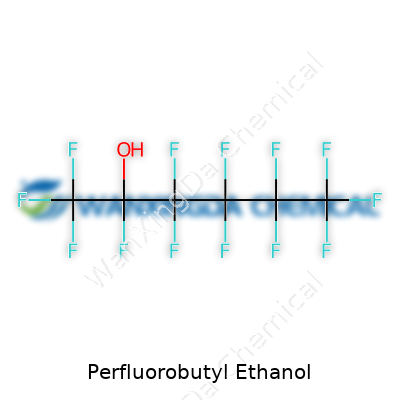
Perfluorobutyl Ethanol: Beyond the Lab Bench
Tracing the Origins: A Chemical Decades in the Making
In the world of specialty chemicals, perfluorobutyl ethanol stands out as a product of both necessity and ingenuity. The roots trace back to the post-war era, when chemists started to play with perfluorinated compounds, dreaming of their use in non-stick pans and high-performance coatings. As technology crept into nearly every part of day-to-day life, the need for unique solvents and specialized additives only grew. Perfluorobutyl ethanol entered the picture during this time, its backbone built for environments where regular molecules just couldn't keep up with the challenges of wear, solvent exposure, or extremes in temperature. My time working alongside polymer chemists showed me this molecule often caused people to raise an eyebrow—a chemical both clear and mysterious, with a set of properties not easily matched.
Getting to Know Perfluorobutyl Ethanol
It’s no secret that perfluorobutyl ethanol is a curious blend of stability and reactivity. The structure—perfluorobutyl group attached to an ethanol moiety—grants it a resilience rare in simple alcohols. Compared to familiar solvents, it resists attack from acids, bases, and oxidizing agents, and keeps its cool under intense heat. These traits come at a price; synthesizing such a compound calls for expertise in fluorination, often in settings few can afford to run safely or environmentally. In my own research, I found that handling these materials involves respect for both their power and their potential downsides, especially when compared to garden-variety organic compounds.
Chemistry That Demands Respect
With a boiling point far higher than many hydrocarbon peers and a density surprisingly high for a liquid, perfluorobutyl ethanol doesn’t behave like water or ethanol. Its low surface tension lets it sneak into tiny cracks and crevices, which earned it a role in the electronics world. It's colorless and nearly odorless, with a chemical stubbornness that makes it tough to break down. In the lab, we measured its solvency and found out pretty quickly it could dissolve fluorinated resins when almost nothing else would budge them. Because of the strength of those fluorine-carbon bonds, attempts to tweak this compound’s structure need more than conventional heating or basic reagents—a real hurdle even for seasoned synthetic chemists.
Preparation: A Balancing Act of Innovation and Precaution
Turning raw fluorocarbons into perfluorobutyl ethanol doesn’t come easy. Industrial processes rely on electrochemical fluorination or specialized halogen exchange reactions. In a pilot plant I visited, technicians worked with care, always double-checking their protective gear. Hiring a chemist isn't enough—one needs an entire safety mindset. These reactions produce heat, corrosive byproducts, and require customized materials for containment. From all those headaches and protocols, out comes a high-value liquid demanded by producers of technical coatings, lubricants, and fuel cell membranes.
Nomenclature and Industry Jargon
Depending on which technical paper you pick up or which catalog you browse, this compound answers to several names. You might see "1H,1H,2H,2H-Perfluorobutanol," "perfluorobutoxyethanol," or simple acronyms like PFBEO. Each name reflects a slightly different context—organic synthesis, surface chemistry, or regulatory labeling—reminding those who work with it that chemical language never stays entirely fixed. In working with international teams, I noticed confusion can stall a project for days if the label on the bottle doesn't match the terminology in the method sheet.
Safety: No Shortcuts Allowed
Safety isn’t just a corporate line item here. Perfluorobutyl ethanol means business in both the lab and plant. There’s been a growing push for tight occupational guidelines, especially after reports of acute exposure risks for many perfluorochemicals. Accident reports from the last decade show why gloves and goggles do more than keep up appearances—skin contact can cause irritation, and nobody wants to find out how tough it is to scrub off. Working environments should rely on robust exhaust ventilation, containment systems, and clearly marked waste disposal. Keeping up with both federal and local regulations has turned into its own full-time job, given how fast both research and public policy are changing in response to the ongoing PFAS scrutiny.
