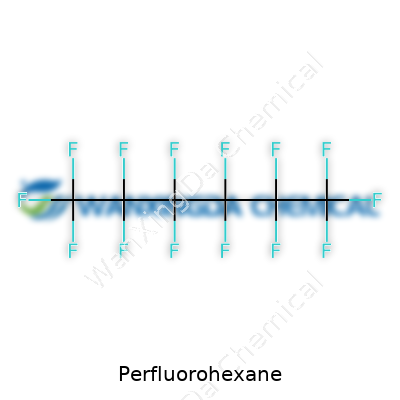
Perfluorohexane: A Closer Look at Its Development, Properties, and Challenges Ahead
Looking Back: How Perfluorohexane Emerged
Perfluorohexane’s story goes back to the mid-twentieth century, right around when chemists started exploring the potential of perfluorinated compounds. Researchers discovered that by replacing every hydrogen atom in alkanes with fluorine, they could create stable and rather inert molecules. Unlike many other organic molecules, perfluoroalkanes resists reacting with acids, bases, or even strong oxidizers. Perfluorohexane stood out because of its transparent, colorless liquid form and unique handling properties. Chemists tinkered with these molecules in search of materials that could serve in specialty electronics and medical applications. Many early studies came from industrial labs looking to move away from traditional organic solvents, which brought notable toxicity and flammability risks.
Product Overview: What Sets Perfluorohexane Apart
Perfluorohexane drew attention quickly as its combination of chemical stability and extreme hydrophobicity opened doors in several industries. It belongs to a class of perfluorocarbons that feature six fully fluorinated carbon atoms—no hydrogens left to grab onto. Because of this, it resists water, barely supports life, and shrugs off most forms of chemical attack. A bottle of the stuff, if left out and exposed, hardly changes over months. Practical folks in labs appreciate that kind of reliability, making it a go-to candidate for tasks like coolant fluids and carrier liquids in precise applications. No one likes cleaning up after a spill, and this liquid wipes clean with no trace left behind.
Physical & Chemical Properties: Unusual, Sometimes Oddly Useful
Perfluorohexane doesn’t freeze until the temperature drops far below what most lab freezers can handle, and it boils at a point well above the room temperature but below water’s boiling mark. Its extremely low surface tension means droplets bead up and roll off surfaces, never soaking in. Odorless, clear, and with a density noticeably higher than water, you can actually float small metal objects in it, which turns heads during demonstrations. Chemically, nothing short of molten sodium or very exotic agents break it down, which matters when precision and cleanliness count. This same coveted stability means perfluorohexane just doesn’t break apart easily in nature, either.
Technical Specifications and Labeling: Eyes on Containment and Purity
Labs using perfluorohexane often keep watchful eyes on purity, as any contaminants complicate both research and commercial work. High-performance liquid chromatography or gas chromatography can verify the material's grade. Storage labels call out the need for tight-sealing caps and non-reactive containers. Being a forever chemical, users don’t relish the idea of spills, so secure containment stays front of mind. Strict labeling in industrial settings aims to remind users that, despite its chemical indifference, its environmental impact commands respect and wide handling spaces are preferred for larger storage.
Preparation Method: Straightforward Chemistry with a Long-Tail Outfall
Most commercial perfluorohexane comes from the fluorination of normal hexane by elemental fluorine—often in the vapor phase and sometimes catalyzed to control extreme reactions. That process, as simple as it is on paper, needs careful control and specialized equipment. The highly reactive fluorine comes with certain dangers that can’t be overstated, and manufacturers spend heavily on containment and environmental controls. On the upside, the chemical’s inertness means even if some impurities linger in the starting material, purification is achievable with distillation, since perfluorohexane's physical properties differ so much from common organics.
Chemical Reactions and Modifications: A Tough Nut to Crack
Many chemists have tried to push perfluorohexane into new forms, but the molecule’s reluctance to react makes most attempts theoretical. Under aggressive conditions, it might exchange fluorine atoms for other halogens or radicals, producing perfluoroalkyl derivatives. Otherwise, it serves as a background solvent or is employed as a blank medium in reactions that can’t tolerate water or polar interference. Those seeking to break or stretch the molecule generally end up producing an array of toxic byproducts that complicate capture and disposal. A more promising route may come from coupling the molecule with modern fluorine-handling materials for electronics, where its unique surface properties become an asset.
