Perfluorobutanesulfonyl Fluoride: Chemistry in a Changing World
Tracing the Path: History Shaped by Curiosity and Industry
Interest in fluorinated chemicals took off along with the chemical industry’s growth through the 20th century. Perfluorobutanesulfonyl fluoride, often abbreviated as PBSF, did not appear on the scene overnight. Its roots can be found in the post-war age when scientists pursued ever-more robust and stable compounds, searching for molecules that could withstand fierce heat, aggressive solvents, and harsh chemical processes. As fluoro-organic chemistry matured, the singling out of sulfonyl fluoride groups represented a moment where industrial chemistry and scientific curiosity collided. Researchers realized that sticking perfluoroalkyl tails onto an already tough sulfonyl fluoride group gave them products with almost unmatched resistance to breakdown. This wasn’t just solving academic challenges. The resulting molecules became the building blocks for a new wave of performance-enhancing agents across electronics, coatings, and even pharmaceuticals. The arrival of PBSF, sometimes known under alternative names like perfluorobutanesulfonic acid fluoride or PFBuSF, marked a checkpoint for the industry, bringing a mix of promise and concern.
A Close Look at the Molecule
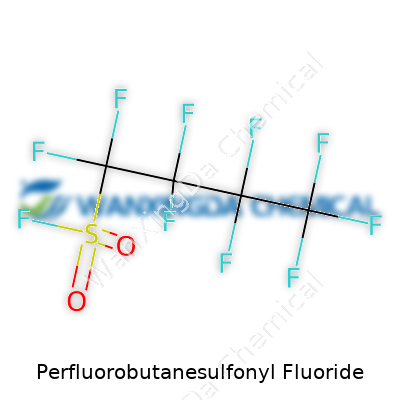
PBSF stands out due to its highly stable carbon-fluorine bonds paired with a reactive sulfonyl fluoride head. The clear to faintly yellowish liquid brings expectations for persistence: it resists heat, repels water and oil, and maintains structure in hostile chemical environments. With a molecular formula of C4F9SO2F, it displays volatility without going overboard—evaporates with a distinct odor, clings to glassware, and ignores most attempts to burn, thanks to that perfluorinated backbone. What matters here is its ability to withstand both acids and bases without losing its identity. For a chemist, this is both a blessing and a curse. It makes the compound wildly useful but also tough to get rid of.
Technical Specifics and Labeling: Reading Between the Lines
Labeling for chemicals like PBSF goes far beyond hazard pictograms. Technical data sources stress the need for high purity: small traces of non-fluorinated material can throw a synthesis off and skew results. PBSF’s uses depend on that molecular precision. It stays stable under room conditions but reacts sharply with nucleophiles, a trait that users leverage when they want to introduce a sulfonyl group in complex molecules. Color-coded labels flag it as a corrosive and an irritant, which matches my experience: open a bottle carelessly, and you’ll regret it for hours. Regulations grow stricter each year as global agencies scrutinize perfluorinated substances—safety data shifts, but the need for clear, informative labeling stands unchanged. Mistakes often sprout from ambiguous containers or missing hazard info, so any operation handling PBSF pays close attention to this.
Born in the Lab: Preparation Routes and Methods
Not every laboratory or plant wants to deal with the headache of making PBSF from scratch. Traditionally, chemists fluorinate the butanesulfonyl chloride or another related precursor, usually by exposing it to elemental fluorine or specialty metal fluorides in sealed reactors. Reagent choices affect yield, purity, and downstream handling risks. The energy needed for that many C–F bonds doesn’t allow for shortcuts or improvisation. Most labs would rather purchase PBSF from a specialist supplier than build the infrastructure for direct synthesis. From what I’ve seen, even experienced chemists respect the dangers of stray fluoride ions and potent byproducts. Each step drags questions about disposal and air quality, hanging over every batch run. After all, there’s good reason why PBSF shows up more often in chemical catalogs than in undergraduate teaching sets.
