Looking at Perfluorodecyltriethoxysilane: More Than Just a Niche Chemical
How We Got Here: The Origins and Evolution
Perfluorodecyltriethoxysilane grew out of a broader movement—basically the exploration into fluorinated compounds that kicked off in earnest in the middle of the twentieth century. Folks in chemical research noticed early on that by combining organosilanes with perfluorinated chains, they could create new materials with surfaces that flat out refused to get wet or dirty. The original drive came from needs in electronics and textiles, but it didn’t take long before industries ranging from construction to medical devices latched on. As more manufacturers explored smart coatings and water-repellent tech, labs started digging into the recipe for longer chains and tighter silicon bonds. If you’ve ever worked in coatings or read about stain-resistant fabrics, you’ve felt the influence of this chemistry, even if you didn’t know the name behind the scenes.
The Stuff Itself: A Concrete Look at Physical and Chemical Nature
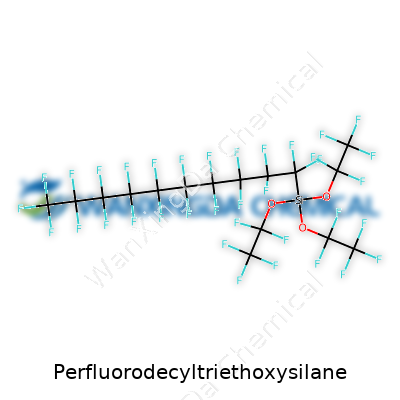
Perfluorodecyltriethoxysilane stands out as a liquid that’s clear and practically indifferent to nearly anything you throw at it—acids, bases, most solvents barely earn a shrug. This chemical, thanks to a perfluorinated ten-carbon backbone, resists just about anything sticky, from water to oil and things in between. Imagine trying to get a bead of oil to cling to a Teflon pan—it slides right off. You see the same sort of action here. Triethoxysilane groups dangle at one end, hoping to catch a roughened or hydroxylated surface (think glass, metal oxides, even natural stones) and build an almost invisible shield. The boiling point comfortably sits above most ambient temperatures, and the vapor pressure stays low, so you won’t sniff it evaporating unless you crank the heat. In practice, though, workers care a lot more about the stubbornness of that perfluorinated chain and the triethoxysilane’s eagerness to latch onto surfaces.
In the Lab and on the Label: Technical Realities and Safety Rules
Anyone aiming to use this in the field can’t just eyeball it and hope for the best. Standard packaging usually comes with regulatory warnings addressing long-chain perfluoroalkyl compounds and local rules about safe handling. Chemical safety data points to the need for gloves, goggles, and solid ventilation. Folks who mix this compound into sprays, coatings, or engineered surfaces need to respect these warnings, since even trace exposure over years has raised health and environmental questions. Proper ventilation isn’t some afterthought; it belongs front and center, especially if you’re prepping surface treatments or working on a production line. Labeling reflects guidelines from major chemical watchdogs, focusing on both human and environmental issues tied to the resilience of perfluorinated materials in living systems.
The Recipe: How the Compound Comes to Life
You won’t see this chemical bubbling away in your standard classroom lab. The usual process takes a perfluorinated alcohol and reacts it with triethoxysilane under a set of carefully chosen catalysts—often Lewis acids. Temperature and reaction time swing the yield, and stray moisture can chop yields and muddy the product. Succeed, and you get a bottle of slippery, nearly-inert liquid that preps glass like nobody’s business. Fail, and you end up with a tacky mess or a bottle of half-reacted precursors. Researchers often tweak this reaction, choosing milder catalysts or less reactive solvents to reduce side-products. It’s an art as much as a science, prone to improvements as new demands emerge for purity or scalability.
Getting Into the Reactions: Tweaking, Changing, and Repurposing
People interested in modifying surfaces reach for this compound because it reacts quickly, forming bonds with metals, oxides, and even polymers topped with hydroxyl groups. After application, the triethoxysilane part hydrolyzes—just a bit of water vapor kick-starts the fun—leading to bonds that stick at the molecular level. As for modifying the compound itself, researchers sometimes swap in different alkoxy groups, lengthen or shorten the perfluorinated chain, or introduce slight tweaks to drive better compatibility with a matrix. Throw it in a mix with other silanes, and you reshape the performance: more flexibility, higher heat resistance, broader solvent block. These little changes can mean the difference between a “meh” coating and a groundbreaking finish in optics or electronics.
