Perfluorobutyl Sulfonyl Chloride: The Science, the Risks, and Paths Forward
Tracing the Roots of Perfluorobutyl Sulfonyl Chloride
Chemistry often feels like a race between new ideas and their real-world impact. Perfluorobutyl sulfonyl chloride walks a path marked by innovation, regulatory catch-up, and discovery. Born from research labs looking for new chemical tools, its history connects with the broader rise of fluorinated compounds in the late twentieth century. These molecules promised a kind of stability rarely found elsewhere—perfluorobutyl sulfonyl chloride stood out because its chemical backbone offered resistance to breakdown, a trait coveted by manufacturers. Over the years, it found its place at the intersection of industrial ambition and technical problem-solving, though with this rise came fresh scientific and regulatory attention.
What Defines the Compound
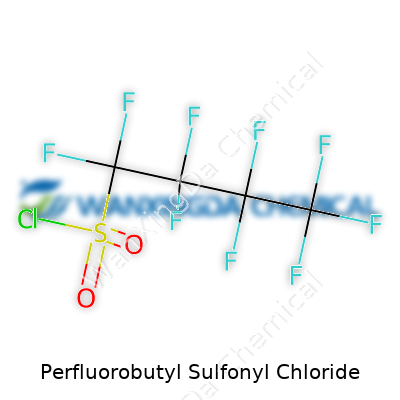
A quick scan through its molecular structure, you notice the hallmark arrangement: a perfluorinated carbon chain capped by a sulfonyl chloride group. That means the carbon backbone gets fully shielded by fluorine atoms, making it both a boon and bane. The fluorine cloak resists attack from almost anything—acids, bases, sunlight, even microbial life. Sulfonyl chloride, on the other hand, delivers the functionality labs often need for further modification. This is no simple building block; its strength and reactivity open doors in synthesis that few other options can match. It’s these chemical traits—robustness, reliability under strain, and reactivity when challenged—that make this compound important across high-performance applications, especially when manufacturers face aggressive chemical environments.
Technical Nuances and Labeling Realities
Labeling in industry requires honesty about both benefits and dangers. Here, regulations serve not only as boxes to tick, but as reminders of why clear information protects everyone up and down the supply chain. Perfluorobutyl sulfonyl chloride must be labeled with its hazardous nature front and center—corrosive, toxic, and persistent. There’s little margin for error in how these warnings get presented; oversight in labeling isn’t just regulatory trouble, it’s a safety disaster waiting to happen. Labels must carry the recognized synonyms, precise chemical formula, and supplier safety guidance. Consistent markings spare no detail, yet the stakes go beyond compliance—workers handling shipments, transporters moving drums, researchers planning synthesis steps all rely on accuracy. Sometimes in the rush of industry, paperwork feels an afterthought, but with a compound like this, clear documentation sets the tone for every safe interaction to come.
How It’s Made: The Manufacturing Approach
Manufacturers synthesize perfluorobutyl sulfonyl chloride through tailored routes—often by direct sulfonation of perfluorinated hydrocarbons, followed by chlorination of sulfonic acid precursors. The chemistry sits at a crossroad between brute industrial scale and delicate control. Working with highly reactive reagents, operators must keep tight grips on conditions. Temperature control, exclusion of water, and precise titration all prevent dangerous runaway reactions. Production generates hazardous byproducts, many of which share the persistent traits of the final molecule. Safe disposal and recycling take as much engineering effort as the climbing revenue charts. The underlying challenge comes from balancing technical ambition with environmental safeguards, pressing for both high purity outputs and stricter pollution limits.
Where Chemistry Meets Reality: Reactions and Modifications
This compound opens possibilities in synthesis that few others can match. Its sulfonyl chloride group attacks nucleophiles—amines and alcohols, mainly—efficiently forming sulfonamide and sulfonate linkages. These modified products find their way into pharmaceuticals, specialty surfactants, and advanced polymers. Because the perfluorinated part resists biological breakdown and most harsh reagents, materials based on these linkages tend to outcompete traditional options in severe conditions. The same resistance that boosts performance complicates waste treatment and environmental safety, setting up a dynamic where each chemical advance invites tough questions on lifecycle management. In my own lab days, handling such reagents carried a certain weight—you feel the risk in your gloves, taste the solvent vapor in the air, and learn the discipline that comes from knowing just how unforgiving these molecules can be outside controlled settings.
