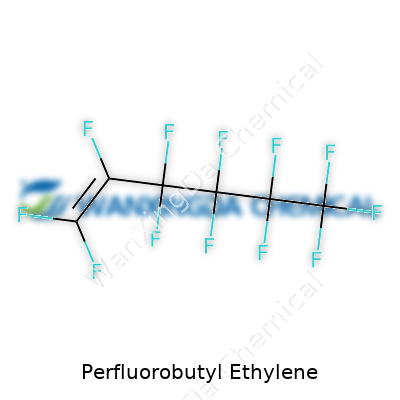
The Many Layers of Perfluorobutyl Ethylene: From Laboratory Curiosity to Industrial Mainstay
Historical Development
Chemistry goes through phases—something starts as a scientific oddity, then slowly finds its place in industry. Perfluorobutyl ethylene has followed this well-worn path. Decades ago, labs experimented with perfluorinated compounds, chasing solutions for chemical resistance in harsh settings. Back then, academics faced expensive reagents and awkward purification steps, so curiosity-driven research delivered new perfluoroolefins only in small flasks. Through the late 20th century, as fluoropolymer use exploded in electronics, semiconductors, and healthcare, more eyes turned toward ethylene-based perfluorinated molecules. Access shifted from the preserve of researchers to pilot-scale projects. By the 2000s, scaling up led to smoother syntheses, new applications in advanced coatings and barrier materials, and a growing understanding of both the benefits and the limits.
Product Overview
Perfluorobutyl ethylene belongs to the wider perfluoroalkylethylene family. Structures with nothing but carbon-fluorine bonds stick out: they offer low surface energy, strong resistance to acids and bases, and real stability under high temperatures. In daily work, chemists and engineers turn to this material when a process throws up challenges that break down conventional carbon chains. Back in a teaching lab, most people see fluorinated chemistry as difficult due to the stubbornness of those C–F bonds. In industry, though, this stubbornness spells fewer failures from oxidation or corrosion. Products show up mainly in the form of colorless liquids, often faintly sweet-smelling, shipped in amber bottles to protect against light.
Physical & Chemical Properties
Looking across data sheets and publications, some trends emerge. Perfluorobutyl ethylene holds a high density compared to hydrocarbons of similar size, reflecting all that fluorine crammed around the carbon backbone. The boiling point ranks well above non-fluorinated analogs. Solubility splits the crowd: water barely touches it, but many fluorinated solvents dissolve it happily—great for those working with specialty surface treatments or preparing microfluidic devices where water’s presence might wreak havoc. The molecule shrugs off acids, alkalis, oxidizers, and most reducing agents. Handling calls for gloves, not because of immediate hazard but to prevent wrecking the compound’s purity. Chemicals like this don’t burn easily. Put a match to it, and the stuff resists. Yet, incomplete combustion can lead to toxic byproducts, making lab managers hyper-cautious about disposal and air filtration.
Technical Specifications & Labeling
Every facility demands solid labeling, especially with new substances. Bottles in research supply cabinets bear systematic names, warning symbols, and concentration details. These aren’t for show; in complex fabrication environments, a stray solvent or cross-contamination ruins batches and bruises profit lines. Regulatory authorities like OSHA, REACH, and others push strict standards on permissible exposure and storage. Those who write SDS sheets for perfluorobutyl ethylene pore over the best available toxicity data and incident reports—something any conscientious chemist checks before first use. Some companies apply tamper-evident seals or track shipment histories, reassuring downstream buyers about supply-chain reliability.
Preparation Method
Synthesis started out as an involved process, usually involving telomerization reactions. This looks neat on paper—throw a perfluorobutyl group onto ethylene, then refine. In practice, manufacturing scale brings headaches like catalyst loss or separation challenges. Watching pilot reactors, engineers fuss over parameters: temperature, reactant ratios, and solvent choice. Small changes in timing or atmosphere spell differences in yield or purity. Recovery needs distillation or extraction, often under vacuum, with an eye toward avoiding fluoride emissions that foul air scrubbers. While recipes exist, each factory tailors steps based on equipment and local rules. What once stretched out over days can now often be done in continuous-flow reactors, boosting consistency and safety.