Aliases and Identity Check
You’ll hear this product under a handful of names, which can cause confusion if you don’t watch the details. Trade catalogs list perfluorodecyltriethoxysilane, 1H,1H,2H,2H-Perfluorodecyltriethoxysilane, and sometimes even abbreviations like PFDTES. The core stays the same—a perfluoro tail, a silicon anchor, and ethoxy groups holding down the fort. Each synonym speaks to the same family, though small changes may signal a slightly different balance between handling requirements or performance on various substrates.
Where It Shows Up: Real-World Uses and Why They Matter
I’ve seen this compound pop up everywhere from slick touchscreen coatings to self-cleaning windows and outdoor sensor housings. In labs focused on micro- and nano-fabrication, it enables silicon wafers to cleanly release from molds. Its stubborn resistance to water and oil keeps graffiti from sticking to buildings, helps pipelines shed ice, and gives medical devices a longer fighting chance against fouling. Some cutting-edge textile manufacturers rely on it for stain-resistance, and the optics world loves how it keeps lenses and sensors free from greasy smudges without impacting transparency. This reach across industries highlights its value: everywhere a clean, shrug-off-everything coating matters, someone’s eyeing perfluorodecyltriethoxysilane.
Behind the Scenes: Research Drives and Unanswered Questions
Research teams keep asking how far they can push the performance and at what cost. The field buzzes with attempts to trim the cost of raw materials and scale up production without loading the world with persistent, hardly-breakdownable chemicals. There’s interest in greener synthesis routes, or in discovering silicone-based stand-ins that keep the dirt-shedding power but break down more kindly after use. In academia, the quest circles back to applications—how certain chain lengths change surface energy, what tailored substrates need for a perfect bond, which catalyst leaves the cleanest product. Even big industrial labs struggle to balance high outputs with environmental and performance targets, especially as regulations tighten.
The Safety and Health Puzzle
Concerns keep popping up, no matter how tough or useful this chemical proves in practice. Perfluorinated compounds, by and large, refuse to leave ecosystems or human bodies once they show up. Their persistence ties directly to bioaccumulation; that’s a mouthful, but it boils down to this: the compounds build up in wildlife and sometimes cross over into human water supplies. Animal studies raise alarms about developmental and organ-level impacts. Official word remains to tread carefully—use where benefit outweighs long-term risk, and always look for ways to reduce release. Safe operation means closed systems, vigilance about spills, and a real push to recapture vapor or wash-water. For researchers, finding safer alternatives or clever recycling strategies matters more now than ever. Every project, it seems, carries the question: Are we solving the right problem, or just swapping one challenge for another?
Where We Head Next: Prospects and Caution
Innovation keeps knocking: materials scientists dream up new uses in advanced batteries, smart windows, or self-cleaning solar panels. Each win brings back the tricky conversation about cost, lifecycle, and what happens at disposal. I’d love to see more transparent data on environmental pathways, not just the bells and whistles about superhydrophobicity. Teams working on biodegradable or short-chain alternatives need support, since tweaking the perfluorinated backbone even a little can make or break performance. Policy and market pressure will keep steering research, and no doubt tomorrow’s version will look a lot different from the legacy stuff. Real progress grows from honest talk about limits as much as technical achievements. For now, everybody who cares about long-term safety, groundbreaking surfaces, and environmental stewardship has skin in the game.
What’s Behind the Buzz About This Chemical?
People rarely overhear a conversation about perfluorodecyltriethoxysilane in the cafe or read about it next to weather reports, but this mouthful of a substance quietly weaves into daily life far more than most realize. Walking over a fresh plaza, eating from a coated frying pan, or looking through anti-fog-framed eyeglasses, odds are this chemical has been part of the background effort to improve surfaces and extend product life.
Why Manufacturers Keep Turning to It
Talking with engineers in building renovations taught me that everyone wants things to last, resist stains, shrug off water, and keep looking new. Perfluorodecyltriethoxysilane—let’s call it by its short name, PFDS—helps make that happen. It doesn’t work alone; factories link it to glass, ceramics, textiles, or stone to build a water-repellent or oil-shedding layer.
You see it at work in the architectural glass used on high-rises. Cleaning skyscraper windows costs time and money, especially above city traffic. Treating glass with PFDS makes rain bead up and dust wash away quickly. The initial investment pays for itself by cutting down on labor costs and keeping buildings looking sharp.