Where It Ends Up: From Circuit Boards to High-Tech Textiles
The market for perfluorobutyl ethanol stretches out in ways casual observers often miss. Electronics manufacturers count on it to clean circuit boards or as a surface treatment for high-voltage components, where even a speck of dust or a droplet could mean the death of a device. Textile engineers use it in finishing processes for water-repellent gear—the kind hikers and first responders rely on. Paint formulators and coatings experts slot it into niche applications where no other molecule stands up to the weather, chemical attack, or repeated wash cycles. For a world that's plugged in 24/7, the utility runs deeper than first glance might suggest.
R&D and the Scramble Toward Safer Substitutes
Long gone are the days when the latest fluorochemical could launch without questions. Environmental persistence and toxicity concerns fueled an entire branch of research dedicated to understanding what these materials do in water, soil, and human bodies. New regulations have forced chemists back to the drawing board for alternatives and improvements. In my own lab days, we tested dozens of possible replacements, but few matched the performance. Universities and private firms now fund studies examining breakdown pathways, adsorption in the body, and ways to improve biodegradability. The scientific community hasn't found a perfect substitute, but there’s an energy in the research that's tough to match elsewhere.
Charting the Future Amid a Shifting Regulatory Landscape
Public debate over perfluorinated chemicals shapes the future of perfluorobutyl ethanol. Activism and evolving science call out the risks—long half-lives in the environment, biomagnification through food chains, detectable traces in drinking water. Industry faces pressure to innovate safer processes, recycle waste, and develop greener analogues without sacrificing function. Where some see this as an existential threat, others see opportunity—a chance to reimagine chemical design, manufacturing, and stewardship. My own conversations with colleagues show both anxiety and hope. If regulators, researchers, and corporations find common ground, tomorrow's perfluorinated compounds might deliver on promise without leaving as much of a footprint.
Most people have never heard of perfluorobutyl ethanol, and to be honest, a few years back, I hadn’t either. But in the circles of chemical manufacturers and industrial engineers, this substance draws a lot of attention. It pops up in places you wouldn’t expect—electronics factories, medical equipment suppliers, even in the labs working on high-tech coatings. I remember the first time I ran into it during a conversation with a coatings specialist. He called it a “workhorse” for modern surfaces that take a real beating.
How It’s Used
Perfluorobutyl ethanol finds its way into compounds that need serious staying power: stain-resistant coatings on carpets, treatments for textiles that hold up against spills, and finishes on smartphones or tablets where fingerprints create havoc. If you’ve ever dropped coffee on your sofa and watched it bead up, there’s a good chance some fluorinated alcohol—maybe even perfluorobutyl ethanol—played a part. Companies blend it into liquid solutions that dry into barrier films, making water and oil roll away.
This chemical isn’t just about keeping things tidy. In the world of electronics, reliable insulation means the difference between a gadget that fries its circuits and one that runs smooth for years. Makers of semiconductors and sensors often turn to highly fluorinated chemicals to get that nonstick, non-reactive layer. Durability is crucial because today’s devices are thinner, lighter, and expected to survive a drop on the asphalt. One forgotten detail, like poor moisture protection, can bring an entire production line to a halt.
Big Benefits, Not Without Risk
Surface protection and chemical resistance don’t come for free, though. Perfluorinated compounds—perfluorobutyl ethanol included—bring some baggage. News stories keep highlighting potential toxicity and persistence in the environment. Scientists call these substances “forever chemicals” because they don’t break down, instead sticking around for decades in air, water, and soil. Evidence keeps piling up that some of them build up in the body, with links to health problems in animals, and concerns for people too.
My own experience interviewing public health experts taught me that breaking the cycle of chemical exposure isn’t simple. Regulations change as science evolves. Europe, for one, puts tight limits on specific perfluorinated compounds, and American agencies keep updating their warnings as new data arrive. In the factories I’ve visited, safety protocols now include full-body suits and meticulous monitoring of emissions and effluent.
Where Do We Go from Here?
The world won’t stop demanding stain-resistant fabrics or wafer-thin electronics. Still, there’s a steady march toward safer chemistry. Some research teams have started hunting for less persistent alternatives—chemistry that gets the job done but doesn’t hang around forever in rivers and tap water. Until those options reach the market, manufacturers pretty much walk a tightrope: balancing innovation, safety, and responsibility to the environment.