Chemical Reactions & Modifications
Perfluorobutyl ethylene claims a special spot in synthetic chemistry—almost untouchable by most common reagents but open to grafting and functionalization when pushed with the right tools. Researchers have explored cross-coupling reactions, adding bits to the ethylene double bond, or even tweaking the tail with photochemical tricks. Scattered academic papers chart efforts to fix functional groups—amines, alcohols, or acids—onto the backbone. These hybrids end up in novel surfactants, membrane anchors, and sensors. Some work focuses on making block copolymers for special coatings or biomedical uses, chasing combinations that tune water resistance, adhesion, or biocompatibility. Chemists interested in smart materials see this as a route toward custom self-assembled structures.
Synonyms & Product Names
Trade names and alternative labels muddy the waters, especially internationally. The academic community sticks to IUPAC, so you get terms like 1,1,2,2,3,3,4,4-Octafluorobut-1-ene. Suppliers, wanting to stand out, coin their own branding—sometimes a string of numbers, sometimes shorthand reflecting the structure’s backbone. Old texts mention it as perfluorobutene ethylene, leading to confusion for anyone combing the literature without synonym cross-checks. For import, customs records call for careful matching, since ambiguous entries can trigger delays or checks for regulated chemicals.
Safety & Operational Standards
Working with perfluorobutyl ethylene, most safety measures center on inhalation risk and environmental escape. Over long shifts, fumes from open containers stack up, but strong exhaust hoods and closed systems keep levels well below established limits. While acute toxicity seldom rivals that of more notorious industrial substances, the long-term story still writes itself—precaution beats cure every time. Waste management pushes up against regulatory walls. Landfill or incineration means contending with perfluorinated degradation products, sometimes classified as persistent organic pollutants. Labs juggling multiple perfluorinated materials put environmental protection front and center, installing carbon filters, tracking disposal flows, and training new staff on spill response. Glove choice and personal monitoring plans go beyond box-ticking, reflecting a growing sense that health and environmental costs outlast any quarter’s production.
Application Area
Walking across sectors, you meet perfluorobutyl ethylene in unlikely places. Electronics manufacturers coat fragile sensor chips, keeping out water and oxygen where even tiny leaks cost millions. Coating lines in packaging plants lay down ultra-thin films, aiming for food safety and shelf-stable flavor profiles. Engineers working on lab-on-a-chip technology love these materials for making channels that nothing sticks to—blood, oil, or aggressive reagents. Specialty lubricants in aerospace or automotive engines draw on perfluorinated chains for wear resistance where other oils simply cook off. Plenty of researchers play matchmaker between biomedical needs and the unique traits these chemicals offer, hunting for ways to make drug-carrying vesicles, implant coatings, or diagnostic devices just a touch more stable or resistant to fouling. Demand tracks advances in industries rather than volume—these chemicals rarely become bulk commodities, but their high value means that a barrel sells for more than a truckload of common feedstocks.
Research & Development
The research scene runs the gamut—from exploring better synthetic routes to fine-tuning functionalization chemistry. University groups design clever catalysts to shrink energy use, or to coax higher selectivity from familiar reactions. Instrument manufacturers watch to see which modifications open new windows for sensors or analytical membranes. Some teams chase greener chemistry angles: halving waste, replacing legacy solvents, testing new solid-state reaction setups that slice energy costs. In the corporate world, R&D focuses on scaling up innovations safely and at consistent quality, often under sharp regulatory scrutiny. Good science here delivers shelf-stable and reliable products for engineers who might never see the molecule itself, but who notice the difference when it performs as promised in the field.
Toxicity Research
The health and environmental footprint of perfluorobutyl ethylene sparks concern as much as curiosity these days. Centuries-old ideas about chemical safety grow thinner with complex, persistent molecules like this one. Animal studies looked at chronic exposure and found perfluorinated compounds stick around in blood and tissues, though each family member varies in how long and in what organs. Knowledge gaps remain, and few clear answers count as settled. Regulatory agencies advocate for cautious use, especially in settings with potential for release to air or waterways. Recent toxicological screens probe for subtle effects: endocrine disruption, immunosuppression, or developmental impacts. The literature teaches humility; even decades of review haven’t chased down every pathway. Users never outgrow the need to check for new data or to plan disposal as if every drop matters.