PFDS also pops up inside homes. It’s used to make kitchen counters less likely to soak up red wine or tomato sauce stains. Outdoor gear companies lean on it to create clothing that pushes water droplets away without making fabric stiff or uncomfortable. That’s good news during sudden summer downpours or winter slush.
Environmental Questions and Safer Practices
As robust as its stain-fighting powers can be, PFDS comes from a family of chemicals under more scrutiny for their persistence in nature. Perfluorinated compounds, including PFDS, hang around in soil and groundwater. They resist breaking down, creeping up in news stories about “forever chemicals” showing up in drinking water or fish. There’s a reason the Environmental Protection Agency and global health groups keep watching these substances closely.
That concern matters on a local and global scale. While chemical engineers and scientists keep working to shrink the environmental footprint, many companies have started searching for alternatives or developing ways to apply these coatings sparingly. Using just the right amount, at just the right place, limits risk while giving people the performance benefits.
Balancing Innovation and Responsibility
Getting more durable, cleaner products helps conserve resources. Nobody wants to replace glass panels, countertops, or clothing every year. Yet if the solutions lead to long-term trouble by building up in water and wildlife, future costs outweigh short-term wins. The challenge comes in being honest about those trade-offs, pushing for more research on safer molecules, and choosing wisely on where and how often to apply these coatings.
Growing up on a farm, I saw a lot of fixes come and go for barn siding and tools—some helpful, some best left in the past. Today’s challenges call for practical wisdom: focus on what really lasts, stay curious about the impact down the road, and hold manufacturers to a high standard. That keeps the balance between making products better for daily life and looking after the world everyone shares.
Perfluorodecyltriethoxysilane, a specialty chemical popping up in labs and industrial settings, isn’t something most folks discuss around the dinner table. Yet, how it’s stored matters in real terms. There’s real weight behind the push to keep chemicals like this safe, not just for compliance but for the people working with it and the communities around these facilities.
The Real Risks No One Wants at Work
Even a quick look at the chemical’s makeup tells an important story: it doesn’t mix well with water, it doesn’t get along with much, and it ranks pretty high among materials that can make a sloppy mess if left unchecked. The dangers come from those long, fluorinated chains. They resist breakdown and can end up lingering in places where nobody wants them. You stack a few barrels of it in the wrong spot, let a little humidity creep in, and you’ve got a problem much bigger than a spoiled batch.
Experience in the Lab, Lessons Learned
In my own time working in university labs, strict rules guided every step around bottles of this stuff. Just tossing it on a regular shelf wasn’t an option. One time, a graduate student left a loosely capped bottle out after use. By morning, an acrid smell filled the storage closet, and everyone spent an anxious day sealing HVAC ducts and preparing spill kits. In the end, nothing major happened, but it was a wake-up reminder on how small slipups can snowball.
Best Practices That Actually Make a Difference
The basics start with keeping it dry and away from direct sunlight. Store it in tightly sealed containers, preferably made of materials that the chemical won’t chew through over time. A lot of folks opt for flammable-safe cabinets, mainly to keep everything together in one controlled spot, since a scattered storage system invites mistakes. Silica gel desiccant packs inside cabinets help deal with stray moisture before it becomes an issue.
Organizing cabinets by chemical compatibility, not by alphabet, seems tedious, but it’s worth the extra step. Remember, PFAS-based chemicals have gained a reputation for persistence—they’re tough and can linger in the environment or on equipment for ages. Storing them carelessly opens the door for cross-contamination, which can affect results in a research setting or lead to regulatory headaches in production lines.
Protecting People—Not Just Paperwork
Proper ventilation isn’t just about ticking off a safety checklist. If a leak or fume builds up, it creates a workplace hazard. Good airflow keeps staff safe and reduces the chances of long-term exposure. Training goes a long way, too. New technicians and old hands alike benefit from refreshers on what to do in case of leaks or spills. Too many assume they’ll just “know” what to do, but muscle memory beats improvisation every time.
What’s Next?
Many companies and research groups are rethinking their approach to PFAS-related chemicals. The shift comes not just from regulatory pressure but from a real understanding of the health and environmental risks. Calling out bad storage habits matters more now than ever. If we treat chemicals like Perfluorodecyltriethoxysilane with the seriousness they deserve—by keeping them secure, dry, labeled, and separated—we protect both our work and our colleagues. That’s worth the hassle and cost of doing things right.