It’s Alive: Chemistry and Reactions
Here’s where PBSF justifies its seat at the table. That sulfonyl fluoride group proves valuable for making sulfonamides, sulfonate esters, and other derivatives prized in polymer chemistry and pharmaceuticals. Toss it into a reaction with an amine, and out comes a perfluorobutanesulfonamide. Expose it to strong bases, and you can swap out the fluoride for other leaving groups, tailoring it for specific projects. Its radical stability and resistance to harsh conditions let it hang on during steps that would wreck other sulfonyl analogs. Real-world labs use PBSF for fluorine chemistry that demands both a stubborn backbone and an optionally reactive end. Nobody with experience mistakes it for a toy: misjudged conditions yield violent reactions, unforgiving fumes, and complicated cleanup.
What’s in a Name? Synonyms and Nicknames
Confusion follows perfluorinated chemicals at every turn thanks to confusing trade names, abbreviations, and product labels. PBSF gets called perfluorobutanesulfonyl fluoride, perfluorobutanesulfonic acid fluoride, and—sometimes—PFBuSF or C4F9SO2F. Chemists trading notes complain bitterly about mislabeling, and a single typo can send a project sideways. Regulatory listings don’t always line up, which adds another layer of frustration for anyone navigating customs, international regulations, or research partnerships. Everyone in the laboratory values clear, unambiguous communication, especially for a molecule with hazards that demand respect.
Staying Safe: Operations and Standards
Safety is never someone else’s job around PBSF. Experience tells me that relying on luck leads to accidents. Splash-proof goggles, heavy gloves, solid ventilation, and sealed containers are the bare essentials. Regular audits and hands-on training matter more than paper checklists. Fumes from PBSF and its breakdown products can irritate lungs and cause lasting harm. Spills demand fast, knowledgeable containment and disposal procedures. Regulations keep updating: some regions tighten controls on airborne releases, disposal protocols, and personal exposure. The best lab managers don't rely on last year’s protocol—active risk assessments and up-to-date hazard reviews shape daily routines. Getting trusted, complete safety data out to everyone on the floor isn’t an option, it’s the starting point.
Where It Shows Up: Applications That Matter
PBSF’s reputation as a starter for advanced chemicals spans the performance coatings world, specialty plastics, and even the semiconductor sector. In making fluorinated surfactants, the molecule pops up as a precursor where durability and resistance win out over price or ease of disposal. Environments that chew up standard chemicals—think jet engines, advanced microchips, fire-fighting foams—rely on perfluorinated chemistry’s robustness. PBSF plays an unglamorous but vital role: it does the job that no other group of molecules can. That persistence delivers reliability, but it’s a double-edged sword. Downstream users face uncomfortable discussions about environmental build-up and tough cleanup costs. Cities, factories, and even rural communities see these molecules show up in water, air, and soil, sparking long-term questions about responsibility and stewardship.
Looking Ahead: R&D and Innovation
Modern R&D labs see both problems and promise in PBSF and its cousins. Researchers push for alternatives that match performance while breaking down faster in the environment. Studies chase ways to degrade sulfonyl fluoride groups after their useful life, turning the tide against decades of persistence. It’s a race that needs new reactions, better catalysts, and fresh thinking. At conferences, you hear frustration and hope in equal measure. Some research groups develop trapping agents and filter technologies that lock up stray molecules. Startups look for entirely new classes of surfactants, gambling that green chemistry can outpace regulatory crackdowns. Inside every team tackling PBSF sits a blunt math problem: can humanity keep the benefits of ultra-stable molecules without saddling future generations with permanent problems?
Toxicity: An Unfinished Verdict
Talk to people outside the chemical sector, and PBSF’s danger sounds simple: toxic, persistent, best avoided. Reality smudges the line. Animal and cell studies show that, much like other perfluorinated compounds, PBSF binds to proteins in blood and can bioaccumulate. The problem is not just acute poisoning but a slow, stubborn buildup over time across food chains. Scientists debate links to cancer, immune disorders, and reproductive harm, but the data don’t always agree. Regulators worldwide now err on the side of caution, driving manufacturers to invest in clearer labeling, better emissions controls, and serious toxicity studies. For workers and communities, there’s little satisfaction in uncertainty—testing every exposure, tracking long-term outcomes, and sharing risks as public science, not private secrets, is the only path that earns public trust.
