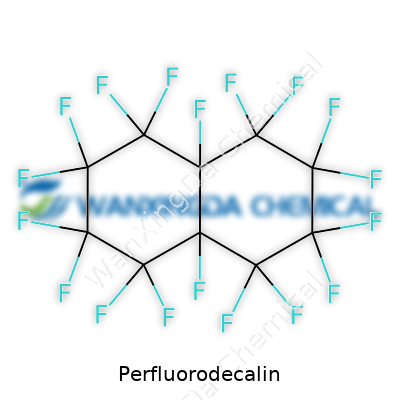
Perfluorodecalin: Commentary on Innovation, Safety, and Future Value
Historical Development
Perfluorodecalin holds a fascinating position in the history of modern chemistry. Its story goes back to the postwar era, when chemists explored stable, high-density liquids based on full fluorination. Part of a family of perfluorocarbons, perfluorodecalin grew out of work on oxygen carriers and blood substitutes. Researchers in the 1960s and 70s started paying attention because these compounds stuck around instead of breaking down, handled oxygen unusually well, and resisted chemical attacks most substances face in ordinary life. Over time, scientists saw more value for these chemicals outside straight synthetic labs. Decades later, the same slow-to-react, gas-carrying traits drew in pharmaceutical, medical, and technological researchers. This progression from obscure lab curiosity to multifaceted research tool shows how persistence and curiosity often spark the biggest shifts in scientific use.
Product Overview
Looking at perfluorodecalin, people see a clear, odorless liquid, almost ghostly in its lack of obvious chemical signature, but hiding some almost outlandish possibilities. Its value does not come from being flashy. What stands out is what it lets other people do—solving problems in breathing, drug delivery, and imaging thanks to its stubborn resistance to attack and broad gas solubility. Instead of making or breaking bonds in the typical sense, perfluorodecalin functions as a medium—showing up in artificial blood experiments, joining oxygen therapy research, and finding a spot in ophthalmic surgery. It carves a place for itself through utility, not appearance.
Physical & Chemical Properties
This liquid’s chemical backbone—decalin, but with every hydrogen swapped for fluorine—forms a tough molecular fortress. Nothing fizzles, burns, or reacts without overwhelming force. That molecular structure gives perfluorodecalin an odd mix: the slick feel of an oil, low surface tension, no flavor or smell, and a density higher than water. But what really stands out is how it pulls in gases. Few materials dissolve oxygen and carbon dioxide as well, which hints at why it comes up in studies on blood substitute technology or deep-sea breathing research. On top of all of that, it won’t mix with water or most organic solvents, keeping it separate and stable, which is exactly why scientists keep coming back to unlock more uses.
Technical Specifications & Labeling
The technical guidelines for perfluorodecalin reflect hard-earned research experience. Labels usually highlight purity levels, water content, and allowable levels of various organic or metallic impurities, because the intended application can swing from medical to industrial. In regulated settings, such as clinical trials or surgical use, purity standards matter a lot—a few stray molecules from old synthesis routes can mean the difference between sterile use and rejection as a medical-grade substance. Researchers expect full transparency, as do regulators. Strict labeling on hazards, transport, and disposal stem from both the environmental and human health perspectives, shaped by growing attention to persistent fluorocarbons in the long term.
Preparation Method
Making perfluorodecalin takes solid industrial chemistry—starting with decalin and blasting it with elemental fluorine. This isn’t backyard chemistry, since working with raw fluorine poses major hazards. Reaction control matters just as much as equipment maintenance. After the main fluorination, high-purity batches go through distillation and purification to strip out under-fluorinated leftovers and mixed-chain byproducts. The process evolved over decades, with improvements reflecting better reactor materials and monitoring, only possible once fluorine-resistant metals and specialty plastics became affordable. The end result—stable, colorless perfluorodecalin—stands as a testament to controlled, deliberate chemical engineering.