Understanding what’s inside our devices and finishes matters. When companies, regulators, and the public get behind safer technology, we all benefit in the long run. Honest conversations drive change more than any alarmist headline. Perfluorobutyl ethanol remains woven into the story of modern industry, and that story grows more urgent as new solutions take shape.
Everyday Hands and Unfamiliar Chemicals
Not every bottle in a lab or workshop sparks concern, but sometimes I’ve paused before picking up a new chemical, especially if the name sounds more at home in a tongue-twister than in daily life. Perfluorobutyl Ethanol sounds technical, but at its core, it’s a fluorinated alcohol. Over the years, I’ve learned that “fluorinated” doesn’t equal “harmless”—particularly when handling substances that have generated headlines for environmental or health worries.
What Perfluorobutyl Ethanol Does in the Real World
This chemical crops up most often in specialty settings. Coatings, surface treatments, electronics—places where durability or unique chemical properties are prized. Because of its structure, this compound resists water, oil, and stains, which has led operators and material scientists to value it. Yet, from my own time working with surface treatments, I’ve learned that the story rarely stops at performance.
Health and Exposure Risks
One thing scientists and regulators have agreed on: many perfluorinated compounds, particularly those like PFOS and PFOA, can stick around in the body or in the environment for years. While Perfluorobutyl Ethanol isn’t as deeply studied as some of its relatives, studies have raised questions about possible health consequences if it enters the bloodstream. I remember chatting with an environmental scientist who said, “If something resists breaking down in nature, it’s wise to ask what it might do inside us.”
Accidental skin contact or inhalation could be easy to overlook in a rush, but these are real paths for exposure. Even low-level exposure over time can add up. Those working in labs or industrial settings—where the chemical is more likely found—know that gloves, goggles, and decent ventilation matter. At home, such chemicals almost never show up, but people who handle fluorinated compounds can bring invisible traces with them.
The Bigger Environmental Picture
Consider the attention given to PFAS, a broad group that contains Perfluorobutyl Ethanol. These “forever chemicals” have been found in soil, water, and even rainfall. The long-term effects of accumulation remain under review, but the push for more information comes from concern, not unfounded anxiety. I’ve seen communities demand answers after PFAS were found in drinking water, worried about links to cancer and other illnesses. Though definitive proof for every PFAS chemical doesn’t always surface right away, the pattern calls for respect and caution.
Safer Habits for Healthy Outcomes
At its core, chemical safety comes from common sense and good habit. I think back to safety trainings: nobody wants a reminder that a simple mistake can have big consequences. Protective gloves, eye covering, and a fume hood feel like second nature with good habits. Never eating, drinking, or touching your face in a workspace—these habits stick with you because small exposures can add up unnoticed.
Disposal matters too. Pouring leftovers down a drain or tossing material in with household trash ignores the journey chemicals take through our shared environment. Treatment facilities struggle to break down fluorinated chemicals. Secure hazardous waste disposal keeps exposure lower for everyone down the line.
Moving Forward with Caution, Not Fear
Perfluorobutyl Ethanol deserves care in handling, especially since much remains to be learned about its long-term impact. Until research clarifies the risks or regulators set stricter guidelines, it makes sense to treat this compound with the same respect given to other persistent chemicals. Health and safety never go out of style, and the cost of a few extra precautions weighs less than the risk ignored.
Looking Closer at the Risks
Working with chemicals in a lab or industry always means paying attention to how they’re stored. Perfluorobutyl Ethanol isn’t something to treat lightly. It typically serves as an industrial solvent and comes with a mix of hazards due to its composition—perfluorinated substances have a reputation for environmental persistence and health risks. Many folks know about PFAS or “forever chemicals.” This ethanol is another extension of that concern. Poor storage won’t just mess up an experiment; it can put workers and the environment at risk, thanks to risks like toxic releases or spills.
Keeping Containers Tight and Materials Right
One lesson I learned early in the lab: never trust a cracked cap or the wrong bottle. Perfluorobutyl Ethanol needs sealed containers made from plastics or metals that resist strong solvents. Glass or cheap plastics sometimes seem like they’d work, but over time, aggressive chemicals can degrade the material. Leaks rarely happen at the convenient time, and the mess is rarely small. I always double-check the labels and gasket seals before putting anything like this on a shelf.
