Perfluorohexanoic Acid: A Clear-Eyed Look at Its Path, Purpose, and Challenges
The Story So Far: Historical Development
Perfluorohexanoic acid, which some folks in the chemical business call PFHxA, shows up in conversations not because it’s a household product, but because of what it shares with a class of chemicals known as PFAS. These fluorochemicals have been around since the early twentieth century and really took off after the Second World War, when industry got serious about making tough, non-stick, and grease-resistant materials. Chemists looking for stable compounds made with carbon-fluorine bonds landed on longer-chain acids like PFOA and PFOS, which by now are notorious for their environmental persistence and impact on health. Over time, industry bets shifted, trying shorter chains, like PFHxA, as a workaround for toxicological concerns. But, as often happens, trading one compound for another doesn’t always sidestep the bigger questions.
What Is PFHxA, Exactly?
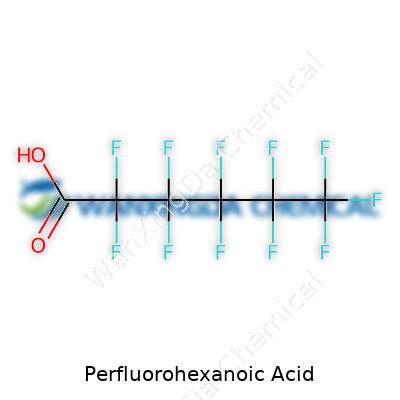
PFHxA is a perfluorinated carboxylic acid, with a six-carbon backbone fully shielded by fluorine atoms. This molecular structure resists breakdown by heat, sunlight, acids, or bases. Unlike more familiar household acids, PFHxA doesn’t react much with its surroundings. Chemists have pursued this kind of stability for decades, fascinated by the things you can do with a molecule that just won’t wear out. It gets used for surface treatments and coatings—think stain-guarded carpets, water repellent sprays, and greaseproof food containers.
The Nuts and Bolts: Physical & Chemical Properties
PFHxA looks like a clear or white solid at room temperature. It dissolves slowly in water, more so than its bigger cousins with longer carbon chains—a detail that matters for understanding how it moves in the environment. Its melting and boiling points push higher than most household acids, which signals how tough the carbon-fluorine chain is. In labs, you see it described as both lipophobic and hydrophobic, which basically means it doesn’t want to mix with fats or water. This stubbornness means PFHxA lingers in streams and soils where it gets dumped or seeps out over time, posing footprint-management problems for a range of industries.
Technical Specs, Labels, and the Language Around It
Terms get tossed around with PFHxA: hexanoic acid, perfluoro-n-hexanoic acid, and even sometimes as part of trade names specific to the application. Regulatory agencies want clear labeling, especially in places where PFHxA rides along with other fluorinated compounds in sprays, paints, or consumer goods. Specs include purity grades and thresholds for other PFAS contaminants. As countries get more watchful about perfluoroalkyl chemicals, more products carry traceability and disclosure requirements. For consumers, it is hard to decipher which "fluorinated" label means a long-standing chemical legacy and which one comes with promises of rapid breakdown or lower toxicity.
How Industry Makes It: Preparation Method
Factories get PFHxA mainly by breaking down larger perfluorinated compounds or by direct synthesis from smaller building blocks—often by electrochemical fluorination or telomerization. Once, companies leaned heavily on processes that spit out a mix of short and long chains. Now, as regulation tightens, operations try to pull out only the desired product, cutting down on unwanted PFOS or PFOA leftovers. Waste controls have improved, but there’s no magic bullet: each batch still leaves a mark somewhere along the supply line. Having visited facilities where fluorochemicals are made, I’ve watched teams juggle the demands of safety and efficiency. Every extra dollar spent capturing waste or tightening purity can mean the difference between routine operations and a regulatory headache.
Chemical Reactions and Modifications
PFHxA itself shrugs off most chemical attacks—by design, of course—so not much happens if you mix it with acids or bases under regular conditions. To modify it for industry use, chemists usually change the terminal carboxylic acid group. These tweaks make it stick better to substrates or blend with plastics, which helps it perform in water repellency or as an emulsifier. Downstream, the challenge becomes reversing that stability if you want to clean up contamination. This means researchers spend a lot of time thinking about photochemical degradation, advanced oxidation, or biotransformation methods that can chop PFHxA into safer pieces.