Chemical Reactions & Modifications
Turning perfluorodecalin into a building block for other chemical work faces real hurdles. Its structure barely bends for most reactions, a double-edged sword. On one hand, that keeps it safe as a carrier or medium. On the other, it challenges chemists hunting for new perfluorochemical derivatives. Specialized methods such as photochemical transformations or extreme temperatures can force the molecule to give up a fluorine here or there. Some researchers focus on blending instead of direct reaction, using perfluorodecalin’s inertness to pair with biologically active cargo for drug delivery or contrast imaging.
Synonyms & Product Names
Common names for this liquid—such as perfluorodecaline, FC-77, and octadecafluorodecalin—show up in research papers, medical device literature, or industrial catalogs. The actual molecule stays the same, but branding reflects the purpose behind each use. Sometimes the full chemical name appears on certificates for export or regulatory submission, reinforcing the need for clarity and traceability, which matters more each year as scrutiny on per- and polyfluorinated compounds increases worldwide. Seeing alternate synonyms like perfluoro-bicyclo[4.4.0]decane in dense technical documents can be intimidating at first, but all roads lead back to the same core chemical skeleton.
Safety & Operational Standards
Working with perfluorodecalin needs a clear-eyed focus on both acute and long-term safety. Inhalation of vapor or fine mist leads to mild respiratory irritation, but lower volatility minimizes that risk in most applications outside of mist sprays. The main worries show up in scaling and disposal: perfluorodecalin resists environmental breakdown, leading to caution about waste management and accidental releases. Regulatory focus, especially in places where “forever chemicals” raise worry, demands life-cycle tracking in supply chains. Good practices come down to protective gear, controlled handling, and waste management that prevents slow buildup in soils or waterways over years. Trained chemists and industrial hygienists continue to revisit these standards, reflecting growing awareness of environmental challenges posed by these persistent fluorinated materials.
Application Area
This liquid’s big claim to fame started in medicine, where perfluorodecalin carried efforts toward artificial blood and liquid ventilation for premature infants. Emergency medicine specialists once hoped to save lives by streaming oxygen-rich perfluorodecalin into damaged lungs. While practical hurdles limited broad adoption, those experiments paved the way for today’s clinical uses, including retinal surgeries where surgeons use it to flatten and stabilize delicate tissues. Its non-reactive character keeps it from interfering with tissue chemistry. Pharmaceutical researchers still look at perfluorodecalin as a unique vehicle to shuttle drugs or imaging agents into hard-to-reach tissues. In technology, companies experiment with perfluorodecalin as a coolant or cleaning agent for delicate electronics, since it won’t corrode parts or leave gooey films. Each field wrestles with ways to make the most of this stubbornly stable liquid, hoping to stretch current limits.
Research & Development
Research tracks involving perfluorodecalin rarely move in a straight line. Early hopes of replacing blood through full-body oxygenation faded, but the side routes opened up new ideas. Today’s labs test it for wound healing, as a delivery vehicle for hard-to-dissolve drugs, and even as a medium for high-resolution microscopy. Pharmaceutical companies and biotech startups circle around, digging through older patents and combining new ideas about surface modification with traditional uses. The goal is to blend stability, gas solubility, and biological neutrality. Development stands strongest where teams recognize limits—knowing that medical breakthroughs take years of controlled trials, but environmental safety issues can emerge just as slowly. This demands a measured pace, with researchers showing patience, transparency, and adaptability as plans shift with new findings.
Toxicity Research
Toxicity reports on perfluorodecalin steer clear of alarm, but steer just as strongly clear of complacency. Animal studies across decades show little acute toxicity at levels seen in medical use, since most of the perfluorodecalin either leaves through respiration or other routes. What worries regulators and environmental health scientists is the long-term side: this molecule doesn’t break down in soil or water the way organics do. Trace buildup, measured in slow drifts over years and decades, adds a note of caution to every new application. Recent regulatory pressure around per- and polyfluoroalkyl substances forces manufacturers and researchers to share full toxicity profiles and chase new data on chronic low-level exposure. Patients in clinical trials see robust consent forms reflecting these questions. Safety research never truly stands still in this space—it’s a work in progress shaped by every new study and regulatory review.