Future Prospects
Looking ahead, perfluorobutyl ethylene faces both opportunity and scrutiny. On the one hand, specialty coatings, advanced composites, and miniaturized electronics drive demand for chemical robustness and tailored interfaces—here, perfluorinated building blocks shine bright. Yet, environmental watchdogs call for new stewardship standards. The future likely involves tighter controls, better life-cycle tracking, and design-for-disposal built in from the start. Calls grow louder for biodegradable or reversible alternatives. Some hope springs from work in enzyme-catalyzed defluorination, where microbes tackle bonds once thought immortal. Chemical designers bear a burden: how to balance unique performance with public and planetary health. Each step forward needs a clear-eyed review of safety data and creative efforts to reclaim or recycle what’s already made. If the last fifty years taught anything, it’s that scientific promise and practical responsibility must walk in step.
Invisible Shield for Electronics
If you’ve ever had a smartphone that survived spilled coffee or a laptop that made it through a rainstorm, chances are, a chemical like perfluorobutyl ethylene had something to do with it. Modern electronics face dust, moisture, and temperature shifts. Manufacturers use perfluorobutyl ethylene to build moisture barriers inside the devices. Unlike older methods that added bulk or weight, coatings with this fluorinated compound create thin, nearly invisible protective layers, helping circuit boards last longer and work harder in rough conditions. Studies show electronics treated with fluorinated films report fewer failures due to corrosion or water damage.
Surgical Precision in Medicine
Doctors and medical tech developers count on reliability, especially with critical components like sensors inside heart monitors and drug delivery systems. Perfluorobutyl ethylene doesn’t stick easily to other molecules, so companies use it to keep medical sensors clean and free of buildup. A friend working in medical device manufacturing once told me about the headaches from organic buildup on sensors, and how these durable coatings reduced cleaning cycles and downtime. Hospitals demand this kind of consistency since even minor contamination risks patient safety.
Chemical Wonder in Coatings and Films
Paint companies and coating producers regularly experiment with additives to boost stain resistance or water repellence. Years ago, I saw an industrial warehouse floor treated with a fluoropolymer film. The difference was clear—a quick mop-up kept the place spotless, while untreated sections stained and wore out faster. Perfluorobutyl ethylene ends up in these specialty films and coatings, from high-end automotive paint to advanced anti-graffiti solutions for public structures. This chemical takes a beating from chemicals and weather, often outlasting traditional coatings.
Chasing Sustainability
Chemists once leaned on longer-chain perfluoroalkyl compounds for similar results, but the spotlight quickly shifted. Shorter chains, like those in perfluorobutyl ethylene, break down quicker in the environment. Environmentalists and regulators call out legacy substances for lingering in water and soil—what scientists label as “forever chemicals.” Shorter molecules offer a potential compromise, with less risk of accumulation and fewer known impacts on wildlife or human health. Regulatory changes now steer many companies toward safer alternatives, so I expect more research into shorter-chain fluorinated compounds, including better recycling and disposal methods.
Building Better Batteries
Storing more power in smaller, lighter batteries drives everything from electric cars to backup power grids. Battery developers look for parts that handle heat and chemical stress. Manufacturers use perfluorobutyl ethylene to strengthen separators—the thin membranes between a battery’s anode and cathode—since these need both flexibility and resistance to breakdown. Fires and leaks in batteries usually make headlines, so improvements in chemical stability grab the industry’s attention. Every incremental gain in reliability opens doors for safer, longer-lasting energy storage.
Looking Ahead
With research pushing the limits in technology, health, and sustainability, perfluorobutyl ethylene seems poised to secure a bigger role. Companies chasing safer, tougher, and longer-lasting materials look for chemicals that combine performance with reduced environmental impact. While this molecule won’t grab headlines, the way it quietly shapes new products deserves some attention. The conversation about chemical safety is just beginning, and I hope for even smarter uses backed by real transparency—because we all live with the choices made in manufacturing.
