Perfluoroethylene: A Perspective on Its Place in Modern Science and Industry
Tracing the Historical Arc of Perfluoroethylene
Stories about synthetic chemicals usually start with a hunt for something more efficient, more durable, or less reactive. Perfluoroethylene’s history follows this pattern, emerging decades ago as chemical firms competed to solve challenges in materials science through the use of novel organofluorine compounds. During the mid-to-late 20th century, chemists found that swapping hydrogen atoms for fluorine radically altered a molecule’s character. In the case of perfluoroethylene, these carbon–fluorine bonds delivered exceptional thermostability and chemical resistance, making it attractive for situations where ordinary hydrocarbons would quickly degrade. This compound didn’t just drop out of the sky — it grew from a culture of innovation that often prioritized pushing materials beyond conventional limits. As an observer watching the evolution of industrial chemistry, I view perfluoroethylene as a symbol of both human curiosity and our enduring appetite for “miracle” molecules.
What Perfluoroethylene Looks and Acts Like
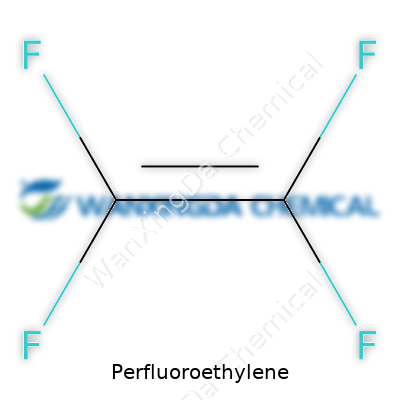
Try to conjure an image of perfluoroethylene and you’ll probably picture a transparent, colorless gas in a sealed cylinder or a process line. Its molecular structure (C2F4) gives it a heavy, almost “greased” feeling in industrial extraction and handling systems. The physical properties are instantly recognizable to anyone used to working with fluorocarbons: a low boiling point, high density compared to hydrocarbon analogues, and a resistance to solvency in both water and many organic liquids. The double bonds and the full fluorination—where all hydrogens are replaced with fluorine—set it apart. It does not burn easily, nor does it react enthusiastically with most chemicals. This quality isn’t just trivia; it’s what underpins everything from gaskets to cable insulation, right down to the maintenance rooms of semiconductor plants.
Technical Nuances in Specification and Labelling
Labelling and technical specifications for perfluoroethylene don’t swing wide of what industry standards call for. The detail that matters most isn’t only purity, though high-grade forms boast impressive purity percentages. Lab staff watch out for impurities like hydrogen fluoride or unreacted perfluorocarbon chains, especially in sensitive processes. Cylinder labeling routinely flags the nature of the compound, its hazards, and batch number for traceability. These aren’t bureaucratic details—they’re a hard-earned response to decades of safety learning and regulatory pressure. In my time following workplace chemical safety, small discrepancies on a label can lead to major operational headaches or even regulatory penalties.
Making Perfluoroethylene: Laboratory and Industrial Approaches
Manufacturing perfluoroethylene calls for fluorination, and that’s a word that carries some weight in chemical circles. Direct fluorination of ethylene typically does the trick, but executing it without setting off unwanted side reactions demands careful process controls and corrosion-resistant apparatus, often fabricated from nickel or Monel alloys. This isn’t a kitchen-sink chemistry project; reactors are built to withstand the aggressive nature of elemental fluorine gas. In addition, operators must balance pressure, temperature, and reaction times so that the outcome isn’t contaminated by unwanted byproducts. From the perspective of process engineering, the challenge of handling such a highly reactive gas sits alongside supply chain logistics and waste handling in the chemistry of advanced manufacturing.
Reactivity: More Than Inertness
Despite its reputation as an inert gas under ambient conditions, perfluoroethylene tells a different story under the right circumstances. High temperatures, electrical discharge, or powerful catalysts can open up channels for polymerization or addition reactions. The most famed transformation converts perfluoroethylene into PTFE (polytetrafluoroethylene)—better known as Teflon—a process that changed the world’s approach to nonstick coatings and chemical-resistant seals. In research circles, chemists look for ways to nudge this molecule into new forms, often chasing materials with even greater stability or specific electronic properties. It sometimes seems like a paradox: a molecule simultaneously prized for its unreactivity, but which only reaches its commercial promise when gently coaxed into controlled transformations.
