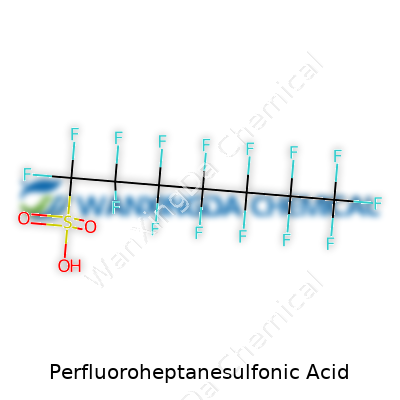
The Story of Perfluoroheptanesulfonic Acid: Hazards, History, and Hope for Change
Historical Development: A Cautionary Tale from the Fluorochemical Industry
Perfluoroheptanesulfonic acid, often found under acronyms like PFHpS or through synonyms, first entered the picture during the later stages of the chemical boom of the 20th century. Companies chasing performance chemicals looked to the ever-expanding world of fluorinated sulfonic acids. By the 1970s and 1980s, demand for stain-resistant coatings, waterproofing for fabrics, and firefighting foams pushed chemists to create compounds that could deliver on toughness. Industry gave little thought to environmental fate. Much like its notorious cousin PFOS, this acid found its way into countless industrial supply chains before scientists and regulators began to worry about persistent organic pollutants. Regulations lagged for decades, with bans and restrictions only appearing after environmental sampling showed just how stubborn this chemical could be in soil and water.
Getting a Grip on the Substance: Physical and Chemical Realities
In real-world conditions, perfluoroheptanesulfonic acid sits as a white powder or solid, not much to look at but tough as nails in chemical terms. Its molecular structure means it shrugs off acids, bases, and nearly all natural weathering. Water barely makes it flinch, giving rise to one of the biggest headaches for environmental clean-ups: once it’s in the water table or soil, almost nothing breaks it down. Boiling point lands high, solubility low, but that distinct set of fluorine atoms locks down reactivity. Plenty of chemists remember fluorinated acids not for their versatility, but for the stubborn aftertaste they leave in lab glassware, as traces remain even after several washes.
Unpacking Technical Standards and Labels
Walk into any research lab or browse a regulatory catalog, and you’ll notice the strict attention given to labeling perfluoroheptanesulfonic acid. Safety sheets highlight its extreme persistence and challenge in disposal. There’s a patchwork of regional standards — Europe typically marks it under persistent, bioaccumulative, and toxic substances. Safety labeling chases new findings and tighter restrictions, especially as toxicity studies catch up with mounting pollution trends. Unlike legacy solvents or reagents, fluorinated acids found their clarion call in environmental monitoring and toxicology circles more than in the mainstream press.
From Synthesis Bench to Factory Scale: How It’s Made
The origin story for compounds like perfluoroheptanesulfonic acid usually runs through electrochemical fluorination routes. Several generations of chemists spent careers refining the process, feeding hydrocarbons into aggressive reactors alongside sulfur trioxide or fluorinating agents. The finished acid comes out of complex mixtures, demanding advanced purification. Though these steps once sounded like triumphs of ingenuity, the legacy leaves cleaning crews contending with stubborn byproducts and nearly permanent waste. I’ve watched research partners shake their heads at the prospect of cleaning up after a mishap or spill involving a heavily fluorinated acid; stories circulate about traces lingering for months, even after aggressive washing.
What Happens in the Reaction Flask: Chemical Behavior and Modifications
Chemically, perfluoroheptanesulfonic acid doesn’t play well with others. Its main moment in the spotlight comes through sulfonation and fluorination reactions which push the boundaries of stability. Attempts to break down or modify the molecule usually meet fierce resistance due to the energy packed into those carbon-fluorine bonds. Some skilled synthetic chemists, craving challenge, take a shot at defluorination or derivatization purely for the scientific curiosity, but commercial operations stick to using it as is, often recognizing that modifying this molecule spells trouble beyond the effort.
Names and Lab Lore: Language and Legacy
If you’ve spent time in the laboratory, you’ll know how these chemicals travel under many banners. Alongside its formal name, it pops up in published work and catalogs as PFHpS, perfluoro-n-heptanesulfonic acid, and a sprinkle of foreign-language variants depending on where the work got done. Names sometimes show up attached to related salts or as parts of technical mixtures, confusing newcomers and veterans alike. Over time, researchers devise their own verbal shorthand for persistent pollutants, reflecting both respect and frustration.