Forward Motion: Prospects Shaped by Experience and Responsibility
PBSF stands at a crossroads shaped by decades of industrial growth, hard-won scientific knowledge, and mounting environmental pressures. Experience shows the industry moves only as fast as its sense of urgency and the laws that hold it to account. Future prospects for PBSF depend on whether leaders respond to today’s scrutiny with innovation instead of delay. Safer alternatives, greener methods, and open science all point toward a needed shift. Researchers and workers, taught by past mistakes, now look beyond the next batch or quarterly report—they remember that cleanup, accountability, and trust outlast the utility of any one chemical. PBSF may not grab headlines on its own, but the conversation about how society manages chemicals like it shapes everything from public health to industrial progress.
Some chemicals float under the radar until their true impact comes out in the open. Perfluorobutanesulfonyl fluoride, or PBSF, fits in that category. Known mainly inside chemical production plants and research labs, PBSF rarely makes headlines, but its ripple effects stretch far beyond those walls.
What PBSF Actually Does
PBSF plays a behind-the-scenes role in manufacturing. Companies use it to help produce specialty fluorinated materials—think advanced plastics, coatings, and water-resistant fabrics. PBSF serves as a starting block in creating chemicals that help everyday products shrug off stains and repel water. These “fluorinated” products show up on carpets, outdoor gear, and some food containers.
Chemists lean on PBSF when they need a building block that won’t react with much else except under tightly controlled conditions. Its structure lets researchers bolt on tough, water-shy groups to other molecules. That means more resilient end products—harder coatings, stubborn stains, gear that lasts longer in bad weather.
Concerns That Go Beyond the Lab
The trouble with PBSF, and many compounds in its chemical family, stems from their environmental journey. Once these chemicals escape the factory, nature struggles to break them down. They can stick around in soil, waterways, and even the animals and people living nearby. That’s not just theory; several studies in the past decade have flagged fluorinated compounds for showing up in drinking water, fish tissue, and human blood samples across the globe.
No one sets out to pollute, but sometimes industry outpaces oversight. For years, regulatory agencies have caught up only after routine use of such compounds left fingerprints in places they didn’t belong. In my own neighborhood, a small spill from an old plant took years to clean up, mainly because regulators didn’t expect the chemicals to move so fast or to hold on so tight once in the ground.
Steps Toward Safer Use
Industry relies on PBSF and related chemicals for good reasons—there isn’t an obvious substitute for every use case. But that doesn’t mean standards should sit still. Companies can rethink how much PBSF they need, tighten up where and how they store and dispose of it, and keep a closer eye on how much slips away into the environment.
Product designers and buyers also have a role to play. If water-resistant gear or stain-proof carpets matter, it’s fair to ask companies if they use cleaner alternatives or have strong containment protocols. Governments ought to step up, reaching for science-based regulations that don’t just react to the latest incident but look ahead at the chemicals coming down the pike.
Public pressure can change boardroom math. Customers asking tough questions about how things are made—whether it’s a raincoat or a pizza box—push brands to look deeper into their supply chains. That may sound idealistic, but we've seen real change in the past with other persistent chemicals. Progress takes clear rules, honest disclosure, and investment in safer options. Without that, the hidden story of PBSF and its cousins keeps leaking into places it never belonged.
A Closer Look at the Formula: C4F9SO2F
Sometimes, it’s easy to get lost in the jargon of chemistry, but the chemical formula for perfluorobutanesulfonyl fluoride — C4F9SO2F — carries a story that stretches far beyond the walls of a laboratory. As someone who’s spent time both in research and in classrooms, I’ve seen firsthand how a single compound can spark conversations about environment, health, and responsibility. This formula looks simple, but it points to a bigger picture.