Synonyms and Product Names
PFHxA goes by a slew of synonyms beyond perfluorohexanoic acid or hexanoic acid, including hexafluorocaproic acid and some trade-specific codes. This naming shuffle does little to clear up confusion for non-scientists, since most people lack a sense of which chemicals end up where. Regulators have begun pushing companies to untangle this mess by forcing transparent ingredient disclosures, which can only help when it comes to tracking what gets poured into rivers or dumped into soils.
Safety and How Operations Handle It
Workers dealing with PFHxA follow strict rules to avoid spills, inhalation, or accidental releases. While PFHxA acts less bioaccumulative than some of its longer-chained relatives, it can still escape into water and persist for years. Companies responding to scrutiny over PFAS build-in redundant systems for air and water treatment and make it a habit to monitor soil and groundwater nearby. Protective clothing, closed vessel systems, and training routines have all improved, though nothing substitutes for the constant vigilance bred by risk.
Where It Shows Up: Application Areas
The draw of PFHxA comes from its ability to impart resistance to stains, grease, and water. It gets worked into food packaging, textiles, carpets, and sometimes even fire-fighting foams. As concern over the more infamous perfluorinated compounds grew, manufacturers slid PFHxA in as a drop-in replacement, thinking a shorter chain would bring a lighter environmental load. Still, no material is immune to scrutiny, and regulatory trends make it all but certain that uses will be more tightly controlled than ever before.
Research and Development: New Directions
Scientists stay busy probing the smallest details of PFHxA’s fate in the body and in nature. More labs take up high-resolution mass spectrometry, which helps measure even minuscule amounts in blood or drinking water. My own work touches on environmental sampling, where the hunt for PFHxA turns up surprises: it travels farther and lasts longer than early studies guessed. Academic and government labs both turn to machine learning models to predict toxicity and find ways to break the carbon-fluorine bond that stymies conventional cleanup.
Toxicity and the Human Dimension
Back in the early days, little data surfaced on what PFHxA did to people or wildlife. Now, the science has caught up. Animal studies report low-level changes in organs and development at high doses, sparking debate about what counts as "safe" for long-term water exposures. Human studies lag, limited by the practical challenges of measuring a molecule that’s everywhere and nowhere all at once. Community groups near contaminated sites keep up the pressure for more testing and transparency, which pushes regulatory agencies to fund more expansive health surveys. One thing’s clear: what science doesn’t know keeps local families up at night.
Where Next? Future Prospects
Industry stands at a crossroads, juggling durability against responsibility. PFHxA’s promise—non-stick and water-resistant coatings—faces growing pushback from environmentalists, regulators, and health professionals. By now, a shift looks inevitable: fewer uses, stronger controls, and growing research into alternatives and cleanup technologies. Some companies pivot to biodegradable options, but questions persist on how well these work in real-world settings. In the end, the story comes down to balancing technology’s reach with public trust. Sustainable materials can’t just work in the lab; they have to fit into local communities and ecosystems without leaving scars for generations. Watching these trends, one lesson rings loudest: the path forward means staying open to revision, for technologies and for the standards we set as a society.
The Many Faces of Perfluorohexanoic Acid
Whenever I hear folks talking about perfluorohexanoic acid (PFHxA), the conversation usually tilts toward two things: how it’s all over modern manufacturing, and how it’s showing up where nobody wants it. PFHxA belongs to the family of PFAS, those long-lasting chemicals people call “forever” for a good reason. In my own dive into this issue, I learned that companies use PFHxA mostly for making goods water, stain, and dirt-resistant. That includes all kinds of products: carpets that shrug off juice stains, jackets that keep you dry in a rainstorm, and even food wrappers that stop grease from soaking through. The compound’s chemical toughness keeps it from breaking down, which is handy when you don’t want things to stain — but not so handy when thinking about where these chemicals end up.
The Risks Spread Beyond the Factory
PFHxA doesn’t always stay put after the sticker label goes on at the factory. Washing those water-resistant jackets or grime-proof carpets sheds some of the chemical, sending it into laundry water. Treatment plants aren’t set up to pull it out, so PFHxA can travel into rivers and deeper into the ground, showing up in water sources. Way back, I remember folks worrying mostly about PFOA and PFOS, older PFAS types, because of clear links to health issues including cancer. As those got phased out, PFHxA showed up as a “safer” stand-in, but there’s little long-term research on what it does to people. Recent studies suggest it doesn’t build up in our bodies as quickly as other PFAS, but once it’s out there, it’s tough to get rid of. The Environmental Protection Agency tracks it closely now, finding it in rivers, drinking water, and even rain.