Staying Safe: Operational Realities Beyond the Safety Data Sheet
Anyone handling perfluoroheptanesulfonic acid gets familiar with gloves, fume hoods, and obsessive cleanliness. Even trace skin contact can trigger irritation, and inhalation presents risks, though the true worry comes from environmental contamination. I still recall a situation where student workers went overboard on decontamination after a minor spill—in many ways, industry fears regulation less than unforeseen exposure or long-term liability. Disposal practices lagged behind best practice for years, so older sites often become notorious sources of groundwater and river pollution. Newer protocols ask for double containment, tracked movement, regular monitoring, and staff education across every handling point.
Application Areas: Surprising Spread Despite Stubborn Risks
Perfluoroheptanesulfonic acid spent decades woven deep in industrial supply chains. It turns up in surface treatments, technical cleaning, flame retardant foam, sometimes treatments for textiles and carpets. Many manufacturers now try to scrub their ingredient lists of such persistent chemicals, helping consumers dodge footprint build-up in living spaces. Regulatory pressure pushes industries to seek replacements, but some uses persist, particularly where no other chemical delivers the same dirt-repellent or weather-proofing punch. Some specialized electronics makers point out that while researchers warn against long-term risks, the performance edge still sways purchasing decisions.
Science Marches On: Research and Changing Attitudes
Those of us tracking this topic saw a shift about two decades ago. Scientists published evidence from wildlife and human blood samples, piecing together routes from factories to fish and up the food chain. Funding agencies now prioritize studies on fluorinated pollutants, particularly for ways to break down or capture these compounds before escape into the wild. Academics with memories of the earlier optimism about “inert” chemicals echo a changed tune as climate change, bioaccumulation, and ecosystem stability come front and center in both public dialogue and lab research. Groups look for molecular sponges, smart catalysts, or unusual biological pathways to grab or degrade perfluorinated acids. It’s a stubborn class of molecules, but not beyond scientific imagination.
Looking for Answers: What Toxicity Research Tells Us
Evidence from community and occupational studies, as well as laboratory animal work, points to serious risks: bioaccumulation in blood, liver, and key organs; links to immune suppression; and disruption of hormone systems. It’s not just the specter of cancer; chronic low-dose exposure leaves fingerprints in metabolic changes and developmental issues. I’ve listened to toxicologists weigh data from real-world exposures, acknowledging that while animal models show effects, the bigger question surrounds long-term consequences for both people and wildlife. Blood surveys from highly exposed communities continue to influence policy, driving bans and substitution programs on both sides of the Atlantic and in parts of Asia.
Prospects for the Future: Lessons in Persistence, Innovation, and Policy
With global awareness of “forever chemicals” now front-page news, industry faces a moment of reckoning. Pressure from both regulators and consumers shifts the dial, pushing for cleaner replacements or new technologies to clean up contaminated sites. Some promising bioremediation ideas start to move from the bench to pilot scale, driven by young researchers convinced the status quo won’t do. Local contamination scandals push lawmakers to close loopholes, ask more of polluters, and demand proof of safety before clearing new chemicals. With so much still unknown about subtle, long-term effects, vigilance and transparency look to define the next generation of chemical oversight. Most researchers agree: trust grows not from secrecy, but from open sharing of findings, mistakes, and improvements. In the end, the story of perfluoroheptanesulfonic acid reads as a warning and a challenge, and a call to look hard at both legacy problems and the choices we make going forward, for health, science, and the communities around us.
Why This Chemical Shows Up in So Many Places
Perfluoroheptanesulfonic acid, commonly called PFHpS, pops up in conversations around water safety, environmental health, and industrial chemistry. Not because folks love talking about it—most people have never heard of it outside a science textbook. In reality, this chemical makes life easier for factories and product designers, even though it comes with baggage for everyone else.
PFHpS comes from the bigger family of so-called “forever chemicals.” Companies often reach for it when they need water, oil, or dirt to slide right off a surface. Carpet against muddy shoes, takeout wrappers keeping grease at bay, or firefighting foams smothering flames all turn to chemicals like PFHpS. It’s not the oldest or best-known forever chemical, but PFHpS works a lot like its infamous cousin PFOS—strong, tough, and persistent way beyond its welcome.