Perfluorobutanesulfonyl Fluoride: Not Just Another Chemical
The moment you see all those F’s in the formula, you know you’re dealing with a perfluoroalkyl substance, part of the wider PFAS family. That means strong carbon-fluorine bonds. In plain terms: they don’t break down easily. I remember chatting with an environmental chemist a few years ago, and his message was clear. These chemicals last, practically forever, in soil and water. Perfluorobutanesulfonyl fluoride is no exception. It’s used to make specialty chemicals, surfactants, and can end up in products that people touch every day.
Why It Matters to Real People
People rarely stop to think about the invisible progress of a chemical from the factory line to their kitchen table. But this story has played out over decades now. These PFAS chemicals, including perfluorobutanesulfonyl fluoride, can travel through air and water, and sometimes, they accumulate where they’re not wanted — in lakes, in fields, and sometimes in us. Studies from the CDC and EPA have turned up PFAS in drinking water supplies across the United States. Even if perfluorobutanesulfonyl fluoride isn’t topping the list, its molecular cousins do show up. As of 2024, many groundwater samples in industrial zones still show traces of various PFAS compounds. They’ve become the “forever chemicals” in news cycles, with good reason.
Health and Environmental Concerns
I’ve spoken to parents, teachers, and small-town leaders — most of them never heard of this compound. But they do worry about cancer rates, birth outcomes, and chronic disease. Some studies link high levels of PFAS exposure to health issues, though researchers still debate details and thresholds. What’s not in question: these chemicals last a long time, and cutting exposure matters. Regulatory agencies in the US, the EU, and parts of Asia have started taking serious steps, including drinking water limits and tighter industrial rules. Research from Harvard and other universities keeps building the case for action. We need more data, but we already know enough to seek safer paths.
Ways Forward
The problem feels overwhelming at first, but there are lessons to draw. Alternatives now exist for many of the functions these chemicals provide. Manufacturers have begun switching to less persistent compounds. In my own experience working with industrial partners, I saw firsthand how public pressure and collaboration can nudge companies away from legacy chemistry. Municipalities have invested in better filters and monitoring for drinking water. Even small towns, working with universities, are finding new tools for PFAS clean-up using activated carbon, advanced oxidation, or reverse osmosis. Each step helps.
Knowledge and advocacy matter. Knowing the formula C4F9SO2F helps shine a light on the chain that links chemistry, policy, and daily life. For me, the story of perfluorobutanesulfonyl fluoride stands as a reminder: chemistry shapes the world, and it’s up to all of us to ask questions and push for better answers.
Understanding the Stakes with Fluorinated Chemicals
Perfluorobutanesulfonyl fluoride, or PBSF, belongs to the family of per- and polyfluoroalkyl substances, better known as PFAS. Hearing “PFAS” sends up a red flag for good reason. For decades, PFAS earned a reputation as “forever chemicals” because they stick around in the environment and resist breaking down. My years following chemical safety issues taught me that the smallest details can shape big environmental outcomes, especially where fluorinated compounds are involved.
Evidence of Hazard: What Research Shows
If you ask about PBSF’s toxicity, the alarm bells don’t ring quite as loudly as they do for some older PFAS like PFOA or PFOS, which came under sharp scrutiny and bans in parts of the world. Still, caution is the name of the game with PBSF. The structure carries the same sturdy chemical bonds, meaning it won’t simply wash away or disappear anytime soon. Scientists found that some PFAS disrupt hormones, cause organ damage, and linger in human blood. Specific studies on PBSF suggest its breakdown products can be persistent and have some toxicity in living organisms. That should prompt serious reflection on its use and disposal.
Why We Should Care
Nobody wants a pile of jargon or abstract risk. The concern with PBSF boils down to where the chemical goes once it’s released. PFAS waste doesn’t vanish. Reports from communities near PFAS-emitting factories show contaminated drinking water, with families facing tough questions about what’s safe to eat or drink. PBSF gets used as a synthetic intermediate, so it pops up in the manufacture of other chemicals and specialty products. Exposure might come for workers who handle it directly or through downstream environmental spread, especially if waste gets mismanaged.