Time to Pay Attention
Most people I talk to agree that consumers deserve to know when these chemicals pop up in everyday products. A problem like PFHxA doesn’t stay trapped in a spreadsheet at a manufacturing plant. It spreads into the air, water, and soils — and eventually into us. Even if companies claim “better safety” compared to older PFAS, communities downstream or downwind of factories can’t just trust a promise. In places where the water tested high for PFHxA, families had to buy bottled water just to make dinner. These aren’t issues anyone expects to face in their own kitchen. That is why I believe open reporting, clear labeling, and real oversight need to be part of the solution.
Looking Beyond Simple Substitutes
Switching from one PFAS to another doesn’t give the world a clean slate. From what I’ve seen in industry news and science journals, real progress comes through safer materials research — not just swapping out a single molecule in an effort to hide from regulation. A lot of clothing and packaging can use coatings made from non-fluorinated chemicals and still get the job done. Laws in some states already push companies toward these safer choices, but loopholes and trade secrets stall broader change. Pressure from consumers matters, and public spending can back companies trying to move away from PFAS altogether.
Standing Up for Safer Choices
PFHxA paints a picture of modern convenience tangled up with long-term risk. Convenience isn’t worth much if future cleanup costs and health concerns pile up. As more people ask questions — and as government oversight catches up with science — I see hope that safer, more open industry practices can take root. Change often starts with a single question: what’s in this, and is it safe for my family? That’s the conversation that needs to keep growing.
Getting Real About Everyday Chemicals
Perfluorohexanoic acid, or PFHxA, crops up a lot where folks least expect it—inside raincoats, food packaging, floor wax, and stain-resistant carpeting. Despite the harmless-sounding name, this stuff belongs to a bulky family called PFAS, short for “per- and polyfluoroalkyl substances.” Most people know these as “forever chemicals.” They earned that nickname since they don’t break down in the environment, or in your body, for a long, long time.
What Science Really Says About PFHxA
Plenty of studies focus on older, longer-chained PFAS, like PFOA and PFOS. These grabbed headlines after links to cancer, immune problems, cholesterol spikes, and more health problems. PFHxA got less attention so far, since it’s a six-carbon version—shorter than some cousins. Some companies switched to PFHxA, thinking it might be safer. There’s no slam-dunk evidence yet that PFHxA poses the same risks as the heavyweights, but regulators and watchdogs worry since the chemical still sticks around inside us and out in the environment.
Animal testing found PFHxA doesn’t build up inside the body as much as long-chain PFAS. That helps, but persistence alone raises concern. People can’t avoid exposure: water samples in several countries show PFHxA leaks into groundwater and rivers. Crop irrigation and drinking water add up. Scientists keep finding these chemicals in human blood—even in kids.
Everyday Exposure and What It Means
The worry grows each year. PFHxA shows up in places folks touch and use all the time. Rainwear and fast-food wrappers let small amounts into hands and mouths. Nobody signs up to consume PFHxA, yet food and water bring it to kitchen tables worldwide. The Environmental Protection Agency recognizes the risk, warning that “forever chemicals” like PFHxA deserve limits to keep levels as low as possible.
Some say that PFHxA doesn’t hang around in the body like its long-chain relatives, but this doesn’t make it harmless. Scientists point out that just because a chemical leaves your system faster doesn’t make daily, repeated exposure safe over decades. Health effects may take years to show up. The early research raises flags for impacts on the kidneys, liver, and developmental health in lab animals. Few good studies on human health exist so far. That uncertainty is its own problem—nobody likes playing chemical roulette, especially moms, farmers, and families who can’t pick where their water comes from.
Moving Toward Safer Solutions
PFHxA keeps drawing attention from regulators, local water utilities, and anyone who deals with wastewater. Lawmakers in some areas have pushed for bans or tight restrictions. Many companies scramble to swap out PFHxA but still want stain-proof clothing or grease-resistant wrappers. Real change means rethinking whether we want consumer products that need these chemicals in the first place.
We all share the same water supply and food chain. The best solutions rely on strict oversight, more independent research, and better industry transparency. My own experience digging into family water safety makes me believe that clean—and truly safe—drinking water must be a basic expectation, not a privilege. Only public pressure and tougher standards can push companies and regulators to act before the next “forever chemical” becomes yesterday’s regret.