Why Industry Leans on It
This stuff doesn’t break down in heat, light, or regular washing. That gives it a long shelf life in products. Electronics makers want clean workspaces, so they pick PFHpS-based cleaners to wipe dust and oil right off circuit boards. Factories making film, textiles, or shiny paper reach for it to get smooth, stain-resistant finishes. Airports and fire departments once relied heavily on its foam to fight jet fuel spills and blazing runways. Because PFHpS resists degradation, it sticks around long after the work is done.
The Problem That Won’t Go Away
The same properties that make PFHpS useful to companies present headaches for people and the planet. When PFHpS leaves the plant, it doesn’t just disappear. Rain and routine rinsing send it down drains and into rivers. Wastewater plants can’t filter it out effectively. The chemical lives on in soil, livestock, and wild fish long after its original purpose. In my own town, a local river tested positive for several forever chemicals, including PFHpS, just from upstream manufacturing runoff years before. None of us expected the stuff to show up in the water, but testing revealed how hard it is to keep these compounds out once they’re in.
Scientists have linked similar chemicals to a host of health worries—high cholesterol, immune problems, and even increased cancer risk in communities with high exposure. Researchers are still studying PFHpS to see if it carries the same risks, but the early signs look similar: build-up in bodies, long timelines before leaving, real concerns especially for children and pregnant women.
Looking for Better Alternatives
Most communities can’t wait for more research before acting on these chemicals. Some countries and states have started banning the manufacture or use of PFHpS. It’s still a struggle to track which products bring it in the door, though. Companies are tinkering with alternative coatings, foams, and cleaners made from less persistent compounds. Sometimes the substitutes fall short—they wash out in heavy rain, or don’t block stains as well. Still, these imperfect options seem safer for people and the environment.
Everyday folks can push for clearer labeling and tougher standards, like requiring companies to prove something is safe before coating our world in it. More research funding and independent oversight help, but it’s ordinary people, raising a fuss and demanding answers, who often force companies to think twice about old habits. If nothing else, the story of PFHpS calls for a lot less blind trust in materials that seem too good to be true.
A Closer Look at a Persistent Chemical
Perfluoroheptanesulfonic acid hits the news once folks start talking about “forever chemicals.” Once these substances get in the water or soil, they hang around for ages. That’s just how science built them—to resist breaking down. Factories lean on these chemicals because nothing else quite matches the slick, water-repellent, and grease-fighting properties. But what looks clever on a lab bench turns into big questions for anyone who cares about tap water or what goes into our ecosystems.
Why This Chemical Gets Scientists Talking
Researchers trace this acid to the broader family of PFAS chemicals, which have sprung up in everything from nonstick pans to firefighting foams. Folks measure trace levels in rivers, rainwater, and human blood samples worldwide. My background in environmental reporting means I’ve watched concern rise each time a new study tracks PFAS chemicals slipping into finished drinking water or turning up in schools or public buildings.
Not every chemical in this family acts the same way inside the human body, but scientists studying perfluoroheptanesulfonic acid circle back to one clear point: it sticks. The body just doesn’t flush it out quickly. Buildup is a real concern after years of tiny exposures from water, dust, or food. According to studies backed by the US Environmental Protection Agency and European Food Safety Authority, related PFAS compounds have shown links to problems like immune system shifts, slower child development, changes in liver enzymes, and some cancers.
Health Risks Are More Than Just Numbers
Living with uncertainty about what comes out of the tap changes how you think about what's safe. Families near factories using or releasing these substances might worry about cancer rates or rare illnesses. When blood testing begins to show PFAS family chemicals in nearly everyone tested, trust in safety rules takes a hit. It doesn’t help that research on the direct health effects of perfluoroheptanesulfonic acid lags behind some of the more studied PFAS. Still, lumping it with well-known toxic cousins like PFOA and PFOS feels reasonable until more information arrives.
From my experience tracking public meetings and local outrage, actions can stall while regulators and companies debate what counts as “safe.” Waiting for perfect answers gives these substances a head start. Past decades taught us the cost of ignoring chemical risks until problems boiled over. This time, clinging to uncertainty only guarantees regrets down the road.
Steps Forward Don’t Need to Wait for Certainty
Communities watching the news about forever chemicals want more than empty talk. They want tighter limits in drinking water and faster work cleaning up contamination. Scientists know more about how to filter PFAS at treatment plants or in homes with activated carbon or reverse osmosis filters. Governments in Europe and some US states are stepping out with tougher rules or outright bans where safer products exist.