Getting Real About Chemical Safety
Perfluorobutyl ethylene looks just like another routine chemical, but behind the unassuming name sits an industrial workhorse found in manufacturing and electronics. Ask any chemist, and you’ll probably hear the same thing I learned working shoulder-to-shoulder in a lab: familiarity breeds carelessness, and that's when accidents happen. Wearing gloves isn’t a suggestion, it’s a necessity—one spill on the skin calls for a rinse that nobody enjoys. Simple nitrile gloves get the job done, and my own hands thanked me countless times for the small act of putting them on before starting work.
The Case for Basic Protective Gear
Goggles and lab coats shouldn’t feel like overkill. Protective eyewear matters because splashes don’t ask for permission. I remember a hurried afternoon pour going sideways. The goggles caught every drop. Even if the liquid looks clear and harmless, perfluorobutyl ethylene can irritate the eyes and cause trouble fast. Long sleeves and fully buttoned lab coats keep splashes from skin and street clothes. That bit of fabric often stands between you and a surprise rash.
Well-Ventilated Spaces: The Unsung Hero
Some chemicals stink up a room fast, but perfluorobutyl ethylene won’t always signal a problem with a strong odor. Every competent workspace deserves local exhaust systems or fume hoods, since breathing low-level vapors for hours does no favors to anyone’s lungs. Standard building ventilation rarely cuts it in the long run. Flipping on the fan or kicking open a window isn’t enough—if you work with this stuff regularly, invest in true fume extraction.
Smart Storage Prevents Headaches
Every bottle deserves a clear label and a sealed cap. Leaving chemicals in a sunny corner leads to a mess, as light and heat speed up the breaking-down process. I once saw an unlabeled bottle mix with moisture near an open window and ruin an expensive batch. Store perfluorobutyl ethylene in a cool, dry cabinet, separate from acids or strong bases. Don’t let curiosity or convenience cut corners. Bad storage habits only cause big trouble later.
Respect for Disposal Rules
Dumping chemicals down a drain is a shortcut straight to environmental problems. Fluorinated solvents linger in water supplies and resist breaking down, which means even small spills matter in the long run. Every workplace must set up proper waste containers. I learned that local hazardous waste disposal services give clear directions and usually pick up small-batch chemical waste regularly rather than leaving you to guess. Following their lead keeps toxic contaminants out of the ecosystem.
Training Makes All the Difference
No one starts out as an expert, and those with experience have a responsibility to teach the basics to newcomers. The best labs I’ve joined set aside time every month to review protocols, update spill kits, and remind everyone how to use a chemical safety shower. Hands-on training sticks better than a printed sign. Run drills, keep emergency gear in sight, and ask questions until the process feels like breathing. Safety sticks when everyone invests in each other's well-being.
Spending a few extra seconds on gloves, goggles, proper storage, and good ventilation never feels like wasted time with chemicals that stick around in the environment. The foundations of safety never go out of style—especially when one small misstep makes all the difference.
Before diving into technicalities, let’s be clear—chemicals like perfluorobutyl ethylene aren’t household names, and their properties shape more industries and products than most folks realize. Anyone who has dealt with fluorinated chemicals knows they come with a unique blend of strengths and stubborn challenges.
Why Its Structure Matters
Perfluorobutyl ethylene stands apart thanks to its arrangement of carbon atoms fully surrounded by fluorine atoms, with an ethylene group tagging along. Those carbon-fluorine bonds don’t let go easily. In my experience, working with these types of molecules means dealing with chemicals that laugh off the kinds of acids and bases that chew through lesser compounds. This kind of chemical backbone doesn’t just resist the odd spill—it shrugs off most attempts to break it down.
Anyone in the chemical industry knows: durability can be a blessing and a curse. The strength of those bonds keeps perfluorobutyl ethylene from reacting with most things. That’s part of the reason folks use it in environments where corrosion eats up regular plastics or rubbers. Old pipes, worn valves, failing gaskets — substituting with robust fluorinated polymers knocks those problems back for decades.