Why Some Chemicals Grab My Attention
I like to keep an eye on stories about common chemicals — not because I enjoy stirring up fear, but because too often, danger lurks where it shouldn’t. Perfluorodecyltriethoxysilane isn’t a familiar name for most of us. But if you’ve ever used a product that promises to make things “stain-resistant,” “water-repellent,” or “easy-clean,” you’ve probably brushed up against its effects. It usually shows up in surface coatings or as a treatment that lets spills bead up and roll away. That convenience comes with a conversation that matters to anyone who cares about what gets left behind after the cleaning is done.
What Science Tells Us About Safety
This chemical counts as a fluorinated compound. It joins a family known as PFAS — the so-called “forever chemicals.” Studies link some PFAS to health issues like cancer, hormone disruption, and liver problems. What makes them worrying is their tendency to build up, both in the body and out in the environment. They don’t break down easily. Over time, small exposures add up.
Perfluorodecyltriethoxysilane specifically doesn’t always come up in public research, but its close chemical relatives set an uneasy precedent. I haven’t seen high-profile health warnings about this compound itself, yet scientists have pointed to a risk that these kinds of substances, even if stable in the final surface, can eventually escape. This matters if dust or wear releases tiny amounts into the air or water. Once released, removing these chemicals from ecosystems takes decades or more.
Everyday Exposure and What We Can Do
For most people, the biggest source of risk comes not from working in a lab but from everyday items: fabrics, cookware coatings, and sprays to “protect” furniture. While industrial workers face more direct exposure, regular folks still end up with low but chronic contact that could add up. Spills on treated countertops or dust from abrasion bring the chemical closer than any of us might guess.
If you have pets or young children, the calculus shifts even more. Kids crawl, chew, and stick things in their mouths. Their bodies and immune systems still develop. Little exposures that wouldn’t worry an adult can hit harder. It’s sensible to ask: do I really need a super-hydrophobic spray or a stain-proof sofa if the protection comes with chemicals that might bring long-term health costs?
Looking for Better Answers
It’s not easy to avoid these ingredients entirely. Labels don’t spell out every fluorinated compound, and ingredient disclosure laws lag behind what people want to know. Still, more brands have caught on to demand for alternatives. Some companies now offer PFAS-free options for clothes, cookware, and cleaning products. Sometimes, the switch requires more effort — extra upkeep, or giving up that “worry-free” stain armor.
What gives me hope: broad bans on entire classes of fluorinated chemicals have started to take hold in some regions. Action at the governmental level works best here, since the stuff moves and accumulates over time and distance. Until regulation catches up, people who care about chemical exposure can shift their buying choices, ask companies hard questions, or keep a careful eye out for labels mentioning “perfluoro” or “fluoroalkyl” ingredients.
Let’s Ask Honest Questions
Products built to resist stains or water seem helpful, but the long-term effects of the chemicals involved need open discussion. With more attention on PFAS and similar substances in recent years, the spotlight won’t fade soon. If in doubt, I go with the simpler option — fewer treatments and less chemical armor at home feel like protection of a different kind.
Invisible Protection: Why Surfaces Matter More Than We Think
Think about the last time you spilled something on your favorite shirt or watched rain bead up and roll off a good raincoat. The technology that makes these little miracles happen isn’t magic—it comes from a surprisingly tiny source. Perfluorodecyltriethoxysilane, a mouthful for sure, stands right behind many of the spill- and weather-resistant surfaces you touch daily. It forms a layer so thin you couldn’t spot it with your eye, but this layer can turn ordinary glass or fabric into something that shrugs off water, oil, and even fingerprints.
You’ll see this compound at work on window panes designed to stay clear of fog and dust for longer, cutting down the number of times you need to pull out the Windex. Smartphone makers have caught on, too, adding it to the glass on screens to reduce smudging. Car windshields protected with this treatment stay clearer during rainstorms, which feels especially important when you’re driving late at night or during a downpour.
Helping Out Where Cleanliness Is Critical
Hospitals and labs need surfaces that won’t hold onto germs, liquids, or chemicals. Perfluorodecyltriethoxysilane helps create barriers on medical tools and benches so that harmful substances and bacteria have a tougher time sticking around. In my time working with engineers in cleanroom environments, they always put safety and cleanliness ahead of convenience—no shortcuts allowed. This treatment cuts down the need for harsh cleaning chemicals and frequent deep cleans, which, according to studies, supports both safety and long-term cost savings.
Museums and restorers turn to this material when they need to shield artwork, historical papers, or even old stone against stains and pollutants. Once applied, it repels grime without changing the look or feel of irreplaceable objects. Preserving the past doesn’t always require glass cases—or at least not as many—as a result.