The Landscape of Names and Synonyms
Any discussion about a specialty chemical must acknowledge the confusion that blooms from varied naming conventions. Perfluorobutyl sulfonyl chloride answers to a list of aliases—sometimes just a tweak in the perfluorinated chain, sometimes a full rebranding for global supply chains. In the regulatory world, chemical abstracts numbers become the Rosetta Stone, tying together trade names, supplier codes, and synthesis routes. Researchers and industrial users quickly learn to map synonyms to molecular identity. That isn’t just an exercise in paperwork, it helps prevent deadly mix-ups in high-throughput environments. Keeping the naming landscape mapped and updated rewards diligence and can spare mistakes before they escalate.
Operating Standards and Practical Safety
Walking onto a floor where perfluorobutyl sulfonyl chloride gets processed, you hear the hiss of fume hoods and see full PPE as standard. The compound burns, corrodes, and toxifies with a vengeance if its boundaries are ignored. Industry standards do more than set thresholds for exposure—they enforce process redundancies, spill response plans, and training sessions that leave no gap in operational awareness. I respect those standards because cutting corners with robust halogenated reagents costs more in cleanup, regulatory risk, and human health than any time “saved.” The best shops pair strict barriers—segregated lines, sealed transfer systems—with drills that keep emergency skills sharp. Over time, muscle memory builds up so that safe habits feel second nature, but it never pays to grow complacent when dealing with molecules designed not to relent.
Application Frontiers and Functionality
Perfluorobutyl sulfonyl chloride built a track record in sectors where ordinary materials simply fail. You find it in the manufacturing of high-performance fluorosurfactants, specialty coatings resistant to chemical onslaught, and selective ion-transport membranes inside electrochemical devices. In advanced electronics, it helps fashion dielectrics and etching agents that define the latest chip architectures. All these fields demand reliability when exposed to corrosive or high-temperature settings. In water treatment and oil recovery, its derivatives offer surface-active properties that remain intact even when exposed to brine, acids, or organic solvents. Years on the research side convinced me that no generic substitute matches the performance floor these chemicals bring, but with strength comes responsibility. Many manufacturers and end-users grapple with the knowledge that downstream waste contains lasting signatures—traces that challenge today’s infrastructure and yesterday’s assumptions about “disposable” chemicals.
Driving Research and Development Forward
Over time, the promise and the peril of perfluorobutyl sulfonyl chloride spurred innovation across academic, corporate, and policy circles. Researchers focus on new synthesis variants and look for milder, more selective modification protocols that trim hazardous byproducts. Universities work closely with industry to design derivatives that break down more easily after use—an answer to the saga of “forever chemicals.” Breakthroughs involve swapping out the perfluorinated chain for more degradable fragments, or attaching reactive groups that snap bonds under tailored environmental triggers. Peer-reviewed studies in respected journals point toward tangible progress, though uptake in industry moves slower than many would hope—it’s tough to unseat the champions of reliability, even as scientific consensus grows around the environmental stakes.
Toxicity and Environmental Burden
Hard conversations about perfluorobutyl sulfonyl chloride don’t dodge its toxic legacy. Animal studies and environmental survey data converge on findings of persistence, bioaccumulation, and organ-level toxicity. Chronic exposure links to liver, kidney, and immune system impacts; regulatory agencies flag tight limits for workplace air and effluent streams. The molecule’s resistance to breakdown ensures that even minor leaks last far downstream, entering soil, water, and food chains. In some heavily monitored regions, scientists detect measurable residues years after phased-out use, warning that society pays a compound interest on short-term chemical convenience. Many older waste protocols show their age—incineration at extreme temperatures, for example, helps but struggles with perfect completeness. Missteps in waste treatment amplify the consequences. These realities force not just chemical makers, but engineers, lawmakers, and public health experts to keep pressing for upgrades to testing, monitoring, and remediation.
