Perfluorohexanesulfonic Acid: Its Journey, Impact, and Where Science Goes Next
The Early Days and Shifting Awareness
The name Perfluorohexanesulfonic acid (PFHxS) has surfaced in environmental circles for longer than most folks realize. It came up as a commercial surfactant not long after the bigger, more notorious PFOS made headlines. Early on, companies sought out these “long-chain” perfluorinated acids for their ability to repel water, stains, and oil across all kinds of household and industrial products. I remember seeing the boom of stain-resist textiles and firefighting foams back in the 80s and 90s; no one gave much thought to what happened to those chemicals after washing or disposal. Regulatory scrutiny came later. At first, PFHxS often flew under the radar as the industry paid more attention to its cousins. Now, research tells a different story, showing PFHxS lingers just as stubbornly as any PFAS class chemical and deserves every bit of the attention.
Why PFHxS Finds Its Way Everywhere
PFHxS is a persistent molecule with a structure built for staying power: a fully fluorinated six-carbon chain capped with a sulfonic acid. In the world of chemical stability, those carbon-fluorine bonds take the prize. Firefighting foams relied on this stability. So did water-repellent sprays, polishes, mist suppressants in plating, and sometimes even older textile treatments. The stuff is colorless and resists breaking down under sunlight or heat. If it gets into water or soil, you can’t count on nature to solve the problem. The properties that made it popular—like resistance to acids, bases, and heating—now pose a persistent contamination headache.
Chemical Profile: Tough Bonds, Little Escape
It has a mighty molecular structure that translates to a solid reputation for chemical toughness. PFHxS molecules don’t linger on the surface; instead, they make their way into groundwater and move through soil with ease. Their low volatility means once the chemical is down, it doesn’t just evaporate. Take a sample of well water near an older airbase, and odds are, you’ll find traces. The acid form dissolves in water and slides along with runoff, carrying the same threat that docks longer carbon chain relatives as “forever chemicals.” While the molecule seems simple on paper, every fluorine atom contributes to a tenacity that resists anything most municipal treatment can throw at it.
Technical Standards and Labeling in the Real World
Labeling rules changed slowly. In earlier years, PFHxS would turn up labeled as a byproduct or minor listed ingredient on safety data sheets, if at all. Now, recognition of its stubborn behavior has led governments to lower the thresholds that trigger reporting or restrictions. Testing sees variation worldwide, with regions disagreeing on what counts as “safe.” Folks dealing with legacy contamination don’t care much about paperwork; safety comes down to real testing and clear boundaries about usage and disposal. Incidents have shown that technical jargon on hazard labels rarely prepares communities for the actual challenges of contamination.
Production and Chemical Tinkering
Companies made PFHxS either by directly sulfonating perfluorohexane or by electrochemical fluorination of suitable precursors. Both paths create a byproduct stew that often includes related PFAS compounds. The process leaves little room for error, and persistent waste streams often travel outside plant walls. The chemical likes to hold onto its sulfonic group, so researchers trying for breakdown must bring in tough reagents or specialized equipment—this stubbornness means that once it leaves the factory, the chemical doesn’t convert easily to safer forms.
What’s In a Name?
Most folks know it as PFHxS, but look at global inventories and you’ll see alternative labels: Perfluorohexylsulfonic acid, Hexafluoropropylene oxide sulfonate, or trade names obscured by corporate branding. This variety in naming led to confusion in early regulations. Back in the days of looser scrutiny, manufacturers didn’t always make distinctions clear for downstream users. Researchers now unite behind standardized terms, but old records still hold legacy synonyms that fill up environmental reports.
Safety and Operational Practice: Lessons Learned
Experience teaches that PFHxS doesn’t care much about factory gates or facility checklists. Areas with firefighting foam use can show contamination in surface water, groundwater, and even dust. The usual protective gear—gloves, goggles, fume hoods—is not enough to prevent environmental escape. Once used, PFHxS trickles out in wastewater, sometimes travelling far past original site boundaries. Attempts at incineration or advanced filtration bring high financial and technical challenges. Workers remember the days of washing down equipment with little more than a cursory rinse, not realizing those rinse waters might one day lead to fish consumption advisories and hefty remediation bills.
Where Does It End Up?
PFHxS rarely stays where it lands. It finds its way into groundwater and even into animals in remote locations. Testing shows tap water, river sediment, and even the blood of some wildlife have traces. I’ve spoken with water managers in smaller towns who struggle to balance the cost of removing PFHxS with tight budget lines—point-of-use filters for houses, municipal upgrades, and even bottled water in extreme cases. Contaminated soil doesn’t respond well to many clean-up attempts, and the molecules outpace the progress of most bioremediation strategies.