Real solutions go beyond filters and stricter standards. Industry could shift away from stubborn chemicals like perfluoroheptanesulfonic acid, swapping in safer replacements or dropping non-essential uses. People play a key role, too—asking for transparency from brands or supporting public investments in clean water and soil cleanups. Safety isn’t a technical problem alone. It's a question of what kind of future we choose to build once we know the risks millions of people face in the present.
The Stuff That Lingers
Perfluoroheptanesulfonic Acid (PFHpS) isn’t just another chemical on the shelf. It’s a persistent organic pollutant, meaning it doesn’t break down easily. Once it gets released, it sticks around for ages. In my years covering environmental news and interviewing chemists, I’ve seen the headaches these “forever chemicals” cause for communities and workers alike. Forgetting to pay attention to storage and handling can leave behind messes that outlast generations.
More Than Labels and Lockers
Strong containment makes all the difference. I remember a research technician telling me about a leak that led to a full lab shutdown and a four-day cleanup. PFHpS, clear and often odorless, doesn’t warn anybody before it spreads. Sturdy, sealed containers built for corrosive and highly stable organics are essential. In a real sense, cutting corners here means real risks—not just for the person next to the drum, but for the river, the neighborhood, and the food chain if anything escapes.
Temperature and Spill Plans Go Hand-in-Hand
People sometimes underestimate how quickly things go sideways during a spill or an accidental splash. PFHpS keeps its form at room temperature and survives many common solvents. Refrigerated storage isn’t necessary, but keeping things cool reduces volatilization. Dry storage is key—water brings the risk of wider environmental spread. Any area handling PFHpS benefits from clear, practiced spill response routines. The best-run labs I’ve visited run drills with their team, so no one freezes up if something tips over.
No Lone Rangers
Working with PFHpS isn’t solo work. Proper training and up-to-date procedures make a big difference. You don’t want someone guessing what to do with unused acid or waste. In places with strict regulatory oversight, documentation and clear waste procedures protect workers and downstream communities. Incineration remains the best solution for disposal, done only in facilities capable of handling fluorinated organics.
Protective Gear: Non-Negotiable
Gloves, splash goggles, and chemical-resistant coats or aprons are a must every time. I’ve heard from staff who learned the hard way that a thin latex glove offers little defense against a corrosive spill. Nitrile or neoprene stands up better. Being able to change and wash clothing easily helps avoid tracking contamination outside the work area. It sounds obvious, but a pair of chemical-grade goggles has saved more than one researcher from a serious injury.
Better Habits Build Trust
Over the last decade, communities have grown more vigilant about what chemicals pass through their neighborhoods. I’ve seen protests and pushback when companies didn’t take accountability for spills or lax safety standards. Responsible handling isn’t just about personal safety—it shapes how the public views companies and research institutes. Everyone has a role to play in keeping these pollutants out of the environment and in keeping trust intact.
Perfluoroheptanesulfonic acid doesn’t make headline news like oil spills or wildfire seasons, but its reach quietly grows in water, soil, and the living things that depend on them. The compound sits in the PFAS family, also called “forever chemicals” because they don’t break down in the environment. These molecules stick around longer than most people would tolerate a leaking roof. Industries used them for coatings, repellents, firefighting foams, and all kinds of manufactured goods. The trouble starts with the qualities that made PFAS irresistible to industrial engineers: their resistance to heat, oil, and water means they linger wherever they turn up.
More Than Just a Lingering Ghost
Since these chemicals last for decades, contamination creeps outward. I’ve seen small-town wells test positive for perfluoroalkyl substances—residents didn’t ask for it, and it’s almost impossible for local utilities to filter out. PFHxS, the salt cousin of perfluoroheptanesulfonic acid, keeps showing up in fish, wildlife, even umbilical cord blood samples. That’s not just a contamination story, that’s a tale of chemical trespass straight through generations.
Most folks won’t willingly drink tainted water or munch on polluted fish, but nobody gets a choice when PFAS settle in local sources. US Environmental Protection Agency studies flagged that PFAS can build up in bodies over time, causing health effects that range from immune disruption to certain cancers. Europe added perfluoroheptanesulfonic acid to its restricted chemicals list after flagging bioaccumulative risks—meaning living things store it faster than they can lose it. Even low concentrations over years become a major hazard.