Known Synonyms and Product Names in Circulation
The chemical industry rarely clings to a single name for any substance. Perfluoroethylene sometimes answers to tetrafluoroethylene or TFE, reflecting both its systematic identity and common trade usage. This spread of names can create confusion for non-specialists reading regulatory filings or safety bulletins. My experience working alongside purchasing and regulatory teams taught me that a chemical’s synonym list is not “trivia”—it’s a vital link in the chain that connects suppliers, safety officers, and end users, smoothing communications and reducing preventable errors.
Putting Health and Operations First: Safety and Practices
No matter how useful a chemical may seem, its safety profile shapes how it travels from storage to application. Perfluoroethylene demands respect—a single whiff of its Properties and the stories of accidental inhalation or cylinder mishandling stay with you. High concentrations displace oxygen and can suffocate in confined spaces, while long-term leaks may corrode metals that were once thought impervious. Proper ventilation, leak detection systems, and clear labeling minimize risk, and that means regular training and drills for workers. Regulations from agencies like OSHA enforce limits on exposure, and even research labs keep detailed logs on their inventories to avoid “oops” moments when running experiments after hours. Conversations with field technicians reinforce that “common sense” isn’t enough—you need protocols drilled until they’re second nature.
Application Areas That Rely on Perfluoroethylene
If you pick apart the workings of a semiconductor plant, medical device assembly room, or even an automotive plant, perfluoroethylene’s fingerprints show up in surprising spots. It plays a starring role as the monomer feedstock for fluoropolymer production—PTFE, FEP, and similar plastics that resist acids, solvents, and searing heat. Gasket manufacturers turn to it for chemical compatibility. Engineers choose perfluorinated polymers to line vessels storing aggressive chemicals or for wire insulation in aerospace-grade electronics. In each of these environments, the emphasis falls on reliability over decades of temperature swings, chemical exposure, and mechanical wear. Spend any time on a research or industrial site, and it becomes clear that its value lies not in splashy marketing, but the quiet assurance it gives when everything else starts to break down.
Research and Development: Where Curiosity Drives Progress
Curiosity doesn't just come from academic circles. Corporate labs and cross-disciplinary teams continue pushing the envelope on perfluoroethylene-based chemistry. Innovators chase advances in polymerization techniques that use less energy, produce fewer byproducts, or create resins with tailored surface properties for biomedical coatings or advanced filtration. Over the past decade, much attention has shifted to improving process safety and lowering environmental footprints, acknowledging that the chemistry isn’t just about yields or profits anymore. Today’s research groups measure success not only by technical breakthroughs but by how their products, processes, and emissions stand up to public scrutiny and regulatory review.
Toxicity Research: Separating Fear from Evidence
Concerns about toxicity dog nearly every halogenated organic compound, and perfluoroethylene ranks no differently. While short-term exposures may cause irritation and central nervous system symptoms at high doses, studies seeking long-term links to cancers or chronic disease continue. Toxicology isn’t about quick answers. Data accumulates slowly, sometimes contradicting earlier findings. Regulatory bodies often err on the side of caution, setting conservative exposure limits as new research unfolds. Workers and communities deserve transparency about what’s known—and clear planning for what isn’t. Health monitoring, emissions control, and substitution studies represent the kind of behind-the-scenes work that makes the benefits of fluorochemistry possible without repeating yesterday’s mistakes.
Where Things Might Head in the Years to Come
Peering into the future of perfluoroethylene feels like weighing the balance between commercial necessity and social license. The demand for robust fluoropolymers won’t ebb as device complexity climbs and industries pivot toward more extreme performance environments. At the same time, the pressure to “green” chemistry—through energy-efficient syntheses, closed-loop recycling, and development of less persistent alternatives—shows no sign of waning. From my conversations with industry professionals and environmental advocates, a clear consensus emerges: the goal is not to abandon useful chemistry, but to pair technical excellence with a commitment to health, safety, and environmental stewardship. The next breakthroughs will probably come from intersections—where chemistry, material science, engineering, and policy overlap, reminding us that science never stands still.
Seeing Perfluoroethylene in Everyday Uses
Perfluoroethylene might sound like one of those chemicals you only hear about in science labs, but it actually plays a regular part in lots of jobs that keep the world running. In many industries, folks rely on perfluoroethylene as a solvent, especially for heavy-duty cleaning where oil, grease, or wax build up. Think of situations where basic soap and water throw in the towel—perfluoroethylene steps up in places like dry-cleaning operations and certain manufacturing setups.