Charting the Road Ahead: Future Prospects
Progress means rethinking both chemistry and stewardship. The relentless push for safety by design, greener synthesis, and downstream recycling must keep pace with the escalating demand for specialized materials. Academic consortia and public-private initiatives ramp up the search for alternatives that keep performance high but lower toxicity and persistence. Across regulatory tables, stakeholders revisit risk thresholds and push for wider adoption of lifecycle assessment. In the lab and in policy rooms, voices now press for full transparency along supply chains—chemical identity, source, and end-of-life pathways included. Calls for more nimble analytical monitoring tools grow louder, as the environmental persistence problem proves larger and more complex than anyone forecast when the first perfluoro-chemicals were scaled. My hope—shared by many in this field—is that by leaning on ingenuity, transparency, and a willingness to confront trade-offs, future generations can tap into advanced chemistry without inheriting uncontrollable risk.
Perfluorobutyl sulfonyl chloride sounds like something straight out of a chemistry textbook. Most folks outside the chemical industry would never come across it, but this compound plays a big role in some aspects of advanced manufacturing that touch our lives every day. What interests me most is how these chemicals work behind the scenes, rarely getting headlines, but quietly powering the technologies and goods we use without even knowing it.
What's It Used For, Really?
The main draw of perfluorobutyl sulfonyl chloride comes from its chemistry—stubborn carbon-fluorine bonds and a reactive sulfonyl chloride group. That mouthful of a name signals that it’s part of the PFAS family, sometimes called “forever chemicals.” These compounds don’t break down easily in nature, leading to concern, but their toughness is exactly why they end up in specialized applications. Think advanced polymers, specialty coatings, and electronics manufacturing.
Take electronics. Devices need materials that shrug off moisture, high heat, and harsh cleaning chemicals. A compound like this helps create materials that last longer and perform better, especially where regular plastics just don’t make the cut. Fluorinated materials built from these chemicals keep phone circuit boards from shorting out and help aircraft wiring deal with big temperature swings. As someone interested in tech reliability, I can tell you: those little ingredients make all the difference in daily device life.
In industry, this compound acts as a building block. Chemical makers use it to add fluorinated groups into bigger molecules—making new coatings that resist sticking, electrical insulation that holds up for years, or fuel cell materials that stay stable. It’s been used in surfactants and as an intermediate for specialty synthesis. That level of customization means better products, but also a bigger responsibility for safety all around.
The Dark Side: Environmental and Health Worries
Here’s the flip side. PFAS chemicals, including ones like perfluorobutyl sulfonyl chloride, build up in soil, water, and even our own bodies. Their strong bonds make them tough to break down, so once they’re out in the world, they stick around. Studies have tied some PFAS exposure to health risks. A Harvard study from a few years ago found detectable levels in drinking water affecting millions in the US. The science is still evolving, but the persistence of these substances means exposure can accumulate over time.
I follow environmental news pretty closely, and regulators across the world have tightened rules on PFAS compounds. Conversations in policy circles stress the need for safer alternatives and ways to break these chemicals down at the end of their useful life. Big companies now invest in better containment during production, remedial technologies to scrub water, and research into alternatives that balance function with fewer lasting impacts.
What Comes Next?
Society must weigh the benefits of chemistry that’s revolutionized materials against long-term environmental costs. My view: use these powerful chemicals where they truly add value, not just for the sake of convenience. Engineers and scientists have started developing greener materials, using similar performance features but with less permanence in nature.
While it’s easy to get cynical, seeing the chemical industry adapt is a hopeful sign. As a consumer, I keep an eye out for product labels and news on regulations. The more the public knows, the more pressure to innovate in ways that protect both our technology and our planet for the next generation.