Research Community on Edge
Studies coming from universities and government labs keep revealing just how far PFHxS has traveled. Every research paper draws a new line of concern: effects on thyroid hormones, immune disruptions, and hints at connections to high cholesterol in exposed populations. Testing grows increasingly sensitive, so limits that seemed reasonable one decade start looking risky the next. The debate isn’t just about numbers; there’s real anxiety among parents, doctors, and folks near contamination zones who ask, “How do we fix this?” Answers grow slowly, as every year brings new health findings.
The Challenge of Toxicity and Risk
Results from animal studies give reason to pause. Rats and mice exposed to PFHxS show changes in liver function and hormone levels, and regulators now pay much more attention to even low-level, long-term exposure. This isn’t a chemical anyone wants to gamble with, especially since data hints that children and unborn infants might be especially sensitive during certain developmental stages. While studies still hunt for definitive links to human illness, caution wins out in most risk assessments today. No level of routine exposure should be ignored, and the call for precaution rings louder than industry assurances of safety at “trace” amounts.
Searching for Solutions
Solving the PFHxS challenge takes more than technical upgrades at water treatment plants. Some hope rests on aggressive filtration like activated carbon and advanced resins, but these fixes cost big money and still leave questions about safe disposal. Incineration at high temperature may destroy PFHxS, yet few facilities can run such processes reliably, and communities bristle at the prospect of shipping toxic waste long distances. The biggest lesson is to stop the cycle upstream—phasing out uses of PFHxS, pushing for substitutes wherever possible, and tracking waste with ruthless efficiency. There’s no shortcut around public disclosure or community involvement. Success stories will come from shared effort and clear rules, not from after-the-fact cleanups.
The Path Ahead: Regulation, Innovation, Accountability
Regulators grow less patient with forever chemicals each year. Europe has moved to restrict uses, and international treaties edge closer to classifying all forms of PFHxS as substances of global concern. Industry pivots slowly, eyeing alternatives but wary of new costs. Innovation must come from both big chemical firms and scrappy startups—greener surfactants, better containment, smarter treatment methods. Accountability needs to mean something: both for past pollution and future stewardship. Communities will not quietly accept scientific uncertainty or regulatory hesitation, not when kids face potential health risks for someone else’s industrial shortcuts.
Hope in Hard Questions
In talking to folks affected by PFHxS pollution—farmers with fouled wells, parents in towns with boil-water notices, school admins unsure if sports fields carry risks—the one thing everyone demands is straight answers. PFHxS forces society to reckon with what trade-offs are acceptable and how far to go in preventing chemical legacy. New science brings tools to map, trace, and maybe even break down these persistent molecules. The problem won’t disappear overnight, but admitting the scale and not shying away from tough policy or investment choices might just keep history from repeating again with the next “miracle” compound. I see genuine hope in the way voices across science, industry, and community have forced this hidden threat into the light. That pressure will keep the story moving—and drive the real fixes that future generations deserve.
The Backbone of Water and Stain Repellency
Perfluorohexanesulfonic acid, or PFHxS, has found its way into plenty of everyday products, mostly as a workhorse behind the scenes where resistance to oil, grease, and water matters. Anyone who’s used a raincoat that actually works or seen a carpet that shrugs off red wine owes a little credit to chemicals in this family. PFHxS enables surfaces to block liquids, helping fabrics last through mud and coffee spills. In older consumer products, this sulfonic acid played a major role in water and stain repellent coatings for textiles, upholstery, and paper-based packaging.
Firefighting Foam: A Double-Edged Sword
Firefighting, especially at airports and military bases, became much more effective and less risky for responders with the help of PFHxS-based foams. These foams suppress intense fuel fires, sticking around to form a barrier between fuel and oxygen. It’s no exaggeration to say that this type of chemistry saved lives. At the same time, there’s no sidestepping the problem that PFHxS molecules don’t break down easily. Once sprayed, they can show up in groundwater for decades. International organizations and health experts have kept an eye on water near sites where these foams saw heavy use. Some countries have started phasing out this application, pushing industry to look for alternatives that won’t linger in the environment.
Microchips and Photolithography: Precision at a Price
In electronics, making computer chips calls for substances that handle extreme conditions. PFHxS helps etching solutions clean semiconductor wafers by adding surface tension control and acting as a wetting agent. When factories push technology to etch smaller circuits, they turn to materials that keep liquid solutions even and controlled. Small-scale effects on the quality of smartphones, laptops, and cars trace back to these tricky wet chemistry processes. The flipside comes in how the industry deals with used chemicals. Proper disposal and recycling remain a challenge, and that’s a topic every hardware customer would do well to think about in an era of fast technology upgrades.