Over the years, I’ve watched small dry cleaners in my neighborhood swear by this stuff. They tout how it gets out certain stubborn stains and preserves fabrics that water would ruin. In electronic industries, techs often lean on perfluoroethylene to flush away oils and residues from sensitive parts. It’s also used in some specialty coatings and lubricants—people tend to trust its resistance to breaking down under heat or corrosive conditions.
The Health and Environmental Toll
There’s another side to this story. Talk with anyone who’s worked around these solvents for a few years, and safety comes up quickly. The Environmental Protection Agency and health professionals keep tabs on perfluoroethylene’s risks, especially with chronic exposure. People working with the chemical can face headaches, dizziness, or worse if they ignore ventilation rules. Some studies link higher levels in the air or soil to more serious conditions. It’s not just talk—the science points to long-term environmental persistence.
Communities near dry-cleaning shops and factories have raised red flags about possible groundwater contamination. When the chemical escapes into soil, removing it can cost businesses small fortunes. I've seen old industrial areas where cleaning up after perfluoroethylene leaks stretches on for years, with fears about water safety lingering.
Balancing Industry Needs and Safety
Folks running businesses often face tough calls. On one side, they want clean results and dependable products that compete on the market. Perfluoroethylene meets those needs, but not without trade-offs. Some companies have invested in closed-loop systems that catch fumes and recycle solvents, reducing what escapes into the air. Others are searching for less hazardous alternatives—hydrocarbon-based solvents or advanced wet-cleaning tech stand out as two options that keep growing.
Governments have tightened rules over the years. Permits, inspections, and waste controls have become normal, prompting shifts in habits for dry cleaners and manufacturers. I’ve spoken to owners who upgraded to modern machines just to keep up with shifting local laws. It’s a careful dance, and often the true costs—health, environmental, economic—don’t appear until years down the road.
Looking Ahead: Keeping People and Places Safe
Better training and real oversight stand out as strong answers. Employers who bring in comprehensive worker education programs usually see fewer accidents. The push for innovation in solvents could carry us to safer spot, but it takes steady effort and investment. Personal experience shows me that real change often starts when regular workers, not just managers, take these chemical hazards seriously. Communities who ask questions about how their air and water are protected often drive smarter choices by local businesses.
Perfluoroethylene’s role in cleaning up isn’t going away overnight. Balancing the drive for spotless results with a responsibility to people and the planet won’t ever feel easy, but honest talk and smart regulation can keep harm in check.
What Makes Perfluoroethylene a Health Issue?
Perfluoroethylene crops up in everyday life more than most folks realize. Known for its stain-fighting power, it often finds a home in dry cleaning and industrial degreasing. The risk, though, isn’t just for factory workers or cleaners. People bring dry-cleaned clothes into their homes and breathe in chemicals that don’t always vanish after pickup.
Experimental studies in animals have linked perfluoroethylene to a host of problems—from liver damage to cancer. The debate about its impact on people runs deep. There’s little comfort in laboratory findings when stories crop up about health complaints among workers and people living near dry cleaners. The EPA calls it a “likely human carcinogen.” For a parent with a closet full of freshly pressed shirts, that label sounds more personal than it does official.
Everyday Exposure: Invisible and Worrying
Back in the day, the family laundry felt like the safest place in the house. Now, science says chemicals like perfluoroethylene linger in the air and settle into fibers. Tests have measured perfluoroethylene in the air of homes. Reports show higher rates of certain cancers and other illnesses among workers who spend years handling the solvent daily.
A dry cleaner once told me about constant headaches and fatigue after long shifts, even with the door thrown open for ventilation. That’s not just anecdotal. Studies point toward increased risks for kidney and bladder cancer among exposed staff. It’s tough to ignore the mounting evidence that exposure over months or years can lead to more than just a mild headache.
Regulation and Alternatives
The federal government has set workplace limits for exposure, and more states have started to push for phasing the chemical out altogether. Some cities have banned its use in new dry cleaning businesses. People living above or near dry cleaners never signed up for the risk, and regulators know it.
High-tech machines and “green” solvents are gaining ground. Wet cleaning, which uses water and mild detergents, does the job for most fabrics and avoids hazardous fumes. Professional cleaners I’ve met say it’s just as effective for almost every stain. These alternatives don’t come with the cancer warning stickers.
Pushing for Change, One Step at a Time
It’s easy to give up hope facing entrenched habits and powerful industries. Still, consumers drive change. Choosing wet cleaning or liquid CO₂ cleaning over traditional dry cleaning sends a message. Parents ask tough questions at the counter. Cleaners get curious, too, especially after hearing about safer options.
