hurt. An hour later, his gloves showed clear signs of corrosion—a warning that PFBS K-salt bites hard, even in small amounts. Any chemical that upsets proteins in the body deserves full respect. Absorb spills using proper materials—no paper towels or rags that go straight to the trash. Seal up contaminated waste tight and label it clearly for hazardous disposal. PFAS waste calls for disposal methods that keep it from leaching out of landfills or sewage systems.
Regular training makes all the difference. Once a year isn’t enough. Short refreshers every few months help keep everyone’s memory sharp—especially since new faces join labs and others slip into bad habits over time. A culture that encourages questions, rewards diligence, and keeps nobody above the rules serves everyone in the long run.
Respecting the Long Game
Regulators like the EPA have ramped up scrutiny on PFAS chemicals. That’s not bureaucracy run amok—it’s the direct result of seeing toxic impacts on entire towns from careless dumping or accidental releases. PFBS K-salt doesn’t show mercy, either in the short-term on skin or eyes, or in the long-term as it infiltrates water supplies and food chains.
Getting this right means taking everyday steps seriously and looking out for one another in labs, factories, and waste facilities. I’ve learned that the ones who ask extra questions or double-check their gear aren’t paranoid—they’re protecting everyone, inside and outside the lab. Anyone dealing with PFBS K-salt owes that much to the next person picking up the bottle.