Perfluoroheptanoic Acid: Unpacking Its Past and Present
Tracing the Story of Perfluoroheptanoic Acid
Looking back at the long story of perfluoroheptanoic acid, one sees tracks laid down back in the days when chemistry laboratories buzzed not about climate impact or ecosystem risks, but about making substances that did the job in manufacturing and industry. The drive for progress led chemists to seek new molecules that resisted solvents, shrugged at heat, and stayed stable even under tough conditions. Perfluoroheptanoic acid stands as one of the offspring from that era. As a member of the perfluoroalkyl carboxylic acids family, this compound shares the backbone that made fluorine-rich chemistry so attractive: a chain that resists breakdown and does not give up its grip on physical toughness easily. As other perfluorinated acids slipped into public scrutiny, so too did perfluoroheptanoic acid, the debate echoing far beyond chemistry circles.
Getting a Grip on Its Basic Features
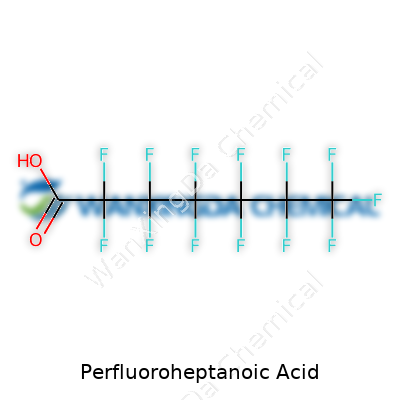
Perfluoroheptanoic acid wears a label that reads like a tongue twister, but its makeup is plain as day to anyone with chemistry under their belt: seven carbon atoms, each crowded with attached fluorine atoms, capped with a single carboxylic acid group. This structure hands it a set of traits that have long caught the eyes of industry: dogged resistance to both heat and many chemicals, a habit of not sticking easily to other substances, and a stubborn persistence that makes it hard to shift from places it’s been used. The acid sits as a waxy or oily solid at room temperature, rarely volunteering to dissolve in water but making itself at home in some strong solvents. Its intense stability, for both good and ill, shapes much of the story that follows.
Behind the Scenes: Synthesis and Reactions
Making perfluoroheptanoic acid does not call for wizardry but it certainly demands caution and the right equipment. The go-to method remains electrochemical fluorination, where a hydrocarbon chain gets flooded by fluorine atoms under high voltage until all hydrogens have been swapped out—a process known to those in the field as both a powerful tool and a source of headaches, given fluorine’s stubborn and hazardous character. As for chemical reactions, this acid sidesteps most traditional chemistry, holding tight to its electrons while most other acids would be swept into reactions. Changes to the chain tend to happen only in the hands of very specialized reagents or under strong conditions, which only deepens the environmental questions tied to it.
Labeling, Synonyms, and Technical Details
Chemists, regulators, and manufacturers all use slight variations of the name, and any quick glance at research or inventory sheets will turn up tags like heptanoic acid, perfluoro-, PFHpA, or sometimes the related chemical shorthand. The acid’s molecular weight, melting point, boiling range, and all those other data points matter more in the lab or in regulatory filings than in a global conversation—yet, knowing what you have in hand shapes every conversation about its safety and risk. Guidance from global regulatory groups pushes a clear label and identity for it, as the risks of misidentification stretch beyond paperwork into real-world exposure dangers.
Safety, Standards, and Operations
Coming face-to-face with perfluoroheptanoic acid, one quickly learns respect is warranted. Exposure can bring toxic effects, ranging from possible irritation on the skin or eyes to potential longer-term dangers if absorbed or inhaled over time. Like many in its chemical family, its resistance to breakdown creates a hidden trap: spills, improper disposal, or accidents lead to contamination that simply does not fade away with the seasons or rainfall. Labs, factories, and warehouses that handle it fall under a strict set of local and international guidelines for storage, transportation, and waste management. Ventilation, personal protection, and spill containment cross the line from good practice to outright requirement.