Synonyms and Commercial Product Names: Navigating the Jargon
Anyone scanning safety sheets or technical catalogs might bump into names like perfluoro-n-hexane or even F-hexane. Vendors often market it alongside other perfluorinated fluids, making for a crowded shelf of similar-sounding choices. The standardized name helps research and regulatory compliance, but confusion can and does persist in commercial ordering. For researchers, knowing the chemical structure prevents grabbing a bottle with the wrong carbon chain length—mistakes happen surprisingly often in shared lab environments.
Safety and Operational Standards: Forethought Pays Off
Most routine work with perfluorohexane avoids drama, given its low volatility and chemical calmness. Dangers rise in larger operations, where spills or vapor vents might escape unnoticed. Regulatory agencies around the world urge strong ventilation, personal protective equipment, and fume hoods. In my own lab years ago, a poorly sealed bottle left a sticky film on the benchtop—luckily with no consequences, but it underscored the stuff’s persistence. Disposal rules now direct even trace amounts toward specialized incineration, since traditional waste streams barely dent it. The more people learn about PFAS impact, the more scrutiny gets applied in waste handling.
Where It Works: Diverse Industrial and Research Uses
Electronics manufacturing leaned into perfluorohexane as a cooling and cleaning agent long before the climate conversation took center stage. Its insulating properties support high-tech electronic assembly, keeping conductors safe from shorts and corrosion. Medical researchers explored its use as an oxygen carrier in synthetic blood substitutes. While early promise dimmed over concerns about long-term retention and breakdown, trials showed it could dissolve huge volumes of oxygen and carbon dioxide, something no hydrocarbon could manage. Precision optics, non-stick coatings, and niche solvents in spectroscopy fill out the main roster of users. Every application stakes out a balance between performance benefits and environmental challenges.
Research & Development: Seeking Alternatives and Safer Paths
Academic and industrial researchers spend considerable energy mapping perfluorohexane’s interactions with human health and the environment. New work explores breaking the carbon-fluorine backbone with specialized catalysts or high-temperature incinerators, aiming to reduce or reclaim forever chemicals. Some newer studies look at blending perfluorohexane with biodegradable co-solvents, trying to retain its chemical advantages while easing its environmental load. Regulatory changes, especially in the EU and United States, drive renewed investment into substitutes for all but the most demanding cases. The field has shifted; chemists now design their work to minimize reliance on perfluorinated organics. When I worked in chemical safety education, the biggest shift came when environmental profiles finally started carrying the same weight as technical performance.
Toxicity Research: A Growing List of Worries
It once looked like perfluorohexane coasted beneath the radar of most toxicological concern. Revised studies and field data have tipped that view, and for good reason. Not only does the molecule linger in soil and water, but trace exposure links to troubling health effects—bioaccumulation catches up fast in complex food webs. Major longitudinal studies flagged concerns about kidney and liver impacts in laboratory animals at sustained exposure. Regulatory bodies set increasingly strict exposure limits. Manufacturers keep revising guidelines and offering ever more rigorous worker protection training. Community groups near industrial plants have pushed for soil and water testing, only to learn that perfluorohexane persists in samples taken decades after the original source dried up.
Future Prospects: Picking a Path Forward
Looking down the road, I see a split future for perfluorohexane. Critical industries will stick with it for now, especially where no reliable alternative delivers the same mix of stability and performance. In fields like semiconductor cooling or select optical manufacturing, perfluorohexane remains as the only show in town. For broader markets, rising regulation and public pressure keep closing doors. Emerging research on safer chemical surrogates could ease the transition, but it demands time and significant investment. Cleaning up legacy waste will likely take center stage for decades. The next generation will have to build a new toolkit for handling specialty fluids, where sustainability factors as much as solvency or stability. Only by grappling with the environmental costs head-on can future chemists hope to shape responsible solutions.
What We Get from Perfluorohexane
No one brags about their knowledge of perfluorohexane at a party. It’s not exactly a household name, but this chemical finds its way into some surprising corners of our world. With roots in the family of perfluorocarbons, perfluorohexane looks perfectly at home in the lab, the hospital, and even the factory floor.
How It Shows Up in Real Life
In hospitals, specialists often reach for perfluorohexane during delicate eye surgeries. Getting a detached retina back in place can mean the difference between holding onto vision or losing it. Surgeons appreciate how this liquid’s density lets them press the retina against the eye wall, buying time for natural healing. I’ve spoken to medical professionals who view it as a marvel—easy to handle, easy to remove, and life-changing for someone worried about blindness.
