The Story Behind Perfluoropentanoic Acid: Understanding Its Role and Risks
A Historical Glimpse into Perfluoropentanoic Acid Development
Perfluoropentanoic acid carves its place in the broad family of perfluorinated chemicals, substances first synthesized in the mid-20th century. The push for more robust, water- and oil-resistant compounds led chemists down the path of manipulating carbon-fluorine bonds, known for their strength. By building out molecules with carbon chains fully fluorinated—meaning every hydrogen replaced by fluorine—researchers aimed for products that could stand up to vigorous industrial tasks. It’s hard not to notice how the scientific era of the 1940s and 1950s threw open the door to ever-newer molecules. Perfluoropentanoic acid, with its five-carbon chain and full fluorination, emerged from this enthusiasm for synthetic durability. Unlike its longer cousin, perfluorooctanoic acid, which attracted global scrutiny, perfluoropentanoic acid arrived quietly and started to find its way into niche applications where other chemicals fell short.
What Makes Perfluoropentanoic Acid Unique
Perfluoropentanoic acid’s profile stands out to anyone who knows their chemistry. It looks like a tough little chain armored with fluorine; each carbon in its five-membered backbone binds only to its neighbors and fluorine atoms—except for the exposed tail, always ready to swap protons thanks to its carboxylic group. At room temperature, it usually shows up as a colorless liquid or a pale solid, with a high melting point for such a short chain. Water and oil bounce right off it. Its stability comes at a price. In practical terms, this stuff refuses to break down easily. Both laboratory and field studies note how it sticks around—resisting sunlight, bacteria, and almost every effort to degrade it. Industrial settings like that. Environmentalists don’t.
Physical, Chemical Realities and Technical Nuance
People who work with perfluoropentanoic acid keep a close eye on its reactivity. It shrugs off most acids and bases, doesn’t rust, doesn’t flinch around harsh solvents—just the sort of qualities that chemical engineers want for making spill-proof coatings or high-humidity insulators. Its carboxylic head allows it to act as a weak acid, and it interplays with metals and certain organic building blocks in creative ways. There’s a science to labeling and handling these bottles: every safety sheet and regulatory label must spell out its stubbornness against breakdown, its tendency to hang around in the environment, and the need for gloves, goggles, or even full protective suits on the production line.
Preparation and Lab Synthesis
If you step into a facility running a synthesis of perfluoropentanoic acid, you’d see a reliance on specialized fluorination techniques. Direct fluorination—where hydrogen atoms are swapped for fluorine under pressurized conditions—creates an unmistakable whiff of laboratory precision, risk, and investment. Carefully controlled temperatures, strict containment systems, and well-trained chemists keep production both safe and predictable. Other synthesis tracks build out longer chains and then “crack” them down to five carbons, trimming away excess with acids or other reagents. Here, procedural know-how trumps brute force: getting those fluorines all lined up requires meticulous timing, the right catalysts, and sometimes a willingness to throw out a batch if purity doesn’t meet standards.
Chemical Pathways and Modifying the Molecule
The backbone of perfluoropentanoic acid opens interesting pathways for modification. Researchers who look for ways to attach this acid to larger polymers or to branch off with functional groups know that each alteration must respect the tough carbon-fluorine bonds. You don’t just bolt on additions; you plan, model, and proceed with care. In practice, scientists use coupling reactions or design specialized intermediates that allow the perfluoropentanoic skeleton to take up a starring role in more complex products. Whether tying it to a plastic backbone for industrial coatings or blending it into surfactants, each change means new questions about behavior and safety.
What Else Do People Call It?
Anyone navigating the chemistry literature finds synonyms flowing freely: perfluoropentanoate, PFPeA, pentafluoropentanoic acid. The industry’s habit of shorthand crosses borders and disciplines, but the molecule always tracks the same tight fluorinated chain, capped by that defining carboxylic acid. Some trade and lab settings also tack on numbers or structural descriptors, but these aliases trace to the same core structure.
Staying Safe in the Workplace
People who work around perfluoropentanoic acid recognize that the qualities making the molecule attractive for industry also ask for an extra level of caution. Splash one drop unprotected and you quickly realize why personal protective equipment matters; long-sleeved lab coats, splash goggles, and chemical gloves don’t just sit in cabinets—they stay in use every time the bottle comes out. Safety standards lean hard on minimizing skin and lung contact, safe ventilation, and strong protocols for spill response. Environmental standards now shape production and disposal. Facilities flush and contain their waste streams, run filter systems built to trap perfluorinated acids, and keep a digitized trail of compliance. Nobody wants a repeat of the water contamination stories that shadow the legacy of older perfluorinated compounds.
Where Perfluoropentanoic Acid Goes to Work
Out in factories and research settings, perfluoropentanoic acid isn’t about one blockbuster use. Instead, it fills smaller jobs—working as a toughening agent in specialty plastics, a surfactant base in manufacturing, and a processing aid for electronics or chemical synthesis. The emphasis lands on resilience. Whether it helps craft high-performance wires, fire-resistant coatings, or repels contamination from circuit boards, this acid earns its keep thanks to an almost stubborn ability to avoid breakdown. Its applications make sense in jobs that punish weaker chemicals—or where a lost coating, a water leak, or a sticky residue simply isn’t an option.