Physical Qualities That Set It Apart
Looking at its physical make-up, perfluorobutyl ethylene comes over as clear and largely odorless. Its boiling and melting points run higher than most familiar organic solvents. During my time handling samples in the lab, that meant ventilation worries dropped down, but you always keep an eye on containment, since volatility isn’t the only risk factor. High density compared to hydrocarbons is easy to spot in the lab: a drop of it almost feels heavier between glass slides.
Non-flammability often crops up as a touted feature, and rightly so. In a world full of fire risks, having a chemical that simply won’t ignite under everyday conditions brings some peace of mind, especially in manufacturing spaces packed with electrical equipment. Health and safety teams appreciate anything that cuts down on potential flash points.
Chemical Inertia—Both Strength and Challenge
This chemical’s main claim to fame is how little it interacts with the world around it. I've seen situations where other materials fail, leaving only the perfluorinated components standing strong. That means fewer product recalls and longer equipment life, saving both money and headaches for many industries.
All that resistance, though, comes at an environmental price. Anyone following PFAS debates knows chemistries like perfluorobutyl ethylene often resist natural breakdown, spreading through soil and water and lingering a long time. The U.S. Environmental Protection Agency and European regulators have started paying serious attention, searching for ways to contain and remediate these chemicals. I’ve seen growing pressure on manufacturers to look for alternatives, and increasing investment in research that aims to turn the tide on persistence.
Finding Solutions
Moving forward, innovation must work in lockstep with regulation. My time working alongside environmental chemists taught me that real-world solutions rarely come by banning chemicals outright. Push for closed-loop systems wherever these chemicals get used. Encourage research into biodegradable fluorinated compounds. Support clear labeling for products, so people know exactly what they’re bringing home. And involve communities in the conversation—after all, the folks downstream live with the results.
Perfluorobutyl ethylene promises plenty with its stable, durable nature. If we harness those strengths wisely and address the fallout from chemical persistence, we can get the best of both worlds—engineering value and environmental respect, side by side.
Why Storage and Shelf Life Matter
Keeping chemicals in good condition matters to anyone working with them. Over my years helping teams in labs and manufacturing, I’ve seen what happens when storage gets sloppy—losses pile up, safety slips, and costs sneak higher. Perfluorobutyl ethylene doesn’t show up in most people’s pantries, but in specialty work, it stands out for its stability and low reactivity. Still, experience shows that ignoring storage guidelines eventually hits projects hard.
Physical Properties and Practical Challenges
Perfluorobutyl ethylene belongs to a class of fluorinated compounds valued for their chemical resistance. This makes people think they can store it anywhere without worry. I’ve had conversations with old colleagues who thought an extra bottle could just sit on any shelf, and nothing would change. Unfortunately, that attitude welcomes trouble. Even resilient chemicals lose quality if exposed to heat, light, or fluctuating room temperatures.
In real-world settings, I’ve watched how even short exposure to sunlight can raise temperatures inside containers. That leads to small changes over months. Unnoticed leaks, vapor buildup, or unexpected pressure shifts can stress packaging, especially if someone reused a bottle or skipped the original packaging. Easy mistakes snowball once someone ignores storage for the sake of convenience.
Best Practices in Storage
Perfluorobutyl ethylene stores best in tight glass or high-grade fluoropolymer containers away from direct sunlight. Cool, dry, well-ventilated storage spaces protect both the chemical and the team. Laboratories or shops slack off when rushed, but leaving a drum next to a heated pipe or an always-open window invites small, preventable accidents. I’ve seen labs turn chemicals unstable over a few seasons just from careless shelf choices.
Keeping accurate labels and dates helps everyone know how long each batch has sat in storage. In some places I’ve worked, a quick check of inventory and labeling saved us from using compromised material. It only takes a few minutes to mark things clearly—effort that pays off later.
Shelf Life and Stability
As a fluorinated olefin, perfluorobutyl ethylene resists many factors that break down less robust chemicals. Manufacturers who do publish shelf life usually estimate several years under tight storage conditions. Still, not every batch lasts the same. I’ve learned that temperature swings and poor handling chip away at longevity, so even stable chemicals benefit from regular checks. Responsible teams track opening dates and inspect for any signs of discoloration, cloudiness, or pressure changes.