The story’s not just about immediate danger, but about what builds up. Even if PBSF is less bioaccumulative than some other PFAS cousins, it doesn’t mean we get a free pass. Regulators have started tracking and restricting entire PFAS groups, signaling that waiting until the next health study isn’t the best approach. In the past, slow regulatory response meant communities had to live with contaminated soil and water for years.
Better Paths Forward
Modern industry faces a challenge balancing performance with precaution. In the places I’ve seen with strong chemical controls, progress comes from simple ideas: keep exposures low, switch to safer alternatives where possible, and prioritize clear information over technical loopholes. Regulators in the US, Europe, and Asia have begun to look at PFAS more broadly, using group restrictions rather than acting chemical by chemical. This approach nudges companies to step back and rethink the legacy of their products before the damage spreads too far.
Waste treatment needs upgrading, too. Incineration, for example, must hit high enough temperatures to break those stubborn bonds, or else PFAS slips right through. Community watchdogs, independent scientists, and unions can push for honest disclosure. When I see industries and governments working with local communities, it almost always drives lasting progress over flashy promises.
The Real Test of Responsibility
Perfluorobutanesulfonyl fluoride may sound like a niche concern, but its story stands for how we deal with chemical risks as a society. If history with PFAS tells us anything, it’s that waiting for disaster before acting never ends well. Today, there’s enough science, enough warning signs, and enough public demand to raise the bar for chemical stewardship. Switching to safer chemistry, enforcing accountability, and giving communities a real say will decide if we learn from the past or keep repeating it.
Getting Serious About Chemical Safety
Perfluorobutanesulfonyl fluoride isn’t the sort of compound you just toss on any warehouse shelf. My experience working with specialty chemicals has taught me that even a small error in storage or handling sets the stage for accidents. Keeping everyone safe in a lab or production facility always ties back to basic respect for chemical properties. Companies sometimes dismiss the importance of storing chemicals correctly, but the long-term costs show up in damaged equipment and, sadly, in personal injuries.
This particular compound falls under the class of perfluorinated sulfonyl fluorides, notorious for their reactivity and persistence. Perfluorobutanesulfonyl fluoride easily reacts with water and strong bases. Once moisture gets into the container, you wind up generating toxic and corrosive byproducts. Every chemist I’ve spoken with agrees: don’t let your guard down around moisture-sensitive reagents. Once, I saw a senior chemist double-bagging a bottle, only to discover the inner cap wasn’t tight – a stubborn lesson learned after corrosion ruined several hundred dollars in reagents.
What Secure Storage Looks Like
Solid practice starts with a tight, properly labeled container, sealed immediately after use. You want these bottles kept in a cool, dry, and well-ventilated area. Locked cabinets with chemical resistive trays offer another layer of protection, not only from curious hands but from the kind of slow leaks that can ruin a whole inventory. I once watched a chemical-resistant tray save an entire lower shelf from a slow drip, which would have otherwise forced us to dispose of a dozen unrelated compounds.
Light and temperature matter more than you’d think. UV light causes some perfluorinated compounds to break down or change over time, so storage away from windows pays off over the years. Heat speeds up decomposition and raises the risk of pressure building in sealed bottles. In one lab, failure to control for ambient heat led to a minor bottle rupture – while it didn’t cause injury, it added a chemical smell that lingered for days. A dedicated chemical fridge at around 2–8°C strikes a good balance, especially in warmer climates or facilities with fluctuating HVAC.
Handling—A Ritual, Not a Shortcut
Personal experience proves that shortcuts during handling always catch up with you. Nitrile gloves and goggles sound simple, but sweat or a small tear turns basic PPE into a liability if you’re not focused. Fume hoods aren’t just for exotic reactions—transferring any reactive liquid there keeps fumes from escaping into breathing zones, especially with potent fluorinated vapors. Pouring or pipetting outside a hood should never become routine, no matter how experienced the staff.