I remember the first time I worked in a chemical laboratory where every substance seemed to carry its own story and its own risks. Perfluorobutyl sulfonyl chloride stands out among chemicals that deserve both respect and careful handling. This isn’t some benign lab material. It can do serious harm if treated lightly. My hands-on experiences with corrosive and toxic chemicals made it clear how preparation and diligence make all the difference.
Understanding the Risks
Let’s not sugarcoat it: perfluorobutyl sulfonyl chloride reacts aggressively with water and moisture in the air. This means inhaling its fumes or getting it on your skin turns into a real danger. The risk of severe burns and lasting harm is high. Inhaled vapors can damage your respiratory tract. Get any on your skin or in your eyes and you might be dealing with chemical burns that don’t just disappear overnight.
Safety Gear: Your First Line of Defense
No seasoned chemist trusts luck to handle chemicals. Gloves, safety goggles, and laboratory coats are standard, but with this compound, you need extra attention to detail. I always reached for nitrile gloves and a face shield when there was even a slight chance for splashing. Run the experiment inside a properly functioning fume hood. This step makes the difference between clean breathing and coughing fits—or worse. Fume hoods protect not just you but everyone else in the room. Before letting any student or lab partner near perfluorobutyl sulfonyl chloride, I checked their gear twice. There’s no overkill when it comes to safety here.
Storage and Handling Routines
Perfluorobutyl sulfonyl chloride doesn’t belong on a crowded bench. It needs to sit in a tightly sealed container, away from sunlight, away from humidity, and well below eye level to avoid accidents. Label everything clearly. I’ve seen too many near-misses when folks tried to skip this step—mislabels turned minor experiments into hazardous events. Chemical storage cabinets with robust ventilation and secondary containment trays limit the spread if a leak ever occurs. Never put this type of substance anywhere near acids, bases, or anything that could spark a violent reaction.
Clean-Up Isn’t Optional
Spills don’t wait for a convenient time. That’s why clean-up kits with neutralizing agents, absorbent materials, and protective equipment should be within arm’s reach before any work starts. I always keep emergency eyewash and shower stations clear and functioning. Quick action makes a world of difference if the worst-case scenario becomes reality. Everyone in the lab needs to know where these stations are located and how to use them—no exceptions.
Learning and Sharing
Training goes a long way. People tend to remember real stories and concrete instructions over lists of dos and don'ts. From my experience, practicing emergency drills and teaching the reasons behind each rule leaves a lasting impression. Tired of one too many close encounters, my lab group held regular refreshers. This helped everyone take personal responsibility and spot issues before they turned into trouble.
Regulatory guidelines, especially from OSHA and EPA, shape our safety standards for a reason. These aren’t senseless rules, but lessons learned from past mistakes. Chemicals don’t forgive shortcuts. Treat perfluorobutyl sulfonyl chloride with the control and seriousness it deserves. Trust me—your future self and everyone you work with will thank you for it.
Looking Beyond the Formula
Perfluorobutyl sulfonyl chloride stands out mainly because of its strong reputation in industrial chemistry. Its formula — C4F9SO2Cl — doesn’t have everyday familiarity, but in the world of synthetic chemistry, even a quick glance kicks off a conversation about environmental responsibility, safety, and persistent compounds.
Understanding Its Backbone
There’s a reason chemists take extra notice of perfluorobutyl groups. When every hydrogen in a carbon chain turns into fluorine, you get a structure that barely reacts with much of anything. Add on a sulfonyl chloride group, and the compound shifts into an entirely different gear: reactive enough for specialty chemical synthesis, yet so stable that natural breakdown becomes a rare event.
Folks in chemical engineering reach for perfluorobutyl sulfonyl chloride to build high-performance products—think surfactants with properties you just don’t get from regular hydrocarbon chains. Fluorine atoms dominate the molecule, forming a perfluorinated tail linked to a sulfur atom, then ending with a chlorine atom hanging off the sulfonyl.