Boosting Durability in Construction and Industry
Modern architecture uses more glass and open designs than ever, but glass isn’t exactly low-maintenance. Treating glass, steel, or tile facades with perfluorodecyltriethoxysilane means fewer streaks, less corrosion, and a longer lifespan for expensive building materials. Factories and food processing plants also use it so that oils, powders, and even sticky syrups don’t cling to machine parts. When you can keep things moving without nonstop cleanups, operations run smoother.
The Balancing Act: Innovation and Environmental Responsibility
This isn’t the most eco-friendly compound on the shelf. Since it contains fluorine, some environmental groups raise fair concerns about buildup over time and the need for safer synthetic chemistry. The industry has started working on alternatives and improving disposal practices. Finding the sweet spot between advanced performance and sustainability challenges researchers. Until then, smart regulation, proper application by trained professionals, and continued investment in green chemistry can help minimize risks.
In the end, perfluorodecyltriethoxysilane isn’t just a lab curiosity—it shapes what we touch, see, and clean every day. Whether you work in healthcare, take pride in polished architecture, or want your gadgets to last longer, you’ve likely benefitted from this scientific workhorse without even realizing it.
No Shortcut to Safe Handling
Perfluorodecyltriethoxysilane shows up in a lot of conversations in labs and factories that work with coatings, electronics, or even textiles. Some see it as just another chemical, but anyone who has dealt with it for a few years knows it deserves a little more respect than a basic bottle of acetone. Chemicals like this have caught the attention of health experts and regulatory bodies because they don’t just vanish after use. They often hang around both in the environment and, sometimes, in our bodies.
Nobody wants to discover problems the hard way. People who skip gloves or ignore fume hoods might not feel any trouble instantly, but that doesn’t mean the risks fade. I remember hearing stories from older colleagues who worked in labs before safety became such a big deal. Long-term effects weren’t obvious at first, but they started showing up years later: respiratory trouble, skin problems, and issues easy to sweep under the rug until too late. These risks aren’t theoretical, either. The Occupational Safety and Health Administration (OSHA) and similar groups in Europe keep nerves on edge about fluorinated chemicals because they’re good at penetrating protective barriers and tough to break down.
Why This Chemical Matters
Perfluorodecyltriethoxysilane’s structure helps make things water-repellent or increase durability, but those same unique bonds make it hard to get rid of. Some relatives of this compound have even popped up in groundwater and animal tissues. Small things add up—droplets on a glove or splashes that evaporate into the air. Research points to possible links between long-term exposure of fluorinated compounds and effects on cholesterol, immune system function, or even some cancers. Nobody can afford to brush that off.
People sometimes grow lax around chemicals they use every day. “It hasn’t hurt me so far” is wishful thinking. Community and workplace health depends on consistent diligence, not luck. In the US, for instance, agencies have placed focus on limiting occupational exposure to persistent chemicals because of the lasting impact even trace levels can have on soil and water.
Right Tools, Right Training
Every workbench with this chemical sitting on it should have more than goggles and gloves nearby. Good chemical-resistant gloves—nitrile, not thin latex—are worth the expense. A proper lab coat isn’t just something for company audits; it keeps droplets off your skin and street clothes. Lab teams must know the spill procedure like they know their favorite radio station. Inhalation can cause headaches or nausea, and skin contact brings irritation and possible absorption. Routine use of a functioning fume hood—not just opening a window—keeps the workspace safer for everyone.
Waste management shouldn’t be an afterthought. Chucking leftovers down a drain guarantees contamination somewhere downstream. Closed, labeled containers help, and trained staff need to manage proper disposal. Sometimes, people think waste vapor or rinse water isn’t a problem, but regulations keep evolving to seal those gaps.
Shifting Mindsets Goes Farther Than Rules
Safety around chemicals like perfluorodecyltriethoxysilane sticks best when it becomes a culture, not just a checklist. Companies who talk openly about mistakes—rather than punishing or hiding them—see fewer accidents because lessons get shared. Ongoing safety briefings and updates on chemical risks make a bigger impact than a yearly training video. People remember stories and habits, not just rules in a binder.
Choosing safer alternatives where possible, or using the least amount necessary, also changes the game. In industries where alternatives haven’t caught up, vigilance with protective equipment, ventilation, and waste management forms a line of defense that pays off over time. Every safe practice sticks with a worker and keeps communities around factories and labs a little healthier.