Perfluorohexyl Iodide: Tracing the Story of a Niche Fluorochemical
Historical Development
Back in the mid-20th century, chemists eager to develop ever-more inert, stable, and robust compounds turned to the fluorinated world for inspiration. Perfluorohexyl iodide wasn’t part of the early flurry of Teflon and refrigerant hype, but as researchers dug deeper, they unearthed new applications for perfluoroalkyl compounds, often by swapping out functional groups for iodine. The track record of perfluoroalkyl iodides is told in patents and research papers rather than headline-making discoveries. Over time, these compounds became valuable building blocks in industries such as specialty materials, pharmaceuticals, and advanced coatings, a testament to the doggedness and gradual curiosity of those working in less crowded corners of chemistry.
Product Overview
Perfluorohexyl iodide steps onto the scene as a six-carbon linear molecule, decked out entirely in fluorine atoms except for one—where a single iodine clings at the end. This molecular design gives it a unique position among the family of perfluorinated compounds. Unlike the short-chain materials that are drawing regulatory attention for their environmental persistence, this molecule finds favor mostly for downstream synthesis and not for mass consumer products. The chain length provides a balance between reactivity and manageable handling, which sets it apart from longer, more unwieldy perfluoroalkyl iodides that often come with trickier logistics and costs.
Physical & Chemical Properties
Holding a bottle of perfluorohexyl iodide, I’m always struck by its distinct heft. The liquid is dense, oily, and resistant to outrageously high and low temperatures—a direct benefit of the fluorine atoms locked along its backbone. One of the telling features is its chemical inertness toward most common reagents. The iodine atom, by contrast, marks the only site prepared for further chemical modification. Reactions with perfluorohexyl iodide need precision and respect; mishandling the iodine could easily derail the best-laid plans for downstream chemistry. The color ranges from colorless to pale yellow, and it resists the slow creep of air and moisture, which makes storage less anxiety-inducing than with some reactive intermediates.
Technical Specifications & Labeling
Preparing to use this compound, researchers study documentation not simply for purity but for subtle marks of quality. Perfluorohexyl iodide typically meets stringent gas chromatography or NMR requirements for identification. Safety information deserves close reading, especially for proper storage, transport, and use—given that iodinated organics sometimes leave residues that can trip up sensitive analytical equipment or leave persistent odors. It’s routine to see sealed glass ampoules for packaging, with clear attention paid to avoiding light and air exposure when possible.
Preparation Method
In the lab, synthesis of perfluorohexyl iodide tracks a route paved by earlier generations of fluorochemical production. The well-worn approach involves telomerization of tetrafluoroethylene, sometimes followed by halogen exchange reactions where hydrogen or chlorine atoms are replaced by iodine. The process isn’t glamorous but it speaks to the old-school skills of stepwise synthesis, careful distillation, and purification. Unlike the more headline-grabbing “green” syntheses of today, these methods lean heavily on industrial infrastructure, high-vacuum systems, and meticulous exclusion of contaminants. Each step brings opportunities to reflect on both the power and responsibility of wielding fluorous chemistry—there’s satisfaction in executing the craft well but always a note of caution about waste streams and process emissions.
Chemical Reactions & Modifications
The beauty of perfluorohexyl iodide lies in its eager response to radical initiators. The iodine makes it a launchpad for C–C bond formation, perfect for introducing fluorous tags onto other molecules or for building out complicated perfluoroalkyl chains. I have seen researchers light up when a reaction goes smoothly—using light, heat, or specific initiators to cleave the I–C bond and attach the perfluorohexyl group to molecules that otherwise would never touch the fluorinated world. Not just for academic bench chemistry, these reactions shape surfactants, lubricants, and specialty monomers. Sometimes the modifications run through transition-metal catalysis, other times straightforward nucleophilic substitution. Each method brings quirks and risks—always demanding a close eye on side products and the broader environmental footprint.
Synonyms & Product Names
Looking through supply catalogs or chemical registries, the same substance comes up under a handful of labels: hexafluorohexyl iodide, perfluoro-n-hexyl iodide, or simply C6F13I. Synonym confusion is a running joke for anyone scouring literature, where the same fluorocarbon shows up under subtle variants. Knowing the differences can mean the difference between a successful literature search and a frustrating wild goose chase. A bit of diligence in cross-referencing CAS numbers or structural diagrams goes a long way.