Challenges in Phasing Out Persistent Chemicals
There’s no doubt that PFHxS got picked for jobs where little else could handle the extremes. But with so many studies connecting this family of substances to persistent pollution and potential health effects, attention shifted from “how well does it work?” to “what comes after?” Some cities banned older versions in local water treatment, and vigilance about runoff at industrial sites has grown. Legal limits on allowable levels in water keep changing, reflecting ongoing research. I’ve watched debates about cleaning up contaminated ground water, and it’s rarely straightforward. Newer water filtration technologies, like activated carbon and ion exchange, show promise, though costly and sometimes slow to roll out.
Looking Toward Responsible Chemistry
In my view, real progress in chemical policy comes not just from banning one substance, but from designing replacements with fewer long-term surprises. Some labs are building materials inspired by natural repellency, while industrial groups invest in better containment. The lesson? Clever design on the front end can save headaches and dollars down the road. By pushing for transparency and backing research in safer chemistry, customers and policymakers can drive the industries that depend on specialized chemicals to act with an eye on both performance and the planet’s balance sheet.
By Someone Who Watches the Details
There’s an alphabet soup of chemicals floating around modern life nobody ever really talks about until something goes wrong. Perfluorohexanesulfonic acid—PFHxS, for short—lurks among them. You probably never heard of it outside a chemistry class, yet it can drift into waterways and bloodstreams after being used in firefighting foam, stain repellents, or metal plating. Those uses brought convenience and durability, but comfort tends to come with a cost people pay much later.
PFHxS belongs to the wider PFAS family, sometimes called “forever chemicals” because they stick around in soil, water, and living tissue long after the factory whistle blows. They never really break down. I spent years reporting on water quality, and the sense of dread among rural communities hit with contaminated wells tells a clear story: nobody wants a chemical in their tap that never leaves.
Scientific studies have raised serious questions. Researchers studying blood samples from places where PFHxS turned up found it accumulates in the body, even more so than some of its PFAS relatives. Higher levels have linked to changes in cholesterol and possible effects on thyroid hormones. In kids, abnormal exposure brings up worries about immune system development. Some governments already twitch when they see the numbers rising in community water tests.
What worries people most isn’t a single study, but a pattern. The United States’ Agency for Toxic Substances and Disease Registry highlighted potential developmental, thyroid, liver, and reproductive changes from PFHxS exposure. Even so, nobody lined up a direct cause-and-effect relationship. Animal testing fills in a few blanks, but the real world gets messy. In public health, if the warning lights flicker, people want answers, not a shrug.
Once these chemicals hit the wider world—spilled along highways after truck accidents, washed down drains after a training drill, buried in landfills—they don’t stay put. High levels have shown up where training facilities used fire-fighting foam, where airports managed runway fires, and near manufacturing plants. I’ve met activists in Michigan digging up old soil samples, worried sick about school playgrounds and rivers. There’s no easy way to mop up a chemical built for survival; activated carbon filters and other fixes prove expensive and incomplete.
The environmental risk keeps growing because PFHxS works its way through food chains. Fish caught downstream of contaminated areas sometimes pack a chemical punch. If you eat local fish or rely on well water, you want to know what hides in your food and drink. Regulatory agencies in Europe and Australia started limiting PFHxS already, sometimes forcing companies to look for alternatives. The US has begun eyeing stricter exposure guidelines and asking for better testing.
This problem calls for sharper oversight in how chemicals make it to market, more thorough testing before use, and regular cleanup efforts in places with known contamination. People can push for answers—calling local officials to set stricter water standards, supporting science that tracks long-term health outcomes, and choosing products with safer alternatives. The cleanup crews need better tools, and communities deserve honest updates rather than technical jargon. Transparency and solid science stay more valuable than indifference, every single time.
What Real-World Experience Teaches about Chemical Packaging
Anyone who's spent some time in a laboratory or a manufacturing plant knows that handling chemicals demands practicality, not just on paper but in the real, messy day-to-day work. Perfluorohexanesulfonic acid, a pretty specialized substance, illustrates that better than most. Here’s a chemical that’s often used for its resistance to heat and stubborn chemical breakdown, but finding it in the right amount and package makes a bigger difference than most folks realize.