Spill response should stay top of mind. You don’t want to improvise after a spill of perfluorobutanesulfonyl fluoride; those moments mean people scramble, and mistakes compound. Spill kits, dedicated absorbent pads, and clear posted instructions put people in control, not guessing what to do.
Solutions for Ongoing Safety
Companies and labs ought to invest in regular training – not just once-a-year seminars, but short, practical refreshers that stress real stories and everyday risks. Relying on a laminated MSDS taped to the wall hasn’t saved a single hand from chemical burn in my experience. Involving the whole team in inspections and storage audits turns safety from a compliance chore into a routine habit. Dedicating space and time for safe storage isn’t just “good housekeeping”—it’s smart risk management.
Storing and handling perfluorobutanesulfonyl fluoride safely never looks flashy. It boils down to respect for the chemical and an honest assessment of your team’s habits. Fact-based protocols and shared vigilance make all the difference.
People working in chemical labs or material science know that chemicals don’t all come in a one-size-fits-all package. Few substances make this as clear as perfluorobutanesulfonyl fluoride, a compound with specialized uses in sectors like pharmaceutical synthesis and electronics. On paper, it’s just another reagent, but the way it gets from warehouse to bench makes a big difference to outcomes, costs, and even safety. That’s where packaging size steps in as more than just a logistical footnote.
Common Options and What They Mean for Users
From what I’ve seen in research and supplier catalogs, perfluorobutanesulfonyl fluoride usually lands in glass bottles and HDPE containers. Volumes stretch from a few grams up to several kilograms. Small packs—say, 10 grams, 25 grams, or 100 grams—show up often, especially for universities and R&D labs. Scale matters here. Nobody in an academic setting wants to store excess hazardous material. Waste is costly, shelf life is limited, and unwanted exposure keeps EHS officers up at night.
On the industrial side, buyers ask for larger amounts. Kilo-scale packs—1 kg bottles, 5 kg drums, or even more for ongoing production—turn up reliably in procurement requests. Shipping larger volumes saves money per unit, but storage conditions and risk management get tricky. That’s an old conversation between lab managers and purchasing teams, and it rarely ends in a single answer. Everyone has war stories about the time a drum exceeded available shelf space or a sudden procedural switch made the whole lot obsolete.
Why the Range Exists
Chemical suppliers didn’t stumble on this range by accident. There’s always a balancing act between keeping stock fresh and making sure buyers aren’t stuck with material quantities they can’t store or use quickly. Some researchers still remember times they poured excess, expired specialty chemicals down designated waste because only jumbo bottles were offered. The cost—both financial and environmental—sticks with you, especially as sustainability reporting ramps up industry-wide.
Impact on Research and Industry
In my own time in lab settings, those small bottles meant more flexibility. Tight budgets could stretch further, and it got easier to rotate chemicals in and out of stock as project scopes changed. On the manufacturing side, the calculation changes. Shaving pennies off per gram by buying in bulk sounds smart, but only if safety, consistent storage conditions, and turnover rates keep pace.
Several major vendors now listen more closely to customer preferences for these reasons. Feedback loops opened as buyers pushed for more varied packaging options. This isn’t just about convenience—smaller bottles cut waste and lower environmental risks in places without specialized chemical storage. For high-throughput settings, being able to order one or five kilograms at a time streamlines procurement and planning. Nobody wants workflow clogged because the only available pack size was either too small or too large.
How to Improve the Packaging Landscape
There’s room to do better. More transparency helps a lot—consistently publishing available sizes, clear shelf-life information, and honest lead times would prevent confusion. Better collaboration between chemical distributors and end users could spark innovation in both package design and pricing. Exploring returnable or recyclable packaging could keep waste down for both lab-scale and industrial buyers.
Ultimately, the exact packaging sizes for perfluorobutanesulfonyl fluoride keep evolving as applications in research and manufacturing grow. People need flexibility, suppliers need accountability, and both sides rely on mutual understanding. Evidence-driven decisions lead to safer, more efficient labs and plants. Choice in packaging isn’t a luxury; it’s a baseline requirement in modern science and industry.