Future Prospects
The future for perfluorodecalin seems to rest on finding the right balance. Medicine, technology, and environmental science each see value, but all have to weigh the benefits against the slow, relentless persistence of this molecule in the wider world. Scientists keep looking for new ways to tap into its gas-carrying powers—exploring treatments for difficult lung diseases, refining eye surgery techniques, and boosting imaging or therapeutic accuracy. The results depend just as much on clear standards, open reporting of negative findings, and honest assessment of waste management. Across every sector, the call for sustainable and responsible chemistry grows louder, meaning the next big breakthrough may rest not in a miracle use, but in clever engineering that lets society have the benefits and leave no chemical trail behind. Perfluorodecalin stands right where thoughtful stewardship and constant research meet—a story still writing itself as global science and policy evolves.
Perfluorodecalin might sound like a tongue-twister from the chemistry classroom, but for anyone with a stake in advanced medicine, eye care, or even skincare innovation, it turns up in surprising ways. I’ve spent enough late nights buried in research papers to feel a mix of awe and skepticism every time a new chemical compound claims to change the rules. Yet, seeing doctors and labs reach for perfluorodecalin offers a strong nudge to pay attention.
A Breath of Fresh Air in Eye Surgery
Surgeons call perfluorodecalin “liquid magic” during intricate eye procedures. Retinal detachment can threaten vision in ways that rattle even the steeliest professional. In these cases, perfluorodecalin steps in to physically flatten the retina, creating a smooth surface for surgeons to work on. Picture a bubble gently nudging things back into place, letting delicate repairs unfold with far less chaos. Studies show that patients benefit from fewer complications and a better shot at keeping their sight. That direct link between a clear solution in a bottle and sharper vision in real life—that’s a big deal in my book.
Fueling Wound Healing and Oxygen Delivery
For years, scientists have explored ways to shuttle oxygen to tissues struggling to heal. Traditional methods, such as hyperbaric chambers, often felt bulky and out of reach for most. Perfluorodecalin carries more dissolved oxygen than water, which makes it an obvious contender for helping wounds breathe, literally. In my experience talking to doctors in burn units, topical treatments containing this compound have nudged slow-healing wounds toward recovery. These aren’t miracle cures, but real, visible improvement can mean the world to caregivers navigating chronic ulcers or trauma.
Breaking New Ground in Skincare and Cosmetics
I’ve watched the cosmetics world chase new trends with almost reckless enthusiasm—no surprise perfluorodecalin has caught their eye. Skincare brands turned to this compound to add more punch to their oxygenating creams and serums. The idea is simple: richer oxygen levels might boost skin renewal and brighten tired faces. While some claims drift into hype territory, patches of real science peek through. Clinical studies show increased skin hydration and support for some skin barrier functions. These products don’t flip a switch overnight, but consumers craving innovation have started listening to the research, not just the marketing blurbs.
What’s the Catch?
Nothing shows how complex progress is like chemicals crossing from surgery suites into moisturizer jars. Many regulators raise concerns about long-term safety in consumer products. Researchers I know insist on demanding greater transparency, better labeling, and a more careful look at side effects. No trending buzzword should leave out the simple need for clear risks and benefits, something many companies dance around. Lessons from blood substitute experiments, which exposed rare but serious immune responses, underline the importance of care. Safety studies still unfold slowly, but the medical use track record remains reassuring so far.
Moving Forward with Caution—and Optimism
Perfluorodecalin stands out because real people benefit every day—from regaining eyesight to easing stubborn wounds. Science reminds us that even familiar products must prove themselves in every new arena, and consumers deserve tools for smart choices. Forward-thinking labs and companies working with full transparency create a future where chemical innovation doesn’t outpace safety or trust.