Addressing Issues and Moving Forward
Every professional who handles specialty chemicals can raise standards with small changes—consistent labeling, routine inspections, clear protocols for sealing and storing. In my experience, one overlooked bottle causes far more trouble than steady, low-effort attention to detail. It’s worth talking openly about mistakes; a culture that skips blame and focuses on awareness keeps hazardous materials out of the wrong hands and on the shelf for as long as possible.
The chemical world stands on the shoulders of those who respect its risks. Vigilance doesn’t cost much, yet it makes chemistry safer and more reliable. A little extra care in storage and tracking shelf life guarantees both the safety of people and the reliability of project outcomes.
Looking Past the Chemistry
Perfluorobutyl ethylene isn’t the easiest name to read or pronounce, but the questions swirling around its environmental footprint are a lot harder to ignore. I came across this name digging into chemical ingredient lists, searching for answers about whether it’s a threat to the environment or tightly watched by lawmakers. Years back, these kinds of compounds were rarely discussed outside science labs. Now conversations about “forever chemicals” show up during neighborhood meetings and in frustrated tweets about water alerts.
What Sets It Apart
Perfluorobutyl ethylene fits into a bigger family known as PFAS — per- and polyfluoroalkyl substances. This whole group has landed in headlines for a reason: they stick around in water, soil, and even our bodies. Communities living downstream from factories or near airports often feel the brunt of these substances. Reports show PFAS compounds pop up in drinking water, fish, and wildlife. Old products—from food packaging to raincoats—released these chemicals over time, and now people want to know which specific ones keep showing up and causing trouble.
An Uneven Regulatory Map
Perfluorobutyl ethylene isn’t as notorious as PFOA or PFOS—the two PFAS members that received heavy scrutiny and eventual bans in some countries. Laws catch up slowly. Take the United States: the EPA only recently added some PFAS chemicals to drinking water advisories. States like Michigan, New York, and California moved faster, setting limits or banning certain types from child products and firefighting foam. In Europe, REACH regulations target broad PFAS groups, although the list of regulated substances still grows.
So far, there’s no blanket ban on perfluorobutyl ethylene. Each country makes its own choices. Most research and restrictions focus on PFAS that show up in large amounts or stick around the longest. Scientists are playing catch-up on the rest, including perfluorobutyl ethylene. Without specific laws, companies using this chemical face less pressure to stop or substitute. Regulatory gaps mean monitoring happens piecemeal and communities living near plants feel left behind.
The Stakes for Health and Nature
Last year, I read a story about a river near a chemical plant. People stopped swimming and fishing out of fear that PFAS levels would cause health problems. Evidence suggests members of the PFAS family can disrupt hormones, weaken immune systems, and build up over time. Wildlife takes the hit, too. Birds, mammals, and fish collect these pollutants through food and water. While we don’t have stacks of data on perfluorobutyl ethylene alone, its structure signals similar concerns—resistant to breaking down, stubborn in living tissue.
The lack of hard rules and full research isn’t reassurance. Plenty of chemicals in this class carried “safe until proven otherwise” labels, yet communities living with the pollution suffered first. With perfluorobutyl ethylene, it’s less about waiting for disaster and more about asking honest questions right now.
Solutions: Push for Evidence and Action
People want answers—and fast—when it comes to the water they drink and the safety of local wildlife. More research with real-world measurements can give us the proof needed to guide policy. Companies can take the lead and find alternatives, cutting down on unnecessary risks. Consumers searching for safer cleaning products, fabrics, and food packaging will encourage better practices across the supply chain. Pressure from neighbors, activists, and lawmakers gets the wheels moving in the right direction.
Environmental hazards don’t wait for paperwork to pile up. Perfluorobutyl ethylene deserves more attention, both in labs and lawmakers’ offices, because ignoring it could mean repeating the same mistakes of the past. Communities deserve clear information and stronger safeguards, not just another long chemical name to worry about.