Common Packaging Sizes: Why They Exist Like This
Across the industry, perfluorohexanesulfonic acid rarely lands on a loading dock in large drums or tankers. The risks tied to its environmental persistence and toxicity steer suppliers toward smaller amounts. Most research or regulatory databases, and plenty of direct suppliers, list this acid in packaging no bigger than a liter. It’s sometimes sold in 1 milliliter glass ampoules, 10 milliliter vials, or up to 100 milliliter plastic or glass bottles. There’s good reason for that: it’s tough to justify hauling around or disposing of excess when the substance itself draws regulatory attention worldwide.
The limited sizes keep exposures in check. In my own experience managing a chemical storeroom, requests for larger volumes almost always triggered extra paperwork and safety routines. Smaller units also help cut down on waste. That’s important with any chemical that sticks around in the environment as long as this family of compounds can. Nobody wants to be left with an awkward half-bottle in a storage locker, especially once new regulations hit.
Why Community and Environment Shape Packaging Choices
Perfluorohexanesulfonic acid is part of the PFAS family, so the debate about its handling spills outside factory walls. Public interest and regulations on PFAS waste have grown. For example, the European Chemical Agency and the US EPA both list these substances among priority pollutants. As concerns about water contamination mount, small packaging helps limit potential spills and leakage. It’s not just about meeting a safety checklist; it’s about trust. When communities near research institutions or production sites see smaller, tamper-proof containers, the message is clear: reducing risk matters.
In practice, these smaller bottles also make for much easier accounting. The rules push companies and labs to track every milliliter that comes in and goes out. Stepping through those records myself, I saw how small packaging cuts the margin for error and simplifies compliance. Larger volumes often brought with them headaches—hazmat transport, extra permits, and a timeline filled with delays.
Supporting Better Outcomes through Smarter Packaging
Offering perfluorohexanesulfonic acid only in tightly controlled, smaller bottles isn’t just a nod to following rules. It reflects lessons learned from years of chemical mishaps and mounting environmental concerns. To keep progress moving, suppliers can make even more use of returnable, sealable containers. That would reduce waste and help with safe collection once the acid’s use runs its course.
The industry doesn’t land on these packaging decisions in a vacuum—they come from trial and error, scrutiny by regulators, and a sense of responsibility. Practical experience and real community impact shape how stuff gets packaged, stored, and transported. It makes a difference in how safe people feel, both in the lab and down the street from a facility that works with these persistent compounds. Safety, environmental respect, and efficient handling all get tied together through the simple act of bottling up this stubborn acid in small, manageable portions.
Looking the Real Hazards in the Eye
Folks who work around chemicals often talk about respecting what they handle. Perfluorohexanesulfonic acid, a mouthful even by chemist standards, deserves the kind of respect usually reserved for wild animals. Its danger can’t be brushed aside just because lab protocols exist on paper. Skin contact causes burns. Eyes exposed to a squirt might leave lasting damage. When you breathe it, you don’t just sniff a faint smell — you invite in a chemical that stays in your body for years, quietly stacking up. This C6 PFAS doesn’t behave like vinegar or bleach: spills lead to a mess that water can’t wash away.
Why Regular Storage Rules Don’t Cut It
Most shops and even universities shy away from perfluorohexanesulfonic acid because its reputation precedes it. Stashing a bottle on a regular shelf, with the hope that nothing leaks, stands as hopeful thinking at best. This acid needs containers that won’t react: high-density polyethylene, PTFE, or certain grades of glass. Rusty metal shelves or flimsy bins lead to disaster. I learned that lesson watching a co-worker scramble to contain what looked like nothing more than a dribble, only to destroy an expensive bench and ruin the air in the room.
People might think cold, dark storage protects everything, but this acid calls for more than just a dim cupboard. Strong acids and incompatible chemicals belong nowhere nearby. Stack it with oxidizers or flammable liquids, and the results go beyond a ruined afternoon. If the air gets humid or a lid doesn’t stay tight, the fumes can find metal to corrode or react with glues and seals most people forget to check. Every ounce of sloppiness with this stuff turns into hours of cleaning and risky exposure.
Training and PPE: Skipping Corners Isn’t Worth It
I’ve seen rushed teams treat personal protective equipment as a checkbox to keep administrators happy. Perfluorohexanesulfonic acid punishes shortcuts like no other: no gloves, splash on skin. No goggles, straight to the eye. Open bottle with bare arms, now you deal with red blotches and pain by lunch. Nitrile gloves barely cut it, while thicker neoprene or butyl rubber fares better for unexpected splashes. Full face shields shouldn’t feel like paranoia, but common sense. Aprons don’t just keep uniforms clean, they stop a small spill from sending someone to urgent care.