Why It’s Gained Attention
Over the past decade, global attention around fluorinated chemicals has intensified. Long molecules like this one resist breaking down in soil, water, and even industrial waste streams. These “forever chemicals,” technically known as PFAS (per- and polyfluoroalkyl substances), can move far beyond where they’re produced.
Research keeps uncovering new findings about how persistent compounds impact health and the environment. From factory floors to distant water tables, PFAS compounds like perfluorobutyl sulfonyl chloride leave a fingerprint. They don’t vanish on their own. Media reports have even spurred governments to launch deeper investigations into their movement and toxicity.
Uses and Ethical Considerations
The practicality of perfluorobutyl sulfonyl chloride tempts many industries—electronics, water repellents, specialized surfactants, and pharmaceuticals all tap its capabilities. The catch comes from risk: balancing chemistry’s potential with honest stewardship. Facilities using or making perfluorobutyl sulfonyl chloride wrestle with waste management and air emissions that, without oversight, can contribute to PFAS contamination zones.
Health studies point to links between PFAS exposure and issues like thyroid disruption, changes to cholesterol levels, and even cancer risk. Data continues to evolve, but enough warning signs exist for experts to recommend limited, trace usage and rigorous waste handling.
Managing Risk, Exploring Alternatives
Tough regulations have forced some producers to shift away from long-chain PFAS, encouraging gradual adoption of shorter-chain versions or alternative chemistries that break down more predictably. True, these new chemicals don’t always match the performance of legacy PFAS, but the aim should stay anchored on minimizing risk where possible.
Anyone in a decision-making role around fluorinated chemicals holds power to shape their future. Proactively disclosing ingredients, seeking safer alternatives, and investing in better capture and destruction methods make sense—not just for compliance, but to maintain credibility and protect community health. Experience in process engineering taught me that even small operational changes, paired with clear communication, shape long-term public trust.
The Value of Transparency
People deserve to know how specialty chemicals influence their world, long before groundwater tests or newspaper headlines spark worry. Clear communication about a substance’s structure, potential, and drawbacks is the only way to connect science with ordinary life. In the case of perfluorobutyl sulfonyl chloride, everything starts with understanding its structure—and what it means for the planet that sits beneath our feet.
Storing a Potent Chemical with Respect and Care
Perfluorobutyl sulfonyl chloride isn’t a name that comes up outside of specialized labs or certain chemical industries, but its storage is a simple fact of safety that can’t get brushed aside. Only a handful of people ever lay eyes on this compound, and yet, every time it leaves the shipping crate and enters a facility, each staff member shares a responsibility. People sometimes forget that the story of chemical safety begins long before the first flask is unsealed. The place and way a toxic or reactive substance sits on the shelf mean everything. For anyone who’s watched an accident unfold because of sloppy storage, this feels personal. I’ve seen the result of inattention — and trust me, it’s not pretty.
Keys to Proper Storage—Why Every Detail Matters
Talking about perfluorobutyl sulfonyl chloride, we’re dealing with a reactive, moisture-sensitive substance. It’s got potential to release harmful gases in the wrong environment, something no one wants lingering in a lab or plant. Good storage starts with the basics: a tightly sealed container that keeps out air and moisture. Glass or select plastics hold up better over time than metals, as corrosion creates its own risks. Placing the material in a cool, well-ventilated room — far from direct sunlight and away from sources of ignition — isn’t just a routine step. That cool storage slows down unwanted reactions, and a dedicated cabinet or shed provides a physical barrier should something go wrong. I’ve learned over the years, a little paranoia about spills and leaks pays off big if the worst happens.
Labels rarely seem exciting, but in this case, they do a lot of heavy lifting. Clear markings, hazard warnings, and the date it entered storage go on every bottle. Whenever I’ve had to identify a container during a drill (or worse, during an emergency), that label is the quickest lifeline to the facts. Keeping detailed records allows for timely disposal — nobody wants to store outdated chemicals that have gone unstable.