Impact and Uses: Not Just the Usual Suspects
Applications once set the tone for excitement with perfluoroheptanoic acid making an appearance in the production of fluorinated materials, as a surfactant in emulsion polymerization, and as a tool in both chemical research and analytical work. The same traits that handed it fame—the ability to repulse water and oil, to cling to surfaces without breaking down—drew researchers and manufacturers to it as a fix in tough engineering problems. Industrial dyes, surface coatings, and advanced composite manufacturing once leaned on this acid in processes where nothing else could compete. Over time, pressure from environmental and health research has worked like a slow tide, eating away at the practicality of its use and prompting a fresh look at alternatives.
Digging into Toxicity and Environmental Risks
Reports on perfluoroheptanoic acid’s effects on human health and the environment keep piling up, much as they have for substances like PFOA and PFOS. Persistence plays out as a long-term curse. This acid doesn’t break down easily in water, soil, or even the body; tracking studies link perfluoroalkyl exposure to developmental, immune, and carcinogenic risks in animals, with mounting concern for potential human effects. Studies point to bioaccumulation in aquatic life and possible movement up the food chain. Researchers, regulators, and advocacy groups now shine strong lights on legacy pollution from this class of acids, forcing regulators and companies to address cleanup, mitigation, and long-term monitoring rather than just risk assessments in the filing cabinet.
Chasing Better Answers in Research and R&D
Not enough people outside research circles grasp how much time and money now spins around finding less damaging replacements. Labs from Asia to Europe have thrown themselves into designing surfactants, process aids, and materials that carve out the same spot in manufacturing without leaving a toxic trail. Research tracks how this class of acids move through drinking water, the food chain, and even the air we breathe. Fresh analysis from government agencies and academic teams tracks hotspots of pollution, seeking to link sources, production, and concentrations with health outcomes. R&D in industry now wears a double harness: pushing forward on new uses while fielding pressure to switch out known environmental troublemakers.
Glimpsing the Road Ahead: Policy, Alternatives, and Responsibility
Policy wheels turn slowly, yet each twist and turn of scientific discovery brings firmer recommendations to scale back or outright ban applications of perfluoroheptanoic acid. Some regions have already walled off its use in agriculture, food packaging, and textiles—the places where its tough, slippery chain once made products last longer and repel stains better than anything else. Public pressure, fuelled by a stream of toxicology research and headlines about "forever chemicals," gnaws at the last holdouts of traditional uses. The future rests on finding safer chemistry that still pushes technical boundaries. Practical solutions mean treating contaminated water, cleaning up old spills, and building better detection tools—not just finding substitutes but learning from the slow-motion crisis that perfluorinated acids have built up over decades.
On Experience and Lessons Learned
As someone who has long followed the arcs of industrial chemicals and their aftershocks, I see in perfluoroheptanoic acid a warning against giving progress free rein without checking the rearview mirror. Early advances in performance and material science brought real improvements, but their full cost waited for the fog to clear. The persistence and toxicity tied to this acid lay heavy on any decision to keep it around. Fact remains, real progress comes not from repeating the same mistakes with new names, but from building science, policy, and public awareness into a trifecta that keeps innovation in check with accountability. The next generation of chemists must remember this story, or it will repeat with a new face and the same price.
Everyday Products, Hidden Chemistry
Try picking up a stain-resistant frying pan, or buying rainproof hiking gear, and the promise of “non-stick” or “water-repellent” leaps off the label. Dig a little deeper and you’ll find a family of chemicals often driving these benefits—one of them being perfluoroheptanoic acid, sometimes called PFHpA. This compound doesn’t get the same attention as its cousin PFOA, but it still shows up in many corners of modern life.
Why Companies Turn to PFHpA
Manufacturers started using perfluoroheptanoic acid to produce coatings that keep grease and water away from food wrappers, carpets, and even electronic components. The stuff works because of its unique molecular structure. The fluorine atoms locked tightly around its carbon backbone give it a kind of chemical armor. Grease, water, and stains can’t easily grab onto surfaces treated with this compound. Compared to old-school solutions that washed away or wore off quickly, these coatings seem to last forever.
Health and Environment: The Tradeoff
That’s exactly the issue. These perfluorinated compounds don’t break down. Once released, they enter soil, water, and air, and stick around for decades. I remember reading reports linking this type of chemical to water contamination near factories. Locals and public health officials scramble to test wells. The outcome? It rarely looks good. Long-term exposure can build up in our bodies. While scientists still sort out the degrees of risk, links to health effects—like problems with cholesterol and immune response—keep popping up in new studies.