For those who need to use a dry cleaner, ask how clothes get cleaned. Let them know safer alternatives matter. On a larger scale, supporting tougher regulations and better protections for workers saves lives. These shifts can’t happen overnight, but small steps keep invisible dangers from piling up in our homes and neighborhoods.
Breaking Down Its Physical Side
Perfluoroethylene sounds like a mouthful, but its main traits tell you everything you need to know about how it behaves. As someone who spent time in labs, I learned pretty quick that working with fluorinated chemicals means dealing with slick, almost oily textures and surprising stability. Perfluoroethylene exists as a colorless, non-flammable liquid at room temperature. Even when heated, it refuses to catch fire, which already sets it apart from a lot of volatile organic compounds that have everyone scrambling for the fire extinguisher.
It also gives off an odor that’s not easy to describe—nothing fruity or familiar, just a sharp scent that never lets you forget you’re dealing with a potent chemical. If you pour it, it moves with a distinct slowness thanks to a density heavier than water. Drop some in a beaker of water, and it’ll sink straight down, not float or mix. What caught my attention is its ability to handle rough handling—the chemical structure locks together tightly, so you’re unlikely to find it breaking down or evaporating in a hurry. This kind of stability often brings both confidence and concern, depending on what you want out of the chemical.
Chemical Properties: Not Your Average Molecule
Perfluoroethylene’s backbone, built entirely of carbon and fluorine, cranks up its resistance to most chemical attacks. You won’t see it reacting with acids, bases, or even with water. That’s why chemists turn to it when they need a solvent unphased by aggressive conditions. Fluorine packs a punch—the element forms one of the strongest bonds with carbon, and that leaves each molecule with armor against heat and oxidation. Toss it in a reaction flask where regular hydrocarbons would fall apart, and it’ll hold steady.
American chemists looked at this property back in the twentieth century, driving the birth of fluoropolymer industries. Because perfluoroethylene shrugs off most chemicals, industries often depend on it for specialty coatings and lubricants. In my own tests, it barely blinked in the presence of strong alkalis or acids that normally spell disaster for other compounds.
Why These Properties Matter
A chemical that resists breakdown might look ideal, but that comes at a price. Throwaway convenience turned into a long-term headache once scientists realized how stubborn perfluorinated compounds can be—“forever chemicals” isn’t an exaggeration. Once perfluoroethylene gets into soil or water, it sticks around. Environmental monitoring found these compounds everywhere from Arctic ice to human blood, and toxicity studies keep pushing the value of caution.
Regulators now scrutinize chemicals like perfluoroethylene, asking tough questions about safe limits and possible substitutions. The EPA and European regulatory agencies started looking hard at whether these compounds can be replaced or better managed to protect drinking water and food supplies. This kind of scrutiny forces anyone who uses or produces it to consider not just performance, but what happens after the workday ends.
Taking Responsibility: Where Do We Go Now?
Communities and industries need to treat perfluoroethylene with respect, not just because it works so well, but because it never really leaves. Finding alternatives means investing in green chemistry—molecules that do the job without staying forever. From my own time handling chemicals, it’s clear a quick fix usually brings long-term challenges. Waste treatment and containment need real support, not just rules on paper.
Getting a handle on perfluoroethylene starts with understanding both the power and risks baked into its molecular structure. Anyone who works with these compounds should be tuned in to both their practical uses and foreseeable consequences. The balance between innovation and environmental responsibility isn’t easy, but experience says honest reckoning with chemical properties is the only way forward.
Understanding the Risks Behind the Name
Perfluoroethylene doesn’t come up in dinner-table conversations, but you’ll find it in some labs and industries. Its uses bring about tricky questions of safety. The name itself signals a need for caution, hinting at chemicals tough on both humans and the environment. The facts remind us why proper storage and handling matter. Direct skin contact can trigger redness or irritation, inhalation might cause headaches or nausea, and spills have the potential to linger in soil and water for ages. No one enjoys learning about pollution years after the fact.
What Working with Perfluoroethylene Looks Like
Talking to people on the ground, they paint a picture beyond safety datasheets. Imagine lugging drums of the chemical into a storage room. Your boots crunch over absorbent mats, and you already know not to show up without gloves and a face shield. Ventilation is more than a checklist item—nobody wants to risk a cough that won’t go away.
Proper containers mean more than making sure they don’t leak. Folks rely on solid drum seals and secondary containment trays. Some companies go further—using open racks that give easy access for monitoring. If the drum feels warm or starts hissing, you’ve got early warning before something worse happens. Temperature plays a big role. Stashing containers somewhere hot or crowded can cause pressure to climb. A cool, shaded spot with steady airflow buys peace of mind. Once, I worked a summer job in a warehouse where someone stacked chemicals in a corner sunbeam by mistake. The boss found them just in time, and that memory sticks with me every time chemical storage comes up.
