Perfluorooctyltrichlorosilane: More Than Just a Borrowed Tool from the Chemical Cabinet
Historical Development
Perfluorooctyltrichlorosilane, often tagged by its tongue-twisting name, traces its roots back to the surge in fluorochemical research following World War II. After the legendary story of Teflon hit the mainstream, chemists began exploring the potential of fluorinated compounds. Markets craved coating agents that held up against oil, water, and temperatures that sent lesser polymers running. The drive for ever-more-repellent surfaces led to a series of silane derivatives. By the late 20th century, this compound emerged from the shadows of academia into industrial labs, carving out a niche in materials science that continues to expand.
Product Overview
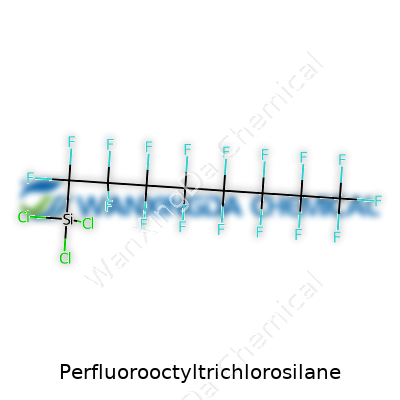
Some chemicals exist just to fill gaps in a list. Perfluorooctyltrichlorosilane stands out for doing something most common chemicals fumble: it shrugs off just about anything a lab can throw at it. Known among researchers and product engineers for its tricky balance between two worlds, it pairs a glass-loving (hydrophilic) head with a slick, water-and-oil-dodging tail. This blend makes it a go-to choice for surface treatments, giving glass, silicon, and metals that signature water-beading effect along with dirt and oil repellency.
Physical & Chemical Properties
You won’t find Perfluorooctyltrichlorosilane behaving like a household cleaner. The compound’s clear to slightly yellow liquid form signals a cocktail of chlorine and fluorine atoms. Chill it, and it won’t freeze easily; heat it, and most air will catch a sniff of something not to breathe in. Chemistry teachers always reminded us: mixing silanes and water triggers a reaction. This one is no different, breaking down, forming hydrochloric acid, and sticking to nearly any glass or metal surface you let it touch. Those ever-present C-F bonds mean the end product holds strong against acids, bases, solvents, and UV, earning its keep in harsh environments.
Technical Specifications & Labeling
Handling this silane means more than just rubber gloves. Big hazmat symbols greet anyone unpacking a shipment. Engineers and lab techs treat the substance with respect, knowing that even a whiff can mean a trip to the eyewash station. Labels warn of water-reactivity, inhalation risks, and recommend seriously robust ventilation. Concentration and purity matter—research typically calls for 97-99% pure material, with any impurities threatening both the intended reaction and user safety.
Preparation Method
Producing Perfluorooctyltrichlorosilane isn’t the work of a garage chemist. Starting with a perfluorinated alcohol, chemists react it with trichlorosilane, squeezing out the product under tightly-controlled, water-free conditions. Every trace of moisture spells trouble because the silane would grab those water molecules, break down, and make the batch useless. Industrial setups employ dry solvents, specialized glassware, and inert gas blankets to keep the chemistry on track.
Chemical Reactions & Modifications
This is where things get interesting for anyone aiming to build water-repellent or oil-proof surfaces. Perfluorooctyltrichlorosilane reacts almost instantly with surfaces loaded with hydroxyl groups. The trichlorosilane part grabs onto glass, forming a covalent bond, while the fluorinated “tail” stands out, conferring slickness that few other chemistries replicate. If someone wants to tune performance, modifications can swap the chain length or play with the silane head for different sticking power or repelling properties, making it adaptable for surfaces beyond plain silica.
Synonyms & Product Names
The compound goes by a handful of synonyms in industry and research literature: trichloro(1H,1H,2H,2H-perfluorooctyl)silane appears in journals; some refer to it more generally as perfluorinated trichlorosilane. Shortened names like "FOTS" crop up in nanotechnology circles. Despite the jumble of syllables, the chemistry and function stay consistent.
Safety & Operational Standards
Real-world handling calls for full respect for its hazards. Labs install local exhaust systems. Workers don chemical splash goggles, chemical-resistant aprons, and gloves rated for highly reactive materials. Laboratories post protocols around emergency neutralization and proper storage, far from moisture and incompatible substances. Disposal isn’t like tossing out old solvent—perfluorinated compounds can build up in the environment, and regulations demand incineration at high temperatures to limit environmental impact.