Experts say the main exposures don’t come from your jacket or the takeout box, but rather from drinking water and some foods. One study from the CDC flagged detectable levels in blood samples from the general public. The uncertainty about how much harm these cause means regulators keep coming back for more data, new toxicology studies, and tougher limits.
Industry Knows Change Is Coming
Living with this long-lasting chemistry feels like squeezing the most out of short-term convenience, at the cost of long-run environmental headaches. I’ve watched companies shift away from the older, longer-chain PFAS compounds, eyeing newer, shorter ones or all-out alternatives. Yet, that change moves slowly. Some alternatives may seem safer in the lab but don’t always perform or behave the same way in the environment. For smaller businesses, the switch takes cash, patience, and know-how.
Personal Choices and Bigger Solutions
As a consumer, I pay attention to labels—sometimes you won’t find these chemicals named directly, but phrases like “fluoro” or “non-stick” drop hints. I try to support companies that cut out PFAS entirely or use transparent supply chains. Still, personal choices only go so far. The bigger breakthroughs come from regulations that take public health seriously and fund real innovation. Phasing out chemicals like perfluoroheptanoic acid won’t happen overnight, but moving the market with better oversight, research, and consumer demand does make a difference.
Learning about compounds like PFHpA serves as a reminder: convenience sometimes comes tangled up with costs nobody expected. Facing up to those risks, keeping pressure on industry, and pushing for better choices helps ensure the story of modern chemistry doesn’t become a cautionary tale for future generations.
What’s the Truth About This Perfluorinated Chemical?
It’s tough to keep track of chemical names that sound like tongue twisters, but perfluoroheptanoic acid gets attention for a reason. Scientists group it with “PFAS”—the so-called forever chemicals. These substances have popped up everywhere from firefighting foams to electronics, non-stick pans, and many industrial processes. Unlike spilled milk that can be wiped up, PFAS can last in the environment and bodies for years. Some regulators have started asking what long-term exposure really means for regular folks.
Why Should People Be Concerned?
The problem with perfluoroheptanoic acid is how stubborn it acts. Drinking water, soil, and even food can build up traces. Bodies don’t get rid of it easily, so it sticks around. The Centers for Disease Control and Prevention and EPA have said some PFAS chemicals link to thyroid disruption, decreased fertility, immune effects, and higher cholesterol. Some research also connected exposure to higher risk of certain cancers, including kidney and testicular. While direct studies about perfluoroheptanoic acid are less common than for its cousin PFOA, the family traits carry enough warnings that people should take notice.
So How Do We Get Exposed?
Most people won’t see a bottle labeled perfluoroheptanoic acid on a store shelf. Exposure tends to sneak in quietly. Treated food packaging, water run-off from factories, and even treated textiles carry these chemicals from point A to point B—and eventually into human bodies. Scientists found PFAS in the blood of most Americans they tested, showing how widespread these substances have become. Few folks knowingly choose exposure; it usually happens through the little things: drinking water, handling wrappers, wearing stain-resistant clothes, or breathing in dust indoors.
What Are the Real-world Impacts?
My own community once dealt with a contaminated groundwater issue. Locals attended meeting after meeting to learn what PFAS meant for their children’s development and their drinking water. Concerns about possible cancer risks hung heavy over each gathering. Parents wanted honest answers about whether lifelong exposure would haunt families decades down the line. Though nobody wants to panic, the uncertainty alone can take a real mental and social toll—one that is hard to quantify but easy to feel.
Looking Forward: Moving Past Worry to Action
Companies can phase out older, more hazardous PFAS, including perfluoroheptanoic acid, but old stockpiles and waste stick around. Government agencies have started to tighten regulations and monitor water more closely. Many scientists argue for more comprehensive testing and stricter safety standards across the whole class of PFAS. Water utilities can invest in filtration upgrades, and households can consider point-of-use filters certified for PFAS removal if groundwater tests high. Sharing information and building pressure for transparency from manufacturers gives communities a way to push back, rather than shrugging off the risks.
The big lesson: Just because a chemical helps make life more convenient doesn’t guarantee it deserves a free pass. Keeping perfluoroheptanoic acid out of our water and bodies means a mix of science, stronger laws, and paying attention to what enters our homes. When health hangs in the balance, speaking up and demanding answers matters most.