Regulation Isn’t Just Red Tape
Laws and rules often get an eye-roll, but with chemicals like perfluoroethylene, they exist for good reasons. Regulations set limits on how much vapor can collect, often requiring fume hoods or filtered exhausts. These aren’t little upgrades. They stop small accidents from turning into full-on emergencies. Labels and training make the difference between a quick cleanup and spreading contamination across a whole facility. The data backs this up; places with regular training see fewer spills and, when they do happen, smaller incidents.
Shared Responsibility in Safety
It’s easy to say that companies should do the right thing, but most accidents happen from habit and shortcuts, not malice. Workers need to know exactly what’s at stake. Simple routines like daily checks for leaks, logging temperatures, and reporting odd smells make all the difference. The same goes for emergency planning. A fire extinguisher looks impressive, but if it’s the wrong type, it won’t help much for a chemical blaze. Neighbors—inside and outside the workplace—deserve to know the risks. If a truck tips over on the way to a facility, or winds carry vapors outside, local safety plans kick in.
The Path Forward
Living with hazardous materials comes with responsibility. Nobody likes rules for their own sake, but with chemicals that last a long time in the environment and carry health risks, cutting corners isn’t worth the gamble. A clean storage area, sturdy containers, solid training, and a watchful team give everybody a fair shot at a safe day’s work. As more is learned about perfluoroethylene’s impact, we owe it to ourselves, and the next crew coming in, to get these basics right.
A Closer Look at What Sticks Around
The name Perfluoroethylene might not spark instant recognition like plastics or pesticides, yet this chemical has found a home in all sorts of industrial processes. Take a moment to look up at outdoor fabrics or consider the coatings on wires—there’s a good chance this compound helped provide stain resistance or insulation. As with many chemicals that promise durability, the conversation soon drifts toward what happens after the useful life ends. Does Perfluoroethylene linger in water, soil, or air? And what does that mean for communities and ecosystems?
Chemical Bonding and Persistence
Like many ‘forever chemicals,’ Perfluoroethylene belongs to the per- and polyfluoroalkyl group, often called PFAS. Scientists have measured these substances in rivers, groundwater, and sometimes in fish or wildlife. The very thing that makes Perfluoroethylene valuable—its stability—also turns into a headache for anyone trying to remove it from the environment. Most natural processes break things down bit by bit, but PFAS compounds hold onto their fluorine atoms like a stubborn stain on a white shirt. Some estimates show these chemicals can linger for years, sometimes longer, without breaking down easily under sunlight, bacteria, or even common filters.
Why Persistence Matters
This chemical durability turns into more than a technical issue. An EPA study found that some of these persistent chemicals travel far beyond the original place of use. Traces have surfaced in watersheds a good distance from any factory—meaning the movement isn’t easy to trace or limit. Researchers have raised alarms about PFAS impacts on human health, pointing toward possible links to thyroid disease, immune response changes, or increased cholesterol. The fact that Perfluoroethylene doesn’t “wash away” means communities can struggle for decades to clear up contamination. Farmers, for example, have voiced concern that soil or water that’s picked up these chemicals could stay unusable for crops or livestock for a generation.
Managing an Invisible Threat
Trying to get a handle on the spread of Perfluoroethylene takes more than an updated filter or a warning label. Regional water utilities now test for PFAS more frequently, but municipalities need clear guidelines about acceptable levels and ongoing monitoring. It’s hard for consumers to know if their drinking water or food carries a burden without transparent reporting from both companies and public agencies. When researchers at Harvard and other leading institutions began tracking the spread of these chemicals, they argued open access to contamination maps could help inform local responses and pressure polluters to clean up their act.
What’s Next? Innovation and Regulation
As the evidence piles up, companies feel the pressure to switch toward alternative chemicals that break down faster or pose fewer risks. Research into new water filtration methods—like activated carbon, reverse osmosis, or even advanced oxidation—could provide some relief. At the same time, policy makers continue considering stricter rules, fines, and bans targeting not just Perfluoroethylene itself but the whole class of compounds. On-the-ground experience says that communities do better tackling chemical threats early, rather than waiting until problems appear in the next generation’s soil or water. When industry, government, and regular people keep an eye out for invisible threats, the chance for real progress grows stronger.