Safety & Operational Standards
In handling perfluorohexyl iodide, the stakes are high, but not always in the way one might expect. The general consensus in physical organic labs is to glove up, work under fume hoods, and double-check connections before heating. Its low volatility doesn’t mean it’s harmless—any process involving iodinated or heavily fluorinated compounds raises flags with occupational health teams. Spills, even small ones, call for serious cleanup to avoid stubborn residues. Waste management plays a critical role, as fluorinated byproducts don’t degrade easily in the environment. Collecting and disposing of all residues and labware properly is non-negotiable, and for good reason—these compounds have a tendency to persist beyond their welcome if mishandled.
Application Area
Perfluorohexyl iodide earns its place as a star in niche applications, not consumer products. Research teams use it to craft surface-active materials, specialty coatings, and intermediates for more elaborate fluorinated compounds. I’ve heard more than one scientist describe the exhilarating chase to make water- and stain-repellent textiles, where just a dash of this reagent unlocks whole new categories of performance. Electronic manufacturers have also found it essential in working with lubricants and insulating materials. Outside of these fields, evidence points to its growing use in drug development, where it can slip a fluorous function into pharmaceutical candidates, sometimes enhancing metabolic stability or bioavailability.
Research & Development
Any compound that delivers both performance and processing headaches naturally draws the attention of R&D labs. Teams probe for safer, more efficient synthesis routes, eager to shrink costs while managing environmental liabilities. A recurring storyline is the search for milder iodination methods, both to reduce waste and make production less costly. Studies into the reactivity of C–I bonds in perfluoroalkyl environments have led to new methods for attaching these groups to otherwise unreactive substrates. On the applications side, partnerships between universities and manufacturers often spark breakthroughs—be it in ultrathin coatings, microfluidic device manufacturing, or next-generation energy storage systems. Each new project serves as a reminder that seemingly esoteric compounds like perfluorohexyl iodide can punch far above their weight when paired with creative problem-solving and interdisciplinary collaboration.
Toxicity Research
Concerns about the health and environmental impact of fluorochemical intermediates have grown. Studies on perfluorohexyl iodide lag behind the heavy scrutiny focused on the likes of PFOA and PFOS, but the structural similarities keep regulators and researchers alert. Data suggests that low volatility may slow off-gassing or airborne exposure, yet its resistance to breakdown makes accidental release a stubborn long-term concern. Toxicity profiles hinge on the persistence and potential for bioaccumulation—topics that have only started to draw systematic investigation. A lesson that’s blindingly obvious to anyone who’s grappled with disposing of lab waste: prevention beats cure, every time.
Future Prospects
There’s no mistaking the uncertain road ahead for perfluorohexyl iodide, or any perfluorinated chemical. On one hand, demand continues for performance materials that shrug off harsh environments or provide unique surface effects. On the other, the lagging pace of toxicity and degradability research means regulatory pressure will almost certainly mount. Innovation won’t come solely from chemistry; improved lifecycle management, greener synthesis, and rapid toxicity screening will have to define the next era. The best prospects look to a synergy between practical breakthroughs and strong oversight—making sure the compound keeps earning its keep, without creating the kind of legacy that later generations curse. Chemistry always faces a balancing act between progress and stewardship, and nowhere is that clearer than the story unfolding around perfluorohexyl iodide.
A Closer Look at a Specialized Chemical
Perfluorohexyl iodide rarely makes the evening news, but anyone working in advanced chemistry knows its role. This compound shows up in laboratories focused on pharmaceuticals, chemical research, and materials science. Its long, stable perfluoroalkyl chain, plus that reactive iodine atom, creates all sorts of opportunities for new molecules. With experience in academic labs, I’ve seen researchers explore how such specialized chemicals allow for creativity, yet bring up tough questions about safety and environmental impact.
Chemical Synthesis and Unique Properties
The secret to perfluorohexyl iodide lies in the perfluorinated carbon chain. Fluorine, in this configuration, creates persistence—a property that makes many fluorochemicals valuable and tricky. Chemists love using perfluorohexyl iodide to introduce perfluorohexyl groups into larger molecules. This has turned out to be a game-changer for making specialty surfactants, designing non-stick coatings, and helping researchers develop new drug candidates.
The chemical’s most interesting trait is the iodine atom. It’s reactive in the right hands, helping link that stubborn fluorinated tail to other structures. For making advanced materials, or even diagnostic agents in medical imaging, this chemical has few replacements. Perfluoroalkyl iodides give scientists building blocks for designing molecules that behave in controlled and predictable ways.