Understanding Why Correct Storage Matters
Some chemicals just stick around, long after the lab has closed. Perfluorohexanoic acid, known as PFHxA, is one of those compounds that scientists and regulators watch closely. This acid belongs to the broader family of PFAS, substances people in water treatment, food packaging, and manufacturing cause headaches for because of how they persist in the environment. Improper storage of PFHxA only makes things worse, risking health and triggering expensive cleanup projects.
Lessons from Labs and Workplace Experiences
A few years back, I worked in an environmental chemistry lab where PFAS standards were a regular fixture. Peers would grumble every time a container leaked or when labels faded. I remember the tension whenever an inspection loomed and someone found a container in a questionable state. That stress wasn’t about paperwork; it came from knowing that a spill or evaporation meant releasing something incredibly difficult to remove. There’s a reason the phrase “forever chemicals” rattles through scientific circles.
Key Principles for Safe Storage
Storing PFHxA calls for close attention to detail. Use tightly sealed containers made from high-density polyethylene or fluoropolymer materials — glass risks etching or corrosion, and regular plastics often fail. The containers sit best in cool, dry spaces away from direct sunlight. Humidity and heat speed up decomposition or accidental release, which can spread vapors or residues further than anyone expects. Secure shelving keeps things upright and safe from bumps, drops, or mistaken grabs during a busy shift change.
Labeling is no small thing here. Forgetting a date or letting a sticker peel off means nobody will remember what’s inside the bottle next month. Strong, solvent-resistant labels cut down on confusion. In my experience, double-checking dates and hazard codes saved more than one person from grabbing the wrong bottle for an experiment. For PFHxA, knowing what’s inside means fewer mistakes and safer handling for everyone.
Why Safety Practices Deserve Real Commitment
Health doesn’t wait for regulations to catch up. PFHxA has slipped into water bodies where scientists can already track its impact on aquatic life. It isn’t as headline-grabbing as some longer-chained PFAS, but it wears away at health standards and makes cleanup projects costlier. Direct contact with concentrated PFHxA can cause irritation, and disposal costs rise when residues end up outside controlled conditions.
Storing it properly avoids costly incidents. Inspections from environmental agencies pick up on lapses — and fines add up, especially after repeat violations. Maintaining a dedicated log book with storage dates, handled by someone with actual hands-on experience, keeps records clear and honest. In the end, it’s about respect for coworkers and neighbors who shouldn’t find out about PFHxA five years down the line when drinking water tests change color.
Practical Solutions for Better Storage
Training doesn’t have to slow down a busy lab. Teams can walk through storage protocols during regular safety meetings. Having up-to-date safety data sheets close at hand, in plain language, stops myths from spreading. Rotating stock and clearing out unused bottles keeps everything within expiration dates, and setting reminders on a calendar works better than relying on memory. These simple steps lower risk and make storage just another habit, not a headache.
Understanding the Place of PFHxA in Our World
Perfluorohexanoic Acid, known by its acronym PFHxA, belongs to a group of chemicals called per- and polyfluoroalkyl substances or PFAS. These so-called “forever chemicals” share one trait: they stay in the environment, seemingly unaffected by time. PFHxA has cropped up in all sorts of places, from stain-resistant fabrics to firefighting foams, and even in our favorite fast food wrappers. Even folks who aren’t entrenched in environmental policy have started to wonder: are there real rules about what companies can do with it?
Is PFHxA Actually Regulated?
Most people have heard about PFOA and PFOS, which got the regulatory spotlight after doctors linked them to cancer and other health problems. In contrast, PFHxA hasn’t had the same headline-grabbing attention. In the United States, regulations don’t specifically target PFHxA on the federal level. The Environmental Protection Agency (EPA) has looked at groups of PFAS, but the strictest rules only focus on a handful of the older, longer-chain chemicals.
Europe has taken some steps farther. The European Chemicals Agency included PFHxA and its salts and related substances on its list of substances of very high concern back in 2020. Some member states are pressing for tighter limits, especially in items that touch food or water. Still, PFHxA-based products continue to show up in places most people wouldn’t expect, and there’s little to stop it.
Why Does PFHxA Matter for People and Nature?
Research points out that PFHxA doesn’t stick around the body the way old-school PFOS or PFOA can. That doesn’t mean PFHxA gets a free pass. Studies in animals have shown changes in the liver and kidneys after exposure. We can’t say with certainty what that means for people, especially with long-term contact. If nothing else, the stubborn persistence of PFHxA in the environment means it piles up in rivers, soil, and wildlife. All that build-up sets off alarm bells for anyone who cares about safe water and food.