Application Area
Surface science thrives on functional coatings, and here Perfluorooctyltrichlorosilane shines. It anchors itself to microchips as a self-assembled monolayer, protecting patterns on next-gen electronics against dust and water. It finds a place in medical device manufacturing—catheters and implants rely on non-stick surfaces to cut infection risks. Researchers use it to keep tiny microfluidic channels flowing, fighting both contamination and clogging. On the consumer side, you’ll see its influence on stain-resistant fabrics and smudge-proof screens, though supply chain shifts and regulatory hurdles mean not every slick gadget draws on it directly.
Research & Development
Universities and corporate labs dig deep into tweaking this molecule’s structure and exploring alternatives. The focus lands squarely on understanding molecular self-assembly, tailoring surface energy, and finding patterns that work at both nanometer and practical, hands-on scales. Nanofabrication, biosensing, and flexible electronics all stand to gain if surface modification becomes easier to control or safer to handle. Every year, new articles delve into how adjusting the fluorinated chain impacts oil and dust repellency or alters optical properties, feeding back into fresh product designs.
Toxicity Research
Few parts of chemistry raise more concern these days than what happens after a product leaves the lab. Research into perfluorinated compounds as a whole keeps surfacing warnings—these molecules break down slowly, showing up in wildlife and waterways far from any lab. Studies link persistent exposure to health risks, including some cancers and developmental issues. Tightening regulation and closer toxicology work aim to clarify exposure pathways and set safe limits. Workers in production or heavy-use environments receive up-to-date training and regularly review new findings, learning from the growing body of occupational health research.
Future Prospects
Pressure mounts to find replacements that perform as well as perfluorinated silanes without lingering in the ecosystem. Entrepreneurs and scientists alike now chase after silicon-based analogues, short-chain fluorinated alternatives, or even non-fluorinated repellents that promise lower environmental lifetime. The world will need more transparent sharing of research, cooperative development between regulatory bodies and private industry, and focused investment to move past a legacy of persistence and toxicity. For now, Perfluorooctyltrichlorosilane continues to anchor a crucial space in the world of surface science, but future breakthroughs in green chemistry may someday ease it off center stage.
Walk into any modern lab with a background in chemistry, and you’ll bump into long names that sound like tongue twisters. Perfluorooctyltrichlorosilane has one of those names. Despite that, it plays a big behind-the-scenes part in a lot of technology and research. My first brush with this chemical came during a college internship, when I watched an engineer coat glass slides for a water-repelling test. Curiosity pushed me to learn why anyone would go to the trouble of using such a difficult-sounding molecule.
The Need for Super-Slick Surfaces
Perfluorooctyltrichlorosilane—let’s call it PFOTS for sanity—gets used for one big reason: it makes things not stick. Water beads up and rolls away, fingerprints refuse to stay. PFOTS delivers that magic, not by sitting on top of surfaces, but by actually attaching itself to them and building a barrier. This repellent layer is a lifesaver in everything from analyzing medical diagnostics to designing phone screens that don’t smudge up as fast as your lunch table.
In research, glass and silicon wafers can drive anyone mad because the tiniest oil or dust will ruin an experiment. PFOTS fixes that problem. A well-coated wafer means samples slide off easily, and cleaning turns into a quick wipe. Picture setting a glass of water on a freshly waxed car hood—no puddles left behind. That’s the level of control PFOTS users seek, and rely on, to run experiments efficiently. I learned early on that this small change in surface chemistry gives results you can trust, with fewer headaches and reruns.
Electronics, Medicine, and Manufacturing Success Stories
Tech giants and small startups both turn to PFOTS for electronics. Touchscreens last longer with resistant coatings, sensors pick up real signals (and not grime), and manufacturing costs go down from less waste and hassle with cleaning. Medical labs want error-free readings and simple cleaning routines. In microfluidics, where tiny droplets zip through microscopic channels, PFOTS stops water and oils from sticking where they shouldn’t, keeping those reactions fast and clean.
Working with nanotechnology, surface science, or precision measurement almost guarantees a lab keeps PFOTS in the cupboard. It’s that good at creating a layer so thin, yet so stubborn, hardly anything wants to cling to it. Aerospace engineers use similar coatings for satellites and aviation, since equipment in harsh conditions can’t afford sticky contaminants. Even art conservators have experimented with these coatings to protect delicate works from the environment.