Not Just Another Chemical on the Shelf
Walk through any chemical storage room in a research lab or manufacturing plant and it’s easy to see how easy it feels to gloss over safety rules. Boxes stack up, bottles cluster together, and sometimes it takes a careful eye to spot trouble before it happens. Chemicals like perfluoroheptanoic acid ask for another level of care. With health concerns and environmental persistence on the news, this molecule lands far from the “just toss it anywhere” category.
Direct Risks Meet Real-World Practice
Experience handling acids teaches a simple lesson: if something can corrode, cause cancer, or leak into the environment, it's best not to trust luck. Perfluoroheptanoic acid is one of those persistent perfluorinated compounds that sticks around for decades. If a bottle cracks or someone leaves the cap loose, vapors can go airborne, and even small spills stretch far past the workbench. Storing this acid in a cool, dry, and well-ventilated spot stops two big risks—fumes in the air and reactions with other chemicals. Glass containers with tightly fitted lids do the job, as the acid chews away at other materials.
Air and Moisture: Invisible, Not Harmless
Humidity sneaks into storage rooms and speeds up chemical breakdown. In my own experience, a summer thunderstorm left a storeroom muggy, which warped labels and even corroded some caps. For perfluoroheptanoic acid, moisture works as a slow fuse, setting off long-term decay or triggering reactions that taint the chemical. Keeping containers dry and protected prevents accidents that don’t show up until too late.
Separation Cuts Down on Surprises
Mixing strong acids with organic solvents, flammable materials, or metals can turn a safe storeroom into a hazard zone. Any experienced chemist will tell you to tuck acids like this one in a separate cabinet or shelf. A mistake with shelf assignments—especially if someone’s rushing—can end in fumes, fires, or worse. Good labeling matters just as much, since one missed sticker sets off a scramble during an emergency.
Regulations Don’t Just Fill a Binder
Rules around PFAS, including perfluoroheptanoic acid, step up year after year. In places like the United States and Europe, agencies keep testing and revising standards for these chemicals because they wind up contaminating water supplies. It pays to follow storage regulations not just out of fear of a fine, but because the cost of a cleanup—both environmental and financial—far outweighs the time spent keeping these bottles locked away and double-checked.
Building Better Habits
Small, steady habits keep people and places safe: double-checking lids, logging inventory, keeping spill kits handy, and reviewing storage layouts. Education and regular training make a difference. Fact sheets pinned to storage room doors beat long-forgotten lectures any day. Reporting leaks or broken containers on the spot, even if it feels like extra work, keeps small incidents from growing out of control.
Solutions That Go Beyond Compliance
Storing perfluoroheptanoic acid safely doesn’t stop with the right bottle or a locked door. Teams should run mock spill drills, rotate stock before expiration dates creep up, and look for safer substitutes whenever research or industry allows. Some places invest in automated monitoring for humidity and air quality, sending alerts straight to staff phones. These steps make it less likely for perfluoroheptanoic acid to turn from a research tool into an emergency.
Learning from Well-Known Risks
Perfluoroheptanoic acid belongs to a much-talked-about group of chemicals called PFAS—sometimes referred to as “forever chemicals.” Once they end up in soil, water, or air, they don’t leave easily. People who spend any time reading about environmental science know PFAS compounds raise alarms worldwide, both for environmental persistence and impact on human health. The scientific crowd doesn’t always agree on everything, but the major health agencies—from the EPA in the United States to the ECHA in Europe—don’t mince words about these dangers. Studies have linked exposure to a wide range of PFAS with immune issues, increased cholesterol levels, and certain cancers.
It’s About People and Planning
Dealing with perfluoroheptanoic acid in the lab or workplace demands real attention. PPE stands for personal protective equipment, but it could just as well stand for “people protect each other”—no mask or glove alone could fix lazy habits. Anyone handling this chemical benefits from gloves made with heavy-duty material, eye protection with full side-shields, and clothing that doesn’t leave skin exposed. Fresh air matters though, especially since inhalation can find its way through the routine setups of fume hoods and vents. I once saw a spill in a student lab handled with panic instead of procedure, and it stuck with me: clear directions and simple drills save headaches down the road.