Avoiding Heat, Flames, and Sunlight
Most perfluorinated solvents have pretty high boiling points and don’t care for sunlight or open flames. With Perfluorobutyl Ethanol, that goes double. I’ve seen solvent containers sweat under heat lamps and crack in direct sun—nobody wants to walk in and find a puddle on the floor or, worse, vapors that could catch fire. Shelving in cool, well-ventilated spaces works best. Chemical storage rooms shouldn’t double as break lounges or storage for electronics. Sprinklers above chemicals don’t mix either. Solvents and water can react unpredictably.
Labeling and Segregation Matters
There’s a reason why hazard labels and chemical inventory systems exist. Mislabeling or mixing incompatible chemicals created real headaches in a few labs I worked in. Keeping Perfluorobutyl Ethanol away from acids, bases or strong oxidizers prevents nasty reactions. Even trace contamination in a shared secondary container can ruin expensive chemicals or worse, hurt staff. Segregation by risk—flammable, toxic, corrosive—lowers the chance for drama.
Ventilation and Environmental Concerns
Tight storage is one defense, but no cabinet seals everything off entirely. Volatile chemicals leach small amounts of vapor. Properly ventilated storage rooms make a big difference. Companies and universities that cut corners here end up explaining mysterious illness reports or equipment corrosion far too often. Any spills or leaks must stay contained, with secondary containment trays catching leaks. Environmental protection gets trickier with perfluorinated compounds since small amounts last in soil and water for decades. I’ve followed up on groundwater studies ruined by improper disposal or storage that let PFAS products leaks into the sample sites.
Solutions Start With Training
Human error tops the list of causes for storage failures. People get busy, skip steps, or forget to replace old seals. I’ve worked in both tightly run and lax labs, and the difference in spill incidents is night and day. Sticking to training schedules, enforcing storage inspections, and rewarding safe practices earns better results than any written policy.
Clear Accountability for a Cleaner Future
Tighter regulations keep popping up for perfluorinated chemicals due to their persistence and toxicity, and that’s not going away soon. But rules only set the minimum line. The best results come from blending solid chemical knowledge, practical habits and a healthy respect for what goes wrong when things get sloppy. This mix isn’t just bureaucracy; it’s basic safety—and it keeps labs and the wider environment safer, too.
Most people never hear about chemicals like perfluorobutyl ethanol. That doesn’t mean their homes, offices, or even clothing are free of their touch. This four-carbon alcohol, trailing a tail of fluorine atoms, slips quietly into firefighting foams, specialty coatings, and even stain-resistant fabrics. It pulls off tricks that ordinary alcohols just cannot, thanks to its unique chemical makeup.
The Backbone: Strong Carbon-Fluorine Bonds
Chemists and toxicologists often discuss the carbon-fluorine bond in reverent tones. It’s one of the sturdiest covalent bonds found anywhere. Perfluorobutyl ethanol uses a string of carbon atoms, each clinging to several fluorine atoms. This structure shrugs off water, oil, heat, and even harsh acids and bases. The ethanol group at one end gives just enough “personality” to make the molecule interact with both water and oil to some degree, which proves essential for its applications.
Why Resistance Means Persistence
Stories in the news highlight just how persistent fluorinated chemicals are. Perfluorobutyl ethanol does not break down easily in the environment. That carbon-fluorine shell shields it against microbial attack, sunlight, and even high temperatures. We’ve seen the impact this can have on water and soil quality where manufacturing takes place. Traces of these substances stick around for decades, snaking through food chains and sometimes ending up in people and wildlife.
Performance and Trade-Offs
As an alcohol, perfluorobutyl ethanol brings some flexibility that straight-chain perfluorocarbons can’t provide. The alcohol group at one end lets chemists hook the molecule onto surfaces or into fluid mixtures. This tweak lets stain-resistant sprays cling to carpets or gives waterproof clothing its magic. It reminds me of how seemingly tiny changes—adding a simple group—shift a molecule’s role dramatically.