Weighing Chemistry Against Health and the Environment
No chemical story lands without a catch. PFOTS is part of the big PFAS chemical family—the so-called “forever chemicals.” They don’t break down easily. That means they can gather in water, soil, and even inside people and animals for a long time. I remember talking with a toxicologist who warned that convenience should never outrank safety. Regulatory agencies across the world keep studying the potential for health risks, searching for alternatives, or setting stricter handling rules.
Practical folks are starting to tinker with new molecules. Some researchers are testing plant-based alternatives or tweaking formulas so breakdown happens more quickly after the product’s useful life. If industry moves faster on this, labs and factories don’t have to pick between modern performance and environmental responsibility.
PFOTS remains the gold standard for making things slick and self-cleaning in labs, factories, and gadgets. As a society that loves its shiny screens and high-precision tools, we have to balance that with some caution and priorities beyond the convenience of a clean swipe.
Understanding Perfluorooctyltrichlorosilane’s Place in the Chemical World
Perfluorooctyltrichlorosilane carries a name that stretches across the label of any lab bottle, but its punch comes from the world of fluorinated chemicals. Used for ultra-thin coatings on glass, electronics, and textiles, this compound helps make surfaces repel stains, water, oils, and dust. It sounds helpful at first, but the story goes much deeper. Behind the scenes, it belongs to a family of substances called PFAS. These aren’t the easy-going sort; PFAS stay in the environment and in people’s bodies far longer than most substances we work with.
The Stubbornness of PFAS Chemicals
Chemists often call PFAS “forever chemicals.” That isn’t just a catchy phrase—it hints at a big headache. Once these compounds enter rivers, farmland, or even our tap water, they stick around for decades or longer. No routine cleaning can remove them. Back in college, I saw firsthand how challenging it was to analyze even tiny traces of fluorinated chemicals in the lab. A small spill lingered for hours, and that was just on a surface.
Studies have linked PFAS to a range of health issues, including immune system changes, hormone disruption, and cancer risks. The evidence for every single PFAS varies, but the pattern stays consistent: once these molecules land in the environment or in people, they don’t leave quietly.
Looking for Trouble: Exposure and Toxicity
Perfluorooctyltrichlorosilane has the added twist of being reactive when exposed to moisture. It throws off hydrochloric acid, which burns skin, eyes, and lungs. In industrial settings, engineers suit up and stick to safety protocols for a reason. Without proper gear or ventilation, exposure can leave workers dealing with nasty chemical burns or lasting lung irritation.
Even if workers avoid the immediate risks, breakdown products or downstream contamination can reach communities. Across the US and Europe, discoveries of groundwater filled with PFAS have sent folks scrambling for solutions. Towns near chemical plants or military bases, where PFAS got regular use, have faced expensive cleanups and growing worry over water safety.
Why This Should Matter to Everyday People
People don’t need to see chemical equations to care about what ends up in their environment. In my own town, nobody paid much attention to PFAS until the water tasted odd one summer. Suddenly, parents asked tough questions at city council meetings. If regulations feel slow to adapt, it isn’t because communities don’t care—often, the evidence builds first. But the damage of persistent chemicals isn’t easy to reverse.
Perfluorooctyltrichlorosilane, thanks to its persistence, raises the same alarm bells as its chemical cousins. Every study showing a risk, every cleanup effort that drags for years, all point to the need for smarter chemical policies and workplace practices.
What Can Be Done?
Engineers and scientists push for alternatives that don’t stick around so stubbornly. For companies, investment in containment, closed systems, and improved waste treatment isn’t always cheap, but the cost of ignoring the problem runs higher in the long run. Regulators need clear data from unbiased researchers, not just company studies, when setting exposure levels and cleanup standards.
Ordinary people can support local groups demanding better transparency and environmental monitoring. Pressuring officials for regular water testing, sharing community experiences, and connecting with scientists driving new solutions keeps the issue from being swept under the rug.
Perfluorooctyltrichlorosilane finds use in high-tech manufacturing, but its risks spread far from the factory. Its place in daily life is a reminder that convenience and performance sometimes come with bigger bills down the line—paid not just in dollars, but in clean water and public health.
Staying Safe with a Tricky Chemical
People who work with high-tech coatings or advanced electronics probably know the name Perfluorooctyltrichlorosilane. In my experience, this compound brings a mix of promise and risk. One side gives you a slick surface and powerful water repellency. The other side brings real hazards if you treat it casually. Safety with this stuff is not just a set of rules. It’s about respect for chemistry and a reminder that short-cuts can cost health or worse.