Pitfalls and the Need for a Safety Culture
People sometimes get complacent. Maybe everyone in the lab knows about the hazards, maybe the best intentions rule the day… until a contractor or new staff member comes through the door. Closed storage alone isn’t enough. Training everyone about the reactivity and dangers of perfluorobutyl sulfonyl chloride stands equally important. Eye protection, gloves, and long sleeves become standard not out of bureaucracy, but out of real-world necessity.
I often think of the little things that drift out of mind until it's too late: rotating stock so the oldest material gets used up first, double-checking secondary containment for leaks, never stacking heavy containers above shoulder height. These aren’t formal policy phrases, they’re the rules that experienced workers teach each new generation. If a spill does occur, having a spill kit and neutralizing agents on hand shows respect for the lives working in that space. In facilities where people take shortcuts, trouble has a way of finding them eventually.
A Community Responsibility in Handling Hazards
The job of storing chemicals isn’t just about compliance, nor is it just about personal safety. It’s about protecting the broader community — everyone who works with the material and those who live nearby. By taking storage seriously, facilities avoid fires, accidents, and environmental contamination that can spiral out of control. It all comes down to caring for people who might never even know what goes on day to day in that storage room. Proper practice takes time and attention, but the cost of failure is written in headlines, not just fine print.
Getting Past the Long Words: What Are We Dealing With?
Perfluorobutyl sulfonyl chloride sounds like science fiction, and for most people, it’s just another chemical name cluttering up material safety sheets. A few years ago, I started digging into industrial chemicals after seeing stories on PFAS contamination in local water. The more I looked, the more I saw how these types of compounds, whether in manufacturing plants, labs, or supply chains, aren’t just technical concerns—they’re threads woven into the air we breathe and the water we drink.
Why Perfluorobutyl Sulfonyl Chloride Raises Concern
This compound belongs to the perfluorinated family. The industry values these substances for their resistance to heat and chemicals. These strengths turn into weaknesses with health and the environment. Perfluorinated chemicals don’t break down easily, and that can lead to them building up over time in soil, water, and living tissue. If you eat fish or drink from groundwater near a chemical plant, those compounds can get into your body.
Several large health studies tie exposure to “forever chemicals”—the wider PFAS group—to issues like cancer risk, hormone disruption, immune system problems, and reproductive issues. The science on perfluorobutyl sulfonyl chloride specifically lags behind more notorious PFOS and PFOA studies, but it shares their chemical backbone and likely sticks around just as stubbornly in nature.
Why the Environment Doesn’t Bounce Back Quickly
Take a small spill. With biodegradable substances, bacteria and sunlight chip away at the problem. But with these perfluorinated compounds, cleanup means battling molecules that hang on for years, if not lifetimes. Scientists find these chemicals in river sediment, deep water, and even in animals thousands of miles from a factory. Wildlife in the Arctic, people who live far from chemical manufacturing—contamination skips borders.
Regulators began to notice that these leftovers don’t actually wash away; they just move around and pile up. The EPA, for instance, categorizes many PFAS as hazardous, and countries in the EU now set strict limits on their release. Still, loopholes let lesser-known cousins like perfluorobutyl sulfonyl chloride fly under the radar.
Keeping People and Places Safe
Access to public information on these chemicals remains patchy. Companies use perfluorobutyl sulfonyl chloride to make coatings, package food, and produce specialty chemicals. If workers have no training or no solid protective equipment, exposure risk goes up. The rest of us face longer-term dangers from slow environmental buildup, especially with weak oversight or poorly enforced rules.
Setting aside industry jargon, people need clear information on which chemicals lurk in their food, water, and everyday goods. I’ve learned that sunlight and hope don’t wash these substances away—it takes law, investment in safer alternatives, and honest disclosure to shrink these risks.
Groups working on environmental health call for more public testing, tighter reporting, and investment in non-persistent alternatives. Pushing companies and agencies to clean up and close chemical loopholes pays off. For now, perfluorobutyl sulfonyl chloride stays off most headlines, but the underlying science and the stubborn persistence of its chemical family leave little doubt: this is a problem that doesn’t just go away on its own.