Safe Storage Means Smart Arrangement
Strong containers matter. Perfluoroheptanoic acid doesn’t play well with everyday plastics. Glass or specialized high-density containers lower the risk of slow leaks or surprise reactions. Forgetting to label a bottle or letting things gather in unmarked flasks sets up bigger problems for the next shift. Storage in a separate, cool place where acids and volatile chemicals stay apart keeps risk far lower than squeezing everything onto the same crowded shelf.
Big Picture: End-Of-Life Strategies
Disposal of PFAS, including perfluoroheptanoic acid, sparks arguments all over the world. Landfills struggle to keep these chemicals out of water tables, and standard incinerators don’t burn hot enough to destroy them completely. Some companies and public agencies fund high-temperature incinerators or advanced chemical treatments, but trust in these fixes runs low if not overseen by experts. Until governments offer systems that make disposal safe and fair, asking researchers and firms to deal with toxic waste on their own just pushes problems down the line.
Building a Culture of Responsibility
Plenty of labs and plants already face strict regulations governing PFAS. Still, rules mean little if ignored, and genuine safety starts with buy-in from every staff member—not just the ones in lab coats. Simple steps, such as keeping real-time logs, running “what-if” drills, and speaking up about near-misses, make the routines stick. When new team members see veterans double-check a seal or clean up right away, that example spreads more good habits than any online training module.
Perfluoroheptanoic acid rarely features in everyday conversation, but the way we handle it affects anyone who drinks water downstream or breathes air in the same city. Common sense, backed by science and steady habits, keeps these risks from falling through the cracks.
Looking at the Structure: More Than Just Carbon and Fluorine
The name Perfluoroheptanoic Acid doesn’t spark much excitement for most folks. Peel back the scientific language, though, and there’s a story in its atoms. Its chemical formula stands as C7F15COOH. Picture a seven-carbon chain where every hydrogen has been swapped out for fluorine — plus a little carboxylic acid group tacked on at the end. The carbon backbone gives it heft and stability, while a full coat of fluorine atoms makes it nearly untouchable by heat, water, or biological processes.
To someone with a background in environmental science, this molecule prompts both interest and caution. The appeal comes with how these carbon-fluorine bonds set a world record for strength. That strength is the very reason you find perfluorinated compounds riding along in waterproof jackets, firefighting foam, and even in stain-resistant carpets. They brush off most things that come their way, a trait that chemists often celebrate in the lab.
Why Structure Matters in the Real World
With perfluoroheptanoic acid, its structure does more than keep stains off your pants. The same chemical stubbornness leads to its nickname as a “forever chemical.” Toss a molecule like this into the environment, and it lingers for decades. It doesn’t just break down or dissolve away like ordinary organic acids. In water, in soil, or in the blood of living creatures, these fluorinated acids stay put. It’s unnerving to think about how this structure ends up in places no one intended. Studies across multiple countries trace perfluorinated acids in rivers, lakes, and even human blood samples.
So, structure tells a bigger story. It decides how something performs in industry, and it shapes how people and wildlife interact with the compound even years down the line. Looking at databases from environmental health researchers, the structure shows up again and again: headlined as a concern much bigger than just a chemistry quiz answer.
Toward Better Solutions: What Can Change?
Growing up around waterways meant watching how fish populations shifted with industrial changes. News of new “invisible” pollutants wasn’t always easy to see in daily life, but the science pointed out patterns. When I first came across perfluoroheptanoic acid during my university days, it felt far from home. Fast forward a few years, and local agencies began tracing these very molecules in our tap water.
Evidence shows that long chains like C7F15COOH pass through many filtration systems. Instead of focusing all efforts on new cleanup tech, smarter choices would start with design and substitution. Research teams have begun to look for alternative compounds with fewer lasting impacts. Some have even returned to nature for inspiration, finding molecules that break down without leaving toxic residues. Limiting production and disposal of the most persistent versions can prevent repeat mistakes.
Regulation and transparency from industrial and municipal sources can push better habits. Chemists, policymakers, and communities all play a role. After all, once a forever chemical enters the chain — from soil to river to kitchen tap — the battle shifts from prevention to costly cleanup. Recognizing the permanence of something like perfluoroheptanoic acid’s structure should drive us to demand more from the next wave of chemical innovation.