Why Clean-Up Doesn’t Cut It
Scrubbing PFAS out of water isn’t simple. Traditional filters fumble. Even high-end home systems funded by alarmed communities only grab some of it. For most water treatment plants, removing PFAS requires expensive upgrades—usually reverse osmosis technology or activated carbon filters. Destroying collected PFAS wastes means shipping hazardous concentrates to far-off incinerators, which themselves offer no guarantee of total destruction. Globally, communities still argue over who pays and where the burden lands, and companies face lawsuits for legacy pollution that may take decades to fade.
Turning From Reaction to Prevention
The science holds up—these chemicals move by water and air. Once released, they spread farther than most people realize. Experience in environmental advocacy suggests most progress comes not by cleaning what’s already polluted, but by not polluting in the first place. That means pushing for safer substitutes in manufacturing and tighter regulation on emissions into water and soil. Several countries have taken steps to ban production, but imported goods or legacy sites can keep the cycle going.
Transparency matters. People deserve to know if their drinking water exceeds advisory levels for PFAS. Community testing, transparent reporting, and strong requirements for industrial disclosure create long-term trust alongside safety. Sources of pollution need clear markers and accountability. For real progress, support must reach all communities—urban, rural, and Indigenous—since exposure risk doesn’t play favorites.
The world can’t unlearn what it knows about perfluoroheptanesulfonic acid and its kin. By putting data, clear guidance, and priorities in place, people and leaders can put health and environment ahead of corporate convenience or short-term profit. The future hinges less on cleaning up yesterday’s risks and more on keeping tomorrow’s water safe from forever chemicals.
Decoding the Molecule
Perfluoroheptanesulfonic acid stands out among fluorinated compounds. Scientists refer to this molecule as PFHpS. The intrigue here comes not just from what it does but from how it’s built. Its formula, C7F15SO3H, leaves no room for accidental mistakes—seven carbon atoms, fifteen fluorine atoms, a sulfonate group, and a single hydrogen. The backbone stretches out in a straight chain of carbon atoms. Each carbon carries fluorines wherever they can bond—none gets left out or replaced by hydrogen. At one end, a sulfonic acid group slots in, ready for action.
Why the Structure Matters
Anyone who has handled surfactants in the lab or industry knows that structure decides fate. The perfluorinated tail isn’t just there for show. Swapping hydrogens for fluorines gives the molecule tremendous chemical stability. This unique structure means PFHpS barely budges in the presence of heat, acids, bases, or even tough oxidizers. That’s great for industrial uses but spells trouble for the environment.
The sulfonic acid head gives PFHpS a handle to grab onto water and surface proteins. If you’ve ever watched oil beads scoot across water, you’ve seen how regular surfactants struggle to keep things together. PFHpS sneaks in with a hydrophobic tail and a hydrophilic head, gripping both oil and water, making it more effective at lowering surface tension than many mainstream surfactants.
Health and Environmental Impact
Reading up on the persistence of compounds like PFHpS points to a big reality check for anyone interested in chemical safety. Fluorinated compounds resist breaking down. That’s a double-edged sword. Once introduced to the environment, PFHpS hangs around in soil, water, and living tissues for years or even decades. Research published in Environmental Science & Technology tracks PFHpS in human blood and wildlife, underscoring how easily this molecule moves through water and food chains.
Unlike many short-lived substances, PFHpS can move across continents. Even places far removed from the industry, such as the Arctic, record the presence of these compounds in snow and wildlife. Science links perfluoroalkyl substances with health effects like liver changes and potential endocrine disruption. Regulators in different regions now place limits on acceptable levels, reflecting real concern over long-term impacts.
Looking for Solutions
Having worked in labs and spoken to industry professionals, one thing stands out: once PFHpS spills or leaks, getting rid of it isn’t easy. Standard water treatment can’t remove it fully. Newer methods, like advanced oxidation or high-temperature incineration, offer hope, but come with high costs and technology barriers.
Some researchers are chasing better answers, like bioremediation or chemical breakdown strategies, but we’re not there yet. I believe moving ahead takes investment in pollution prevention and tighter controls on industrial runoff. Substitution with safer compounds could take the edge off the environmental risk, and better monitoring will keep people and ecosystems safer.
A Call for Responsible Innovation
Anyone using powerful synthetic compounds like PFHpS owes it to society to think ahead about unintended impacts. The unique chemical structure offers industry a useful tool, but every benefit deserves a clear-eyed look at the potential cost. Using all the scientific evidence at our disposal—plus some common sense—can steer us toward products and processes that lock in performance without leaving pollution behind for the next generation to solve.