I’ve watched towns panic after learning PFAS have made their way into local water. Most families can’t afford bottled alternatives long-term. Without solid rules about PFHxA, communities rely on outdated guidelines to judge safety. Scientists agree that “the dose makes the poison,” but with so many new chemicals being made, it’s tough for regulators to keep up with what’s coming out of factories.
Paths Toward Safer Solutions
Stronger action is overdue. Right now, most rules work like a patchwork quilt, where some states or countries ban PFHxA in some uses, and others don’t. This inconsistency creates loopholes big enough to drive a truck through. National and international groups need to broaden their focus beyond just the old PFAS to include newer ones like PFHxA.
Governments could require more transparency from manufacturers. If companies had to test and share where and how much of these chemicals go into their products, folks would have a fair shot at avoiding them. Incentives to invest in safer alternatives would also push the needle forward; businesses tend to move fastest when rules level the playing field.
Bringing the Issue Into Public View
Few people feel empowered to ask hard questions about what’s in their water or food packaging. Sharing clear, honest information about chemicals like PFHxA helps build trust and puts pressure on decision-makers. No one can solve a mystery they don’t know exists, and the public has a right to know what risks may lurk in plain sight. Ensuring safer products won’t happen overnight, but turning up the spotlight on PFHxA marks an important step toward protecting both people and the planet.
Understanding the Risks
Perfluorohexanoic acid (PFHxA) doesn’t show up in the news as much as some chemicals, but those of us who have spent any time in industrial labs or environmental science know just how tricky it can be. This isn’t just another lab solvent. PFHxA falls into the family of perfluoroalkyl substances (PFAS), nicknamed “forever chemicals” for how stubbornly they resist breaking down. There’s a reason workers and researchers keep a healthy respect for substances like this: you don’t want them lingering in your body or seeping into the water supply.
True Protection Comes from Real Precautions
Lab coats do their part, but no one should touch a vial of PFHxA without more than just fabric between their skin and the substance. Chemical-resistant gloves are the baseline. Nitrile or neoprene gloves tend to hold up better against PFAS compared to latex, and no one wants chemicals slipping through unnoticed. Splash goggles stop droplets from getting into your eyes, and a face shield increases security if you’re pouring or weighing out powder. I’ve watched enough accidents happen in shared lab spaces to know that over-preparing beats underestimating a risk.
PFHxA vapor doesn’t belong in your lungs. Working under a fume hood cuts inhalation risk dramatically. Many PFAS carry concerns beyond acute exposure, since the long-term impact on organs and development in children keeps cropping up in studies. Air filtration in the room matters, as does keeping the workspace clean—not just wiping up with any old rag, but following with detergents suited to PFAS. Ordinary soap won’t always do the trick, so treating benches with the same respect you give to biological contamination makes sense.
Hard Conversations About Waste
Walking PFHxA-contaminated waste over to a regular trash can or draining solutions down the lab sink ended long ago. Regulations are catching up with these persistent chemicals because they slip past many typical water treatment barriers. I’ve seen too many labs that cut corners with disposal, figuring low concentrations don’t matter. They do. Proper hazardous waste containers, labeled and managed with site-specific protocols, keep this stuff out of drinking water—at least that’s the goal.
Some companies claim they’ve developed better destruction methods for PFAS, from thermal processes to high-energy breakdown systems. Those tech fixes sound promising but haven’t become routine in most institutions. Until then, the only practical path is to reduce use, contain every step of the process, and never assume someone else will clean up safely later.
Accountability, Not Shortcuts
Handling PFHxA properly speaks to more than just worker safety. It’s how organizations show real accountability to their communities and the environment. Ignoring the guidelines or underestimating chronic effects can cause harm long after someone has left the building. The toughest part is that you can’t see or smell these chemicals much of the time. Policies and training aren’t just legal requirements—they carry moral weight. I once had a trainee balk at the extra time spent storing PFHxA waste, asking if it really made a difference. Watching rivers and towns contend with PFAS pollution answers that question several times over.
Facing the Problem Head On
PFHxA isn’t some theoretical hazard. If you’re transporting, measuring, or disposing of it, use every tool and precaution at your disposal—because the fallout from a mistake sticks around for decades. Science isn’t just discovery or production. It’s about stewardship. There are no shortcuts worth taking with chemicals that outlast us all.