Concerns: Persistence and Exposure
Just as these materials bring promise, they’ve raised legitimate worries. Perfluorinated chemicals, including perfluorohexyl iodide, persist in the environment. Once released, they don’t simply disappear. The stability that makes them so appealing for industrial uses turns around to haunt water systems and living tissues. PFAS (per- and polyfluoroalkyl substances), the class of chemicals to which this compound belongs, have turned up in unexpected places—sometimes in blood samples, sometimes in drinking water.
Scientific reviews and monitoring data have shown that prolonged exposure to some PFAS links to health issues. These risks include thyroid disruption, potential cancer connections, and immune system effects. National health agencies keep ramping up studies and regulatory review, especially for chemicals that share the same carbon-fluorine backbone.
Ideas for Responsible Use
Industry can’t just walk away from these substances, especially where no alternatives match their performance. Yet, every use must come with a plan for minimizing accidental release. Facilities ought to invest in closed systems, effective waste treatment, and recycling programs—something I’ve seen some companies implement with decent results. Transparency matters, too. Chemists, managers, and policy makers should share what’s known about environmental pathways and keep data open to public review.
Regulatory bodies have started taking steps to track and manage PFAS use, pushing for better labeling and more robust disposal practices. Researchers continue hunting for alternatives with similar performance and less persistence. Moving forward, companies should align with those efforts, support greener approaches, and re-evaluate where these compounds are truly essential.
Everyday Action and Long-Term Impact
Most people will never handle perfluorohexyl iodide directly. Yet, products and research that depend on it reach everywhere. Making smart choices in chemical innovation includes looking beyond convenience and performance. Each step toward transparency and responsible management can help build trust between scientists, industry, and the communities affected by these substances. That path demands effort, but protecting future generations always does.
What My Years Around Labs Have Taught Me
Rarely does a chemical make you stop and think about everything from skin protection to waste management quite like Perfluorohexyl Iodide. Most people probably never run into it, but those of us who have seen its quirks in action know it plays by rules you can’t ignore. My years working with specialty chemicals reminded me that casual handling has real consequences—sometimes ones you don’t see until too late. Street wisdom from the lab: treat every unfamiliar bottle with some humility.
Worst Mistakes Happen When Rushing
Lab stories usually feature one common villain: the shortcut. Rushing to finish up, skipping gloves for a quick transfer, not clicking on a fume hood—all rookie moves. With Perfluorohexyl Iodide, that’s an invite for trouble. This compound brings some toxicity and it doesn’t play nice with unprotected skin or eyes. It handles sneaky spills and airborne vapors much better than people do. You won’t see or smell the vapors, but that doesn’t mean they're not doing harm.
Practical Steps, Not Fancy Gear
Gloves actually matter. Nitrile or better. Cheap latex lets chemicals creep through much faster. Goggles become your frontline defense—no exceptions. Open beakers and bare eyes make a recipe for stinging regret if the stuff splashes. A fume hood isn’t just a box with a fan; it’s a barrier between you and irritation, headaches, and worse. Running your experiment in a breezy room or with the windows open won’t cut it.
Lab coats stop more than stains; fewer ruined shirts and less exposure to something that likes skin. Wash hands before you eat or use your phone. Sounds basic, but lunchroom stories always include someone who forgot. Contamination is quiet—until it isn't. I’ve seen folks rub an eye carelessly and regret it for days.
Spills and Bottles Can’t Wait
Don’t leave open bottles around—Perfluorohexyl Iodide doesn’t need an invitation to evaporate. Small spills treated lightly can turn into big headaches later. Go for absorbent pads and good ventilation. Bag and tag waste, never pour it down the drain. I once made the mistake of thinking “it’ll just flush out with water.” The local rules will remind you otherwise, and the environment will pay for the oversight.
Training Isn’t Optional
Doesn’t matter if it’s your first day or your hundredth. Solid chemical safety training saves you from learning by accident—trust me, you don’t want to “just figure it out.” Even seasoned lab rats reach for the data sheets every time logic wavers. Institutional memory gets built from people sharing honest mistakes.
Better Habits, Fewer Regrets
Regulations exist for good reasons, and you don’t really appreciate them until something almost goes wrong. Following established procedures, labeling everything, and respecting disposal rules cuts down on risk. No one wants to spend a night explaining to emergency responders why the air smells off or why their hand looks swollen.
Some chemicals force you to grow up. Perfluorohexyl Iodide fits that bill. Straightforward caution, real-world habits, and genuine respect for risk go a long way. Looking after yourself and your coworkers means less drama and more days ending without a trip to the clinic.