Another job for this chemical pops up in the electronics world. As fans of cleanroom stories will tell you, even a speck of dust can destroy sensitive parts. Perfluorohexane doesn’t just sit idly on the shelf; technicians use it to wash away oils and grime from precision components. Its chemical stability means no unwanted side reactions, a trait prized in settings where a contaminated circuit board spells disaster.
Let’s not overlook engineers and researchers using perfluorohexane as a coolant. Certain high-speed computers and lasers run hot enough to need more than fans. The liquid offers an alternative, carrying away heat where air cooling hits its limits. Folks in research labs have told me how much they value this capability, especially when custom equipment leaves little room for standard cooling systems.
A Double-Edged Sword
There’s a flip side. As a fluorinated compound, perfluorohexane doesn’t break down easily. It hangs around in the air, water, and soil. Studies highlight its persistence, raising alarm bells among environmental scientists. Some experts have linked perfluorinated compounds to a range of health risks if exposure stretches over years, from hormone disruption to other chronic issues. Because perfluorohexane is just one member in a large chemical family, understanding its long-term effects becomes even trickier.
Regulators in places like the European Union have noticed, keeping a close eye on the use of perfluorinated substances. Policies calling for reduced emissions or safer handling stem from real concern about what these chemicals could do if they keep piling up.
Searching for Balance and Better Answers
Industries that depend on perfluorohexane can’t just drop it overnight. Its performance in surgery, cleaning, and cooling gives it a unique spot that alternatives sometimes fail to match. Investment in greener chemistries matters. Companies funding safer substitutes sometimes find tools that work just as well, but convincing every hospital or electronics plant to switch takes more than a few pilot studies.
Awareness helps too. Workers deserve clear information about safe handling practices. Proper disposal, spill prevention, and recycling programs cut down on environmental leaks. That kind of nuts-and-bolts responsibility may not make headlines, but it turns dreamy regulations into real-world progress.
Perfluorohexane shows both the ingenuity and growing pains of modern industry. The challenge, as with so many things, lies in holding onto the benefits while fewer risks spill over to future generations.
Looking Past the Slick Surface
Perfluorohexane pops up in a lot of technical discussions, but most people outside labs or manufacturing never run into it. This chemical sits in a group called perfluorocarbons, which comes loaded with debate. Some industries use it for cleaning electronics or acting as a heat-transfer agent. This keeps it tucked well out of sight for the average person, but that doesn’t mean nobody should care about what it might do.
Understanding the Health Questions
Plenty of chemicals find use in day-to-day products, but perfluorohexane is a bit of a slippery customer. A handful of animal studies hint that it can cause issues if large amounts build up. Workers involved in heavy industrial use have sometimes reported headaches, tremors, or a kind of forgetfulness when left exposed to strong concentrations for a long stretch. The vapor doesn’t play well with lungs, either — breathing lots of it brings an obvious risk. Some safety guidance treats perfluorohexane with the same respect reserved for industrial solvents, meaning gloves, goggles, and plenty of ventilation matter.
Persistent Chemicals: What Makes Perfluorohexane Noteworthy
One thing that stands out about perfluorohexane isn’t what it does right away, but how stubborn it stays in the environment. The bonds inside perfluorohexane barely budge, no matter what nature throws at them. They linger for ages—much longer than something like ethanol or acetone. Once perfluorohexane enters soil or water, it resists breaking down. That’s where people working in environmental safety start getting nervous. Nothing sticks around forever without making some kind of impact.
Regulatory Voices and Public Understandings
Nobody likes to take chances with invisible risks, especially with chemicals. Regulatory agencies in some countries have flagged related compounds as “forever chemicals,” pointing at their ability to accumulate in water, animals, and maybe even the human body over time. While not every perfluorinated compound gets the same label as toxic, scientists group perfluorohexane among chemicals demanding caution, especially in larger quantities or repeated exposure.
How It All Adds Up
I’ve looked into worksite safety and watched regulations around perfluorinated compounds grow stricter. The industries using perfluorohexane now follow tighter rules—engineered containment, spill response, medical monitoring, and limiting how much can end up in wastewater. The science says to respect this chemical’s potential for harm. Long-term exposure, even at low levels, still makes researchers uneasy, since effects can be slow to surface.
Keeping Hazards in Check
Risk shrinks when folks working with perfluorohexane wear proper protective gear and don’t let chemicals sit out in the open. Clear labeling, good ventilation, and company policies aimed at tracking exposure make a difference. The rest of us should demand transparency from companies using these chemicals. Anyone living near manufacturing should be able to learn what gets used and how water and air stay safe.