But this performance comes with a price. Research keeps scrambling to catch up with how these compounds move through the body and the world. Studies published in journals like Environmental Science & Technology have found that even short-chain variants aren’t as innocent as hoped. The longer these chemicals stay in use, the clearer their health and ecological risks appear.
What Real Solutions Might Look Like
Moving forward takes more than simply swapping out molecules for slightly less persistent versions. Regulatory agencies in North America and Europe have started exploring bans and restrictions. Several companies have begun chasing “fluorine-free” chemistries for common uses, including cookware and floor treatments. There’s still a gap between what customers expect and what safer alternatives can actually do.
My experience working in a lab taught me every choice in the supply chain ripples out farther than expected. Solving the challenges of perfluorobutyl ethanol means accepting short-term inconvenience for long-term relief. Clear labeling, public education, and genuine investment in green chemistry offer real hope for moving the needle.
It’s easy to overlook invisible advantages and risks sitting inside a Teflon pan or a spill-proof sofa. Understanding the quirks of these molecules lets us remake the world in healthier, more responsible ways—if we decide the comfort is worth the long-term cost.
Shopping for something as specialized as Perfluorobutyl Ethanol isn’t much like picking up office supplies or ordering groceries online. This chemical rarely pops up in ordinary conversation, but for experts in certain industries, it’s an essential material. The chase for a pure, legitimate source comes with its own set of hurdles—ones that highlight the gap between general retail and scientific procurement.
Why Sourcing Matters
Years ago, working in a university lab, a colleague and I tried to track down a rare reagent. The process showed me just how cautious buyers must be with chemicals that aren’t widely distributed. Getting something as niche as Perfluorobutyl Ethanol from a random supplier can spell trouble, both for the integrity of your work and for your safety. Many folks outside the chemistry and materials world may not realize the checks in place for these substances. Orders often require institutional clearance, background on intended use, and sometimes proof of laboratory credentials. All of this helps prevent dangerous misuse, as well as accidental purchase by those not prepared to handle volatile or toxic agents.
Purity and Credibility: Two Sides of the Same Coin
People sometimes ask, “Can’t you just shop online and pick the cheapest supplier?” The reality is more complicated. For both research and industrial uses, purity isn’t negotiable. Even a small amount of unwanted byproducts or impurities can dismantle a series of experiments or a production batch. In my experience, the difference between published results and confusing failures often boiled down to overlooked contaminants from a shoddy source. Trusted chemical suppliers usually provide documentation—a certificate of analysis, batch traceability, and verification of handling procedures. Well-known names in lab supply, with established reputations, tend to dominate the market for good reason.
The Online Maze
You won’t see Perfluorobutyl Ethanol in general e-commerce markets. Most reputable suppliers operate their own secure sites, which restrict access or require business account verification. Major scientific suppliers often ask for end-user statements, research justification, and other paperwork to guard the chain of custody. This practice doesn’t just protect their reputation—strict oversight ensures buyers know what they’re doing, have the right storage and disposal plans, and understand the legal environment around specialty chemicals. Genuine sellers aim for transparency: providing chemical safety information, clear delivery terms, and options for technical support. Fly-by-night resellers, meanwhile, skirt around these practices and introduce real risk into the equation.
Regulation and Responsibility
Regulators keep close watch over the distribution of select chemicals for good reason. Incautious handling and unregulated sales feed into environmental pollution, workplace accidents, and even criminal misuse. Genuine suppliers stay aligned with these laws to avoid regulatory blowback and to help buyers meet their own obligations. Reliable online platforms will take time to confirm buyer credentials and will stop short of handing over specialty chemicals to unknown parties. This kind of diligence also offers peace of mind: you’re much more likely to receive what you actually ordered, without worrying about mislabeled substances or dodgy documentation.
Improving Access Without Sacrificing Safety
Specialty chemical access remains a challenge for independent researchers and smaller organizations. Many have highlighted the need for clearer pathways that balance accessibility with oversight. Connecting with university purchasing offices, forming consortia, or using recognized procurement partners can smooth the process. On the flip side, the industry would benefit from better guidance and more transparent communication about the rules of entry and handling. Responsible procurement helps protect both the buyer and the broader community. Navigating the process is less about finding a shortcut and more about understanding why the checkpoints exist. In today’s world, that’s an investment in safe progress.