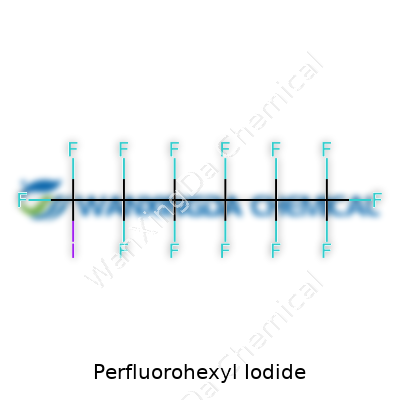
Understanding the Backbone: C6F13I
I remember my first introduction to perfluorohexyl iodide back in a university lab, getting a close look at its chemical makeup. The formula sticks in my mind: C6F13I. What you see here is a hexane backbone, now supercharged with fluorine atoms in place of the usual hydrogens, and an iodine atom hanging off the end. Chemically, you've got six carbon atoms joined in a straight line, every carbon tightly hugging its quota of fluorines except the last one, which bonds to a single iodine.
Visualizing the structure, each carbon links to two fluorines — except for the carbons at the tips. The very end of the chain bears the iodine attached directly to the terminal carbon, so you get something like CF3-(CF2)5-I. That combination pushes some unique chemistry and environmental challenges that experts keep debating.
Why This Structure Makes a Difference
Across chemical labs and industry groups, the pull of perfluorohexyl iodide comes from its chain length and mix of fluorine and iodine. That sturdy carbon–fluorine cage builds a reputation for resistance to heat, acids, and pretty much anything else it faces. That’s what got my attention as a graduate student. In effect, adding iodine gives scientists a useful “handle,” letting them build even more complex molecules, especially when tailoring surface coatings or starting tough chemical syntheses.
Beyond the lab, this sort of chemistry isn’t just neat — it’s the reason water and grease bounce right off treated fabrics or wires. With perfluoroalkyl chains like this, products get their non-stick power or protect sensitive electronics. The downside, as current research and regulatory focus reveal, is that this class of compounds doesn't break down easily. Once they’re in soil or groundwater, they stick around.
Health, Environment, and Solutions
Environmental persistence means scientists and regulators are on alert. Communities living near manufacturing plants have raised serious health questions after learning these chemicals wind up in drinking water and even food. Some peer-reviewed studies link members of the perfluoroalkyl group with immune suppression, elevated cholesterol, and higher cancer risk, though perfluorohexyl iodide itself awaits deeper investigation.
In my own time at a water treatment plant, I saw the real-world struggle with these so-called “forever chemicals.” Conventional filters barely put a dent in them. Activated carbon and advanced oxidation have been tested, but costs surge and waste disposal complicates things. Regulatory bodies are trying to understand how these molecules move and transform in the environment. The bigger question remains: how can industries reduce or substitute these compounds before they scatter even more?
A promising approach starts with green chemistry — encouraging companies to develop alternatives at the molecular design stage. Some researchers have proposed replacing halogen atoms or shortening chain lengths to cut down toxicity and persistence. Others champion better containment, robust waste treatment, and stronger oversight. None of these options feel easy or cheap, but they’re necessary to keep communities safe.
Looking Ahead
People working with perfluorohexyl iodide, or living near its footprint, deserve straight answers. The chemical’s formula and structure explain both its usefulness and risks. Sharper oversight, more transparent reporting, and actual investment in safer alternatives will shape the future for not just this compound, but all its relatives. When science and policy act together, the odds of keeping both technology and health on track get a lot better.
Storing Science’s Powerful Tools
Inside most chemistry labs, the colorful flasks and tidy drawers might seem orderly to a visitor. Beneath the surface, though, each chemical carries its own risks and quirks. Perfluorohexyl iodide is one of those compounds that shows us how a little bit of knowledge, organization, and preparation can make all the difference between safe research and real trouble. Some folks see only the complex name and tune out, but handling this stuff shows that all the small habits around storage matter.
Why Temperature and Light Matter
Heat and sunlight bring out the worst in a lot of chemicals. Perfluorohexyl iodide reacts to both—store it in a warm, sunlit spot and you’re practically inviting it to break down, maybe even spill or off-gas something nasty. I remember starting out in a lab where someone once tucked a similar compound above a radiator for convenience. Bottles like those can warp, and before long, the whole room picks up an odd, acerbic smell. For this compound, room temperature just won’t cut it. That means refrigeration, in an area with clearly marked hazards, with no sun streaming in or heat source nearby. Most labs use dedicated fridges for this class of material.