Switching to less persistent chemicals gives companies a way to lower future problems. Investing in better filtration and recovery technology can stop most releases before they leave the building. If those steps cost more upfront, they save headaches later—both for workers and the wider public.
Being upfront about the risks isn’t just about ticking regulatory boxes. It shows respect for the communities where these chemicals might end up, and helps keep trust intact between industry and everyone else outside the fence.
Understanding What Makes Perfluorohexane Unique
Anyone who spends time in a lab or reads up on specialty chemicals runs into oddball compounds, and perfluorohexane often sparks a curious look. It’s a clear liquid, easy on the eyes but stubborn against almost anything you throw at it. Unlike standard hydrocarbons, every hydrogen atom in its six-carbon backbone gets swapped for fluorine. That’s a big reason behind its chemical personality.
I’ve handled it before, and the stuff doesn’t really have a smell, which already tells you something’s up. Most organic liquids make themselves known nose-first, but not this one. Perfluorohexane’s got a boiling point around 56°C, low for a molecule its size. The density is higher than water, almost twice as much by volume. Pour some into a beaker, and droplets swing past oils and organic solvents like they're not even there. In practical terms, that non-mixing nature gives perfluorohexane real bite for use in labs where separation is tricky.
Incredible Stability—Blessing or Curse?
The fluorine atoms crowding those carbons bring impressive strength. That means the molecule barely reacts with acids or bases and won’t rust out equipment or change much under heat. Such resilience made it a darling in electronics, cleaning tech, and sometimes in medicine for blood substitutes or imaging. The flipside—nature doesn’t break it down easily. If it leaks out or escapes into the wild, it sticks around.
Perfluorohexane also refuses to dissolve in water, so rain can’t wash it away. That’s convenient on a benchtop but troubling for the environment. It doesn’t burn in air under typical conditions, so it won’t feed fires. These traits let it hide in plain sight, often after spills, slipping through soil or waterways while persisting for years.
Addressing the Environmental Impact
For years, everyone loved chemicals that wouldn’t degrade or catch fire. Now, concern grows about what happens when these same features clog up natural cycles. Scientists link perfluorohexane’s family, the perfluorocarbons, to climate issues due to their greenhouse effects. The molecule’s strength becomes a liability when it stands up to sunlight, microbes, and chemistry in the field, lasting for ages.
Addressing this means stepping up both research and regulation. Many voices in the scientific community have pushed for tighter rules on disposal and use, and companies now face mounting pressure to phase out or replace these substances. Groups tracking chemical pollution continue to flag fluorinated compounds for close scrutiny. Industry and academia are tinkering with alternatives that break down faster or pose less risk.
Toward Practical Solutions
One lesson stands clear—choices on a micro-scale can echo for decades. In my own work, caution always comes first. Containment, careful recycling, and investment in safer replacements represent three solid approaches. Expanding efforts in chemical redesign, seeking compounds with similar performance but greater degradability, makes sense both for business and for the wider world.
Perfluorohexane’s physical and chemical strengths serve some unique needs, but these same qualities deserve respect, not just admiration. The challenge falls to all hands—chemists, regulators, policymakers, and everyday workers—to treat these substances responsibly and look beyond short-term convenience.
Real-World Wisdom on Chemical Storage
Walking into a lab stocked with bottles labeled “perfluorohexane” can trigger a second look, especially if you’ve seen stories of careless chemical handling. This clear, colorless liquid often pops up in high-tech fields, including electronics and ophthalmology. It hardly has a smell, doesn’t burn easily, and stands out for its ability to stay stable under a lot of conditions. Still, just because a chemical doesn’t explode at the drop of a hat doesn’t make it a harmless addition to the shelf.
Health and Environmental Factors that Matter
A lot of people overlook how tricky storage can get, even with a substance as seemingly non-threatening as perfluorohexane. Breathed in enough, its vapors irritate eyes and lungs, and there’s growing talk about how these “forever chemicals” build up in nature over time. The science points out that while perfluorohexane doesn’t break down easily, it finds its way into rivers and air, clinging to environments and organisms for years.
Smart Storage Reduces Risk
A key move is to pick storage spots that keep the substance cool and shaded, away from sunlight and any heat sources. Sealing containers tight goes a long way; a leaky jar undoes a lot of good work and risks vapor slipping into the air. Even in workplaces where ventilation already gets attention, extra steps such as chemical fume hoods boost protection for those handling the liquid. Glass or high-grade plastic containers give the best results for avoiding slips, leaks, or chemical reactions.