Looking at the Promise and Limits
You may not hear about perfluorodecalin in most conversations, but this liquid pops up in some surprising places. Its job as an oxygen carrier makes it a bit of a star in certain medical circles. Think about eye surgeries—perfluorodecalin becomes almost a technological hero when doctors need something to support the fragile tissues in a retina. Also, it’s found its way into wound care and even into high-end skin-care products. With such a resume, folks naturally wonder how safe it really is for people.
What Science Tells Us
Perfluorodecalin belongs to a group of chemicals called perfluorocarbons. They carry large amounts of gases, especially oxygen. Because it doesn’t interact much with cells or break down into something else inside the body, scientists often describe it as “biologically inert.” This quality is key for safety. It means surgeons can use the liquid in sensitive places like eyes without worrying about damaging tissue or causing toxic reactions. Medical research backs this up—most studies on eye and wound use show low rates of issues when doctors use it as directed.
The story looks different when perfluorodecalin is inhaled or injected at high doses in ways the body isn’t meant to handle. Studies turn up breathing problems for animals when it gets into lungs in big amounts. There’s no evidence medical professionals suggest this kind of use for people, and that’s important—context always matters. This isn’t a compound for self-experimentation or fringe therapy. In my own work with health science writing, I’ve watched regulations evolve to keep such chemicals aimed squarely at cases where benefits far outweigh the risks.
Cosmetics and Everyday Use
Some skin-care brands add perfluorodecalin to serums, hoping to boost oxygen in the skin or help wounds heal. European and American health agencies haven’t flagged these cosmetic uses as risky, but the science stays thin on everyday skin exposure. The molecule’s size means it unlikely slips far beneath the surface. If there’s a worry, it’s not from soaking in—it’s what happens if someone has a rare sensitivity or if a manufacturer ignores proper formulation standards.
Looking Beyond the Hype
Producers push the idea of oxygen-rich skin creams like it’s a miracle. That feels out of step with what published data support. It’s true that perfluorodecalin helps in the lab and the operating room. It also appears true that daily skin creams aren’t much of a hazard, assuming everything gets made according to safety standards. For folks thinking about home treatments or unapproved uses, the risk sits much higher.
Safety Needs Respect and Oversight
Trust in chemicals like perfluorodecalin should come from strong oversight. Hospitals depend on regulatory review to keep treatments safe. Cosmetic makers need clear rules. Shoppers get the best protections when authorities test new uses and chase down any problems quickly. I’ve covered cases where governments stepped in after knockoff products skirted rules, and the fallout always lands hardest on those least able to spot the fakes.
That’s the landscape today: perfluorodecalin works for certain jobs in skilled hands. Companies, doctors, and users need facts, not fads, to keep that track record strong. Pretending a treatment works for everything only clouds the public conversation. Safety holds up best when it’s built on evidence and honesty.
Trust Starts with How We Handle Chemicals
Anyone who spends time in a lab knows—some chemicals demand far more respect than others. Perfluorodecalin falls into this camp. It comes up everywhere from ophthalmic surgery and cell culture work, to liquid breathing research. It looks unassuming: clear, nearly odorless, heavier than water. In reality, it asks for careful handling at every stage, especially when storage is involved.
Why Storage Choices Really Matter
Poor storage of perfluorodecalin carries real risks. Contaminated or improperly sealed bottles risk degrading quality and endanger research integrity. Too often, good work goes to waste simply because a storage shortcut led to product breakdown. Temperature swings, direct sunlight, or casual exposure to the atmosphere do more harm than most imagine. Heat can nudge decompositions along, direct lighting creates the potential for undesirable reactions, and air exchange may invite moisture, spoiling purity.