What Makes Perfluorooctyltrichlorosilane Unforgiving
You can spot danger right away: “trichloro” means chlorine atoms ready to react, and “perfluorooctyl” signals a stubborn, almost indestructible tail. Even a drop of water near an open bottle launches fumes you do not want to breathe. It doesn’t just smell bad – those vapors go after skin, eyes, and especially the lungs. Long sleeves and chemical goggles show you respect the potential. Forgetting gloves once burns that lesson in deep.
Many people jump right to fume hoods for handling. Solid choice. In my own work, I never trusted just an open bench, no matter how careful I felt. Labs lose power, equipment fails, and sometimes someone bumps your arm at just the wrong time. The margin for error here is slim, and personal protective equipment isn’t a backup. It’s as basic as closing the bottle.
Control Starts Before You Open the Bottle
If your chemical’s shipped with moisture-tight packaging, don’t let it sit out while setting up your workspace. I once rushed through prep and left a vial in humid air. The mess took hours to clean, and I never made that mistake twice. Little bits on a glove spread fast, and soon enough keys, glasses, even doorknobs get a thin coat of trouble.
Good ventilation really helps, but keep spills from happening in the first place. Glass containers work better than some plastics, since some solvents dissolve plastics or let fumes through. I’ve seen people wrap containers in extra-layers to shield them from light or accidental splashes. Store them away from acids, water, and especially anything that reacts with halogenated chemicals. Mixing things up can ruin good work and personal health in one unlucky day.
Thinking Beyond Labels: Training Matters
Written instructions matter, but habits protect people around you just as much as rules. Anyone using this silane in professional settings should advocate for real training—not just a sign-off on paper. Watching a seasoned chemist walk through setup, you pick up tricks only learned from experience. Marking storage shelves and bins avoids confusion, especially at shift changes or in shared spaces.
Disposal counts for as much as handling. This chemical shouldn’t go near regular trash or drains. Get local environmental guidelines. Chemical companies jump to regulatory talk, but the best advice comes from lab techs who know which waste streams are safe and which aren’t. In my time working with silanes, double-tagging waste and updating logs stopped more than one mix-up before it grew into a bigger headache.
Why Getting It Right Pays Off
Mistakes with Perfluorooctyltrichlorosilane last longer than one spill or burned hand. The stuff doesn’t break down easily. Letting a few drops get loose means longer clean-ups and lost lab time, not to mention trouble for cleaners, coworkers, and the environment. Using common sense—splitting up large volume bottles, labeling well, working slowly—can save money, health, and your team’s trust. No fancy equipment needed, just steady habits and respect for the science.
What Makes Perfluorooctyltrichlorosilane Stand Out?
Perfluorooctyltrichlorosilane isn’t something that grabs headlines, yet walk around nearly any industrial campus and you’ll see its impact in subtler ways. You’ll probably run into it in labs, around electronic plants, and in packaging lines. It’s not famous like big-name polymers or plastics, but engineers and researchers rely on it for one reason: it fights moisture, oil, and sticky substances with a stubbornness you just don’t see every day.
Electronics: Keeping Surfaces Dry and Clean
Take the electronics industry: microchips and circuit boards don’t play well with water or fingerprints. Any contamination can wreck a batch, cost millions, and delay production. Tech companies bring in Perfluorooctyltrichlorosilane to coat delicate surfaces. The result? Water droplets bead up and roll away, dust wipes off without a trace, and oils have nowhere to stick. This isn’t just about keeping things tidy — it’s about protecting the heart of everything digital from corrosion and failure.
Glass and Optics: Clarity Above All
Now shift to optics. People need their lenses and screens crystal-clear, whether it’s for medical scopes, camera gear, or smartphone displays. Drop a bit of this compound on a glass slide, cure it, and suddenly the glass shrugs off smudges like a raincoat. I remember touring a lab where scientists measured anti-fog coatings for surgical tools. The glass treated with Perfluorooctyltrichlorosilane stayed spotless through steam and handling, proving its worth where every second counts.
Packaging: Stubborn Stains and Sticking are Out
Modern packaging demands that food, pharmaceuticals, and even cosmetics last longer and stay pure. A thin layer of this silane compound on packaging film or bottle liners makes sure nothing leeches in or out. Ever struggled to pour out honey or shampoo without leaving a mess? There’s a good chance coatings born from this chemical kept those containers slick and easy to use. That smoother flow isn’t just about convenience; it keeps waste down and hygiene up.