Personal Experience from the Lab
Having worked in labs that juggle a mix of chemicals, I’ve seen what can happen when storage shortcuts are taken. Once, a poorly sealed container released a hint of vapor. The smell wasn't strong, but everyone nearby started coughing—an easy lesson in why tight lids and vented cabinets matter. The truth is, taking five extra seconds to check those caps saves a world of stress later. Eye protection and gloves became less about following checklists and more about dodging the “what ifs.”
Steps Forward for Safer Handling
Nobody wants to fill out incident forms after a spill or exposure. Simple habits make the biggest difference—labeling every bottle clearly, logging chemical use, and training new staff until they can find personal protective gear with their eyes closed. In my experience, asking questions turns into the best safety net. If a process needs reviewing, or a safety step feels off, speaking up beats keeping quiet every time.
Government guidelines set down rules for a reason but filing them away after an annual review won’t help much day-to-day. Building a safety culture means keeping up-to-date and looking two steps ahead. Teams that run regular reviews catch problems before they escalate. It’s not enough to count on one person to double-check—shared responsibility spreads out the risk and raises standards across the whole workplace.
Looking Toward Safer Chemistry
The drive for better handling and storage of perfluorohexane speaks to a larger issue: chemicals have consequences that reach beyond the lab. Think long-term, ask questions, and take small actions every day. By staying alert to both health and environmental impacts, anyone working with perfluorohexane can protect people and the planet—one container at a time.
What Perfluorohexane Means in the Real World
Perfluorohexane doesn’t jump off store shelves at a local pharmacy or pop up in the cleaning aisle. You find it in labs, research settings, and certain industrial applications. Growing up around a family of chemists, I remember the hush those bottles inspired—clear liquid, unfamiliar name, demanding both respect and paperwork. It’s a specialty chemical, not a household supply, and that shapes where you can buy it and how much you’ll pay.
Real Sources, Real Challenges
For anyone outside the laboratory or R&D field, searching for this solvent quickly shows it’s reserved for places that work with chemical distribution at scale. Reliable purchases happen through industrial suppliers and reputable chemical distributors. Companies like Sigma-Aldrich, Thermo Fisher Scientific, and TCI America figure prominently in these circles. Each of these firms vets its buyers, and anyone looking to purchase will likely face requests for business credentials, purpose of use, and proof they can safely handle the substance. The days of easy, anonymous chemical purchases have come and gone, driven mainly by safety and regulatory requirements.
Cost—Why Prices Are All Over the Map
The price for a bottle or drum of perfluorohexane rarely matches what you find for household chemicals. In 2024, small research quantities might range from about $200 to $500 for a half-liter, sometimes more depending on purity and grade. Larger volumes cost less per unit, but suppliers tend to pack shipping and hazardous material fees onto every order. Anyone budgeting for a scientific project or proprietary process quickly learns that hazardous material surcharges stack up, and the price reflects more than just the product. It covers handling, shipping, and the legal framework around chemicals with environmental impact.
Global supply chain issues and increased demand from specialty electronics and medical applications have nudged prices higher. Some grades cost even more, particularly where ultra-high purity makes a difference in end use, such as semiconductor or bio-imaging work. Even if the sticker price looks manageable, a chemist’s supply room manager faces a stack of extra charges—anyone working on a tight research budget feels this crunch firsthand.
Why Restrictions Make Sense
Perfluorohexane, like other perfluorinated compounds, raises environmental and health concerns. Studies show these chemicals persist in the environment, and scientists have linked them to health risks from long-term exposure. That matters to communities living near chemical manufacturing or disposal sites. Laws tightened up after years of studying these effects. Distributors respond with strict buyer checks and careful recordkeeping. The result is extra friction for legitimate research but a safer marketplace all around.
Better Paths Forward
Transparency in sourcing and clear communication from suppliers help scientists and innovators keep their work above board. More suppliers now provide documentation about product origin, impurity levels, and environmental handling advice. Customers should expect vendors to answer questions about regulatory compliance and waste disposal processes. This level of openness boosts trust and deflates the risks posed by illegal or substandard chemical supplies.
Researchers can push for sustainable alternatives or methods to recycle and recover chemicals like perfluorohexane. Universities and companies that openly share their chemical handling protocols—rather than keeping them under wraps—offer valuable roadmaps. Sharing knowledge about how to safely, legally, and ethically bring specialty chemicals into the lab ultimately serves not just projects but the wider community.