Through the years, my own lab work with sensitive fluorocarbons hammered home this lesson: sloppy storage quietly saps both time and money. Once, a shipment of perfluorodecalin sat near a window, its cap never properly tightened. Later, when someone couldn’t get reproducible results, everyone blamed the protocol. The culprit turned out to be simple environmental neglect. After that, we treated every reagent as if it cost the project’s success.
Best Habits: Concrete Steps, Not Just Theory
Research and lived experience point in the same direction. Store perfluorodecalin in a cool, dry spot—preferably in a chemical refrigerator that avoids freezing. Room temperature’s usually fine as long as the space stays steady, without wild daily swings. Use impermeable containers; glass with tight, chemical-resistant caps does the trick. Metal, unless specified by the manufacturer, is a gamble, since it can react over time or shed particles. Even plastics that seem sturdy may leach or crack after repeated opening and closing.
Above all, keep it out of direct light. This means turning away from benches under windows or exposed shelves. Everyday lights aren’t as strong as sunlight but continuous exposure still takes a slow toll. When in doubt, dark amber glass or a solid cabinet does wonders.
Why Labeling and Date Tracking Count
Labeling doesn’t just keep inventory neat; it saves teams from using the wrong stock or expired material. Anyone who’s scrambled for documentation during an audit knows that a clear handwritten label with date and initials beats any lengthy digital log in a pinch. Track the opening date and set a policy for replacing stocks after a reasonable timeframe.
Culture of Respect Beats a Written Protocol
Regulators and safety officers have their rules, but in real life, culture trumps checklists. If a team treats perfluorodecalin as precious, mishaps become rare. Clear policies, frequent reminders, and visible good examples from senior staff make these habits stick. For labs working on deadlines or with multiple hands sharing reagents, these small acts keep disaster at bay.
Making Science Work: Practical Over Perfection
Sometimes, supply shortages force labs to stretch a bottle longer than they’d like. Still, good storage practice can prevent needless waste. After years in research, I see responsible storage not as a chore but as an investment in results, reputation, and safety. No experiment survives poor storage, no matter how groundbreaking the science.
Perfluorodecalin’s unique character deserves nothing less than diligence. Store it well, and you protect both people and progress—one sealed cap at a time.
Perfluorodecalin finds its way into all sorts of places. You’ll spot it in eye drops, specialty cosmetics, even some medical labs. Its unique structure lets it dissolve and carry oxygen—a trick that’s inspired cutting-edge treatments for eye injuries or severe skin wounds. Still, as with nearly every new formula that claims a spot in healthcare and beauty routines, concerns and questions start to bubble up. Folks want to know: what kind of risks travel hand-in-hand with its benefits?
Why People Care About Side Effects
I get the curiosity. New chemicals promise a lot but rarely reveal their full story upfront. A bottle might say it hydrates skin or keeps lenses clear, but something that works so powerfully deserves a healthy dose of skepticism too. This stuff doesn’t grow on trees, after all—it’s cooked up in a lab, and every time you add something foreign to your body, you risk tipping the scales. That’s not paranoia, just common sense. Anecdotally, I’ve seen friends and peers wrestle with unexpected reactions after trying new “miracle” ingredients—each one taught us to dig deep before trust sets in.
What Science Tells Us
Researchers note that perfluorodecalin shows up mostly in topical formulas and specialized medical settings. The molecule itself is practically inert, which means it doesn’t break down or react much with living tissue. That’s good, to a point. Hospitals have used it inside the eye for retinal surgery, and labs monitor its movement closely. What stands out most: its safety track record seems strong when doctors use it as intended and in small doses.
But real-life use stretches well beyond the clinic. In skin products, perfluorodecalin usually hangs around on the surface, hydrating or delivering oxygen. Most people don’t notice any problems. A handful will see some redness or irritation, especially if they’re already sensitive or allergic to other ingredients. That irritation looks like a rash or mild swelling—annoying but not dangerous for most. Rarely, it causes a more significant allergic reaction that needs medical attention.