Medical Devices: Reliability Matters
Perfluorooctyltrichlorosilane also found its way into medicine. Medical professionals have little patience for sticky syringes or cloudy surgical covers. A good coating keeps instruments functional during intense procedures. It doesn’t just block fluids — it stops organic material from clinging to device surfaces. Cleanups get faster, sterilization becomes easier, and device longevity improves. In a hospital, every minute matters, and reducing any source of contamination helps cut infection risks.
The Road Ahead: Tackling Sustainability
The shadow over all these benefits comes from environmental questions. Fluorinated chemicals like this one resist breakdown in nature, sometimes accumulating in water or living tissue. Regulators and green chemists have started asking hard questions about whether we should keep using them as we do now. Some companies chase alternatives, using non-fluorinated coatings or inventing ways to recover and recycle waste. Setting stronger disposal rules and finding safer replacements can help balance industry need with environmental responsibility.
Credibility and Evidence
Discussions about chemicals like Perfluorooctyltrichlorosilane must rest on trusted research and transparent reporting. Reviewing published studies, company case reports, and regulatory reviews gives us real insight into both value and risk. The story of this compound is one where progress always walks side by side with accountability.
Most folks dealing with perfluorooctyltrichlorosilane don’t spend much time thinking about shelf life until a sticky bottle turns up on the back of a dusty lab shelf. Silanes like this aren’t just ordinary chemicals you can toss in a corner and forget. For those who work in laboratories, surface sciences, or advanced manufacturing, proper storage is not a suggestion—it’s a necessity. Skipping the basics puts projects and safety at serious risk.
What Makes Perfluorooctyltrichlorosilane Different
Perfluorooctyltrichlorosilane belongs to a group of compounds that bond with surfaces, creating water-repellent coatings that drive the innovation behind everything from anti-fingerprint screens to lab-on-a-chip devices. I’ve seen labs where improper storage leads to crusty, useless silane that ends up costing both time and budget. In the wrong conditions, this chemical doesn’t just “go bad”—it reacts with ambient moisture and the result isn’t pretty.
Real Storage Conditions: No Shortcuts
Just about every reputable lab supplier will tell you to keep this silane in tightly sealed bottles, far from moisture. Humidity cracks open the seal on quality. Even professional scientists get tripped up here. Once opened, the countdown begins: hydrolysis steals away the ingredient’s effectiveness. So I always remind my team to use freshly opened containers for critical surface treatments, especially when research integrity hangs in the balance.
Temperature matters here too. This chemical doesn’t appreciate the heat. Storing it at room temperature in a cool, dry spot gets the job done. I’ve learned from long days in poorly ventilated rooms that warmth speeds up unwanted reactions. Cold storage—in a fridge or freezer—can stretch the shelf life but only if you guard against condensation, especially after pulling it out. Someone in my lab once left a bottle uncapped for an hour on a muggy summer day. That bottle ended up as hazardous waste.
Shelf Life: How Long Is Long Enough?
Under factory-sealed conditions, perfluorooctyltrichlorosilane can last over a year, sometimes two, with little problem. But after the seal breaks, the clock drops fast. Most suppliers I’ve dealt with recommend using an opened bottle within months, sometimes just a few weeks if the lab’s air feels humid. Use-by dates might feel arbitrary but expired chemicals rarely deliver reliable results.
It’s tempting to stretch the life of expensive chemicals. I’ve learned the cost rarely pays off. That “good enough” bottle may end up contaminating surfaces or sabotaging entire experiments. Keeping close tabs—regularly checking for haze, particulates, or changed color—means trouble rarely sneaks up.
Solutions: Make Good Habits the Norm
It pays to run regular inventory checks and write the opening date on every bottle. Training never stops, especially for new hands in the lab. If someone sees moisture inside a bottle, we treat it as contaminated. No half measures. Clever tech like desiccant cabinets, and tracking software can’t replace good habits, but they sure help. Budgets always push back on over-ordering, so estimating annual needs goes a long way in keeping only fresh, ready-to-use stock on hand.
At the end of the day, proper storage of perfluorooctyltrichlorosilane protects not just the quality of research but the health and safety of everyone in the lab. It’s about respecting both the science and the people behind it.