Tight Caps, Clean Shelves
Oxygen and moisture seep in quickly wherever bottles aren’t checked or caps aren’t screwed on tight. Every chemist learns the hard way—loose tops mean contaminated reagents and sometimes damaged experiments. Perfluorohexyl iodide likes to react with water too. I’ve seen forgotten vials at the back of a shelf, sticky with condensation after months of neglect, labels swollen and peeling. Clean, dry shelves and a quick double-check every time you pull out a bottle make a world of difference, even in a busy lab.
Labeling and Segregation
I’ve never worked in a place where the biggest threat was the size of the chemical inventory—it’s always the state of the labeling and separation. Storing this compound away from strong bases, acids, or oxidizers takes only a bit of shelving discipline, yet mistakes still happen. Mixing incompatible chemicals in a cramped storage cabinet doesn’t just risk a ruined sample. It’s a serious safety hazard. Legible labels, consistent storage rules, and trays or secondary containment all help cut down on mistakes that come back to bite later.
Personal Experience and Solutions
No matter how many times a group leader runs safety drills, the best habits grow out of small acts: tracking expiration dates, updating the inventory system, rotating out old stocks. Bringing a new bottle of perfluorohexyl iodide into storage offers a chance to reinforce these good habits. I’ve seen teams thrive when anyone who notices a slip in storage steps up—no one waits for a fire marshal’s report. Digital inventory logs work well for high-turnover labs, while a laminated checklist on the fridge door keeps things simple and clear—no need for special software or gadgets.
The Bigger Picture
Careful handling and storage of tricky chemicals like perfluorohexyl iodide doesn’t just protect the experiment or the researcher. It protects everyone who works nearby, and sometimes even those down the hall. Solid training, a bit of respect for what might go wrong, and a culture where people ask questions and watch out for each other give us safer labs and stronger science. That’s what good chemical storage tells us about responsibility—every bottle and label counts.
Understanding What’s in Our Backyard
The chemical world sometimes brings trouble that most people never see coming. Perfluorohexyl Iodide belongs to a group called PFAS—per- and polyfluoroalkyl substances. These are molecules tough to break down, popular in industrial settings and, over the years, linked to health and environmental concerns. Long-lasting chemicals like this can make a mess that outlives those who created it.
Everyday Life and Long-Term Exposure
Walking into a workplace that uses perfluorohexyl iodide doesn’t set off alarm bells like walking into a smoky factory did decades ago. The real worry comes from repeated and extended exposure. PFAS compounds often build up in the body over time. Studies keep linking them to liver, immune system, and developmental effects. The Centers for Disease Control and the World Health Organization have shown that certain PFAS types spike risks for some cancers, thyroid issues, and high cholesterol.
People living near facilities making or using perfluorohexyl iodide face the biggest risks. Drinking water contamination stories fill the news from communities near chemical plants, and PFAS sticks around in soil and water long after factories close their doors. These chemicals don’t break down, so their impact spreads, hitting fish, livestock, and creeping up the food chain until families far from the source see the effect on their dinner plates.
Environmental Hazards—No Easy Cleanup
Once perfluorohexyl iodide finds its way into the environment, getting rid of it feels a lot like chasing smoke. Water treatment plants aren’t set up to filter these out. Rain washes the chemical into rivers and lakes, where it keeps moving. Soil contamination adds another layer, turning up in crops and animals. This bioaccumulation means even folks trying to eat clean face PFAS in their meals.
Wildlife has shown some heartbreaking results. Lab studies with birds, fish, and mammals show lower birth rates and changes to immune systems after PFAS exposure. Natural resources become contaminated for generations. Companies once pitched these chemicals as miracle products that never wore out. Now, that stubborn durability spells out a generational responsibility for cleanup and mitigation.
Where Solutions Start
History taught us that waiting for perfect science leaves too many at risk. Regulation has started to catch up, setting stricter limits on PFAS, with some governments banning or phasing out certain types. Perfluorohexyl iodide falls under the growing watch list as research continues. That’s not the finish line—constant monitoring, updating water and soil testing, and pushing research into safer alternatives matter.
On the ground, I’ve seen local activists push for better transparency in chemical reporting and real-time leaks monitoring. Public pressure can force plant operators to tighten their operations and take responsibility for existing pollution. Communities linked arms around the world, forcing slow-moving regulatory wheels to turn a bit faster.
The fight against stubborn chemicals calls for a team effort—scientists, lawmakers, and regular folks watching out for their neighborhoods. Honest research, transparent policy, and stubborn advocacy stand as the first real tools available. No one wants these stories to become tomorrow’s cleanup disasters, so keeping perfluorohexyl iodide in the spotlight is not some abstract exercise. It’s about holding on to health—both human and ecological—for the long haul.