Waste: Pretending It Vanishes Doesn’t Work
Plenty of places sink their chemical waste, hoping that out of sight means out of mind. This acid behaves differently. Dumping it goes straight against environmental responsibility and often against the law. Even tiny amounts can slip through water systems and come right back in fish and drinking water. If the phrase “forever chemicals” feels overused, try explaining to a parent why this stuff hangs on in the environment longer than we’ve had cell phones. It’s time teams step up, training on waste labeling, segregated storage, and properly sealed disposal containers — not just looking to pass an inspection, but to keep it out of the planet’s water and soil.
Looking Beyond Rulebooks
There’s value in turning every incident, near-miss, and spill into a learning moment instead of shrugging and pushing a mop. Some of the best changes come from people who work with these chemicals every day and know which shelves bow, which fridges leak, and how quickly “no big deal” turns into trouble. Real safety grows when those voices hit the morning meetings, shaping policy from the ground up. It’s on everyone in the loop — lab hands, supervisors, waste haulers — to speak up before the lid comes off.
The Nuts and Bolts of Perfluorohexanesulfonic Acid
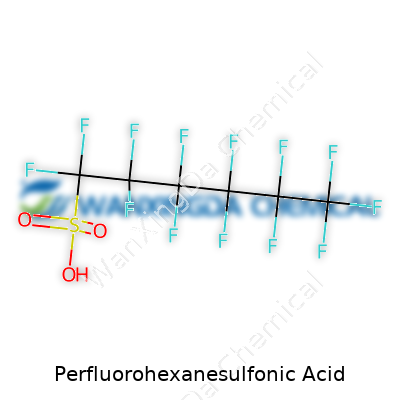
Perfluorohexanesulfonic acid, known in chemical circles as PFHxS, isn’t on the lips of most people, but this chemical isn’t obscure among those who study environmental contamination. Digging into the basic makeup, this molecule carries the formula C6HF13O3S. At its core, you’ve got a chain of six fully fluorinated carbon atoms—no room for hydrogen except at the tail, hooked onto a sulfonic acid group. Think of it as a six-link carbon backbone, each link wrapped, locked, and shielded by fluorine, making it one of the most stubborn substances nature deals with. The sulfonic acid part, SO3H, bolts onto the end. The structure looks simple on paper: F13C6-SO3H. In practice, this simplicity spells trouble for the environment.
Unpacking the Importance: Experience on the Front Lines
I worked with municipal water testing for some years. PFHxS popped up again and again in water results, long after it was phased out by many manufacturers. The molecule refuses to break down, even after years in soil and water. Once it leaches into groundwater, it circles back in drinking taps, long-haul fish, and, eventually, people. The problem links to the strongly polar sulfonic acid group at one end and the greasy, water-repellent tail at the other. Neither nature nor most treatment processes have good tricks for snipping up this kind of structure.
What Makes PFHxS a Standout Chemical Challenge?
This chemical resists heat, acids, bases, biological attack—pretty much every standard environmental process. That super-strong carbon-fluorine bond keeps standard chemistry at bay. The only hydrogen in the formula sits at the acid group, which means the rest of the molecule shrugs off just about everything. The properties that made PFHxS so attractive for firefighting foams, textile coatings, and stain-proof carpets all stem from this tough chemistry.
Beyond the Lab: Why Structure Matters in the Real World
Learning the formula is just a start. In my work sampling water, PFHxS often shows up alongside its higher-profile cousins like PFOS and PFOA. Regulators now realize focusing only on a handful of these chemicals misses half the threat. The pattern repeats: compounds with this same stubborn carbon-fluorine backbone, acid group at the end, and faint industrial smell, marching straight through wastewater plants and into bodies. Blood samples confirm that even communities far from factories have traces of PFHxS. It sticks around for years in the environment—and in people.
Chasing Solutions, Not Just Clean Numbers
Fixing the PFHxS problem takes real-world action, not just tweaks in glass-walled labs. Water treatment partners now test new absorbents and advanced oxidation methods. Instead of hoping conventional filters keep up, some countries invest in source tracking, aiming for upstream bans and cleaner production. Community-level monitoring matters, too. The public doesn’t get much say in molecular diagrams, but everyone deserves to know what’s in their water and food. Scientists push for global agreements to stop the chemical at the source, since cleanup only works so well after it’s spread everywhere.
Trust in Chemistry—and in Vigilance
From the first test vial to the latest international treaty, this acid shows why knowing the shape and formula of a molecule isn’t just academic. That formula points straight to why the world grapples with PFHxS today. It’s a hazard built into the blueprint. The structure of perfluorohexanesulfonic acid gives it power, but also a dangerous staying power in the world outside the lab. Real progress hinges on understanding the chemistry, tracking the spread, and supporting strong decisions at every level.