Why Those Side Effects Matter
Redness or swelling might sound mild, but for someone living with eczema, sensitive skin, or chronic conditions, it makes life harder. I’ve watched family members abandon promising products after just one bad reaction; trust takes a hit, and recovery takes longer than people think. Doctors warn that anything applied near the eyes should get extra scrutiny, especially since even small amounts of irritation there can cause ongoing discomfort. For those dealing with open wounds or compromised skin, the risks rise.
Practical Solutions and Smarter Choices
People who want to try perfluorodecalin-based products still have options to reduce their odds of trouble. Reading ingredient lists pays off, especially if you know you’re prone to sensitivity. Dermatologists recommend patch testing—a little dab inside the elbow or behind the ear—to spot any issues early before you go all in. For anyone with a history of allergies, it makes sense to talk through those concerns with a medical professional ahead of time. And anyone buying products from less-regulated online markets should pause; sticking with reputable brands that back up their claims with public data keeps things simpler.
It’s tempting to chase the next big advance, especially in personal care and medicine. But stepping back and evaluating both the benefits and the risks, without a rush, keeps you in charge of your own health. The more transparent companies and healthcare workers are about perfluorodecalin’s strengths and side effects, the easier it is for everyone to make honest, safe choices.
Perfluorodecalin looks simple at first glance—a clear liquid with no scent. The tricky part shows up when you try to mix it with something else. Anyone who has worked in a lab with this stuff knows it acts a little different than your everyday oil or alcohol. Most stuff just floats or sinks, staying neatly separated. Turns out, that’s a big deal if you care about making real-life products like skin treatments, eye drops, or even shimmering drinks.
Understanding the Science Behind Mixing
Perfluorodecalin doesn't play nice with water or most oils. Pour water over it, they split into two layers. Mix it with regular plant oils or alcohol, and nothing changes—they don’t blend. This happens because of its chemical structure. Fluorine atoms in perfluorodecalin create a slick shell, pushing away anything that doesn’t share that structure.
A lot of folks ask if you can just force it to mix by shaking harder, stirring longer, or using fancy machines. Science says no. Physical force only suspends droplets for a while until gravity wins and the layers split up again. The real trick comes down to something called emulsifiers. These are special molecules designed to bridge the gap, sticking to both perfluorodecalin and whatever you want to blend it with.
Where Mixing Matters in Real Life
Why bother mixing perfluorodecalin? That answer shows up most clearly in medicine and cosmetics. Doctors have leaned on this liquid’s oxygen-carrying abilities for artificial blood research. Cosmetic companies use it to deliver oxygen deeper into the skin or help heal wounds. Without the right blend, though, you get useless blobs instead of smooth creams or serums. Years back, I watched a cosmetic chemist spend weeks wrestling with the problem. Each failed mix cost money, time, and a little bit of hope.
Some people have looked into mixing it with volatile oils or natural extracts to boost product performance. Most of those experiments hit the same wall: perfluorodecalin won’t dissolve the way water does for sugar or salt. Lab folks then reach for surfactants—basically, molecules shaped like a head with two feet. One end likes water, the other loves oils or fluorinated liquids like perfluorodecalin. Shake things up with the right surfactant, and you get an emulsion. That’s the only way these two usually meet in a stable way.
Where We Go From Here
People interested in responsible chemical mixing need facts. Safety matters, of course. If you push the science, say with nanoemulsions, you need strong evidence it does what you promise—especially when lives are at stake in clinical uses. Real progress requires honest reporting about what actually mixes and why. Hype or shortcuts help nobody.
My advice: If you’re aiming for practical results, team up with a chemist who knows fluorinated liquids inside and out. Test thoroughly, especially if anyone uses the result in or on their body. And if your first dozen tries end in ugly layers or oily streaks, keep at it—that’s how most breakthroughs start. If it finally comes together, you’ll know you’ve earned it.