The R&D Race: Where Scientists Push Forward
In university labs and corporate research centers, the energy around perfluoropentanoic acid heads in two directions: finding new places where its toughness pays off, and tackling the problem of cleanup after use. On the product side, innovations focus on integrating shorter perfluorinated chains into safer chemical blends. The thinking is that shorter chains may slip past the persistent bioaccumulation problems that plague longer relatives. Meanwhile, academic groups study how to tweak the molecule so it can do a job and then degrade on cue. Researchers in environmental chemistry trial new ways to capture and neutralize the acid, whether through advanced carbon filters or chemical destruction that leaves no legacy pollutants behind. Funding agencies and journals keep a spotlight on these efforts, knowing the stakes go beyond the chemical business—public trust and environmental health both sit in the balance.
Pain Points: Toxicity and What the Studies Say
For years, few beyond the chemistry world paid much attention to perfluoropentanoic acid’s toxicology. That’s changed. Scientists trace the chemical in water, soil, and sometimes the bloodstream of both workers and neighbors. The shorter chain of PFPeA doesn’t promise safety without proof. Animal experiments track its movement through organs, showing that while it tends not to linger as long as longer cousins, it still resists breakdown and can accumulate with repeated exposure. Concerns concentrate on liver health, kidney function, and possible links to hormonal disruption; the data remains early, but regulators pay close attention. Safe handling guidelines and workplace exposure limits get reexamined each year as toxicologists turn up new findings. People who grew up around the debates on PFOS and PFOA recognize the pattern: don’t wait for a problem to get out of hand before drawing limits and asking tough questions about bioaccumulation, toxicity, and long-term health.
Looking Ahead: Future Prospects for Perfluoropentanoic Acid
Legislation and science drive changes in perfluoropentanoic acid’s future. Some industry players seek full substitutes—alternatives that skip perfluorinated chains completely. Others double down on closed-loop systems, recycling and capturing every drop used. Researchers push to invent molecules that tackle the same tough jobs but break down safely. Many believe that only a mix of stronger regulation, smarter product redesign, and serious investment in cleanup technology can untangle the long shadow left by perfluoroalkyl acids. In this mix, community advocacy also matters. Public demand for transparency on chemical use and water safety forces companies and regulators to stay accountable. It takes real investment to monitor, innovate, and—when needed—phase out products whose risks start to outweigh their industrial payoffs. The story of perfluoropentanoic acid, like so many chemicals of the modern era, is far from finished.
What’s Perfluoropentanoic Acid Doing in Our Everyday Lives?
Most people never hear about perfluoropentanoic acid unless they’re reading the tiny print on an industrial chemical label. This compound, part of the larger family of per- and polyfluoroalkyl substances (PFAS), shows up behind the scenes in more processes than a casual observer might guess. Those who spend time in factories or work with coatings and textiles may have crossed paths with it, even without knowing.
Invisible Helper in Industrial Chemistry
Perfluoropentanoic acid gets picked for jobs that need a serious boost in water and oil resistance. I’ve talked with textile engineers who rely on chemicals like this for their rain jackets and carpeting. They don’t care if a fabric looks great—if it can’t shrug off coffee or rainwater, it doesn’t make the cut. So PFAS chemicals get used in surface treatments and coatings. Something as simple as a stain-resistant couch or water-repellent tent often owes its durability to these compounds.
Besides textiles, manufacturers work pfa into the process for making fluoropolymers. These tough plastics can handle repeated friction, high temperatures, and aggressive cleaning, and they stick around in everything from cellphone cables to chemical piping. Perfluoropentanoic acid works as a processing aid, helping the plastic take the right shape and consistency. The versatility here explains both the deep industry reliance and the challenge of moving away from it.
The Problem That Lurks Beneath the Benefits
Reading studies published by environmental researchers, the warning signs are clear: perfluoropentanoic acid doesn’t break down quickly. Once it gets into water or soil, it sticks around for decades. This property turns out to be both a blessing and a curse. Good if you want a frying pan to stay nonstick forever, bad if you care about what trickles out of a landfill.
Health experts have raised alarms about PFAS linked to certain health problems, including thyroid disease and some cancers. There’s debate on the exact danger level for humans, but enough studies have created a consensus that exposure needs to drop. Drinking water gets tested more often now, with stricter guidelines for what’s considered safe. The “forever chemicals” nickname for PFAS isn’t just clever marketing; it points to their ability to hang around so long that they show up in blood tests decades after exposure.
Industry Change Doesn’t Happen Overnight
Replacing perfluoropentanoic acid isn’t easy when whole production lines and product standards depend on it. Some companies have started to switch to alternatives that break down faster, though they can wind up costing more or performing a little worse. Government regulations push the industry toward cleaner options, and customers push too. Anybody who shops for “PFOA-free” cookware plays a part in this transition.
Watching the PFAS debate unfold has shown me that real change doesn’t come from just one direction. It takes scientists digging into new formulas, regulators holding polluters accountable, and consumers asking what’s in the products they buy every day. Awareness about what goes into household and industrial goods will push companies to rethink what they use, and hopefully, phase out chemicals with long-term harm.
Looking Forward
Companies face an uphill climb balancing performance against safety. Greater transparency about what’s in our environments—and our bodies—drives research for safer chemical substitutes. Government rules and public demand for cleaner manufacturing will stick around. I expect to see more investment in safe chemistry and green technologies, even if the road is long. Knowing the full story behind a chemical like perfluoropentanoic acid makes choosing better paths possible.
Looking at the Risks
Perfluoropentanoic acid (PFPeA) doesn’t stir much recognition in everyday conversation, but it lives in the same family as the chemicals that have made headlines for their durability and potential health risks. Born from the world of per- and polyfluoroalkyl substances (PFAS), PFPeA joins a long list of chemicals that stick around not just in products, but also in our water, food, and bodies. I’ve followed the slow roll of news about PFAS for years, and PFPeA’s profile keeps growing as more labs track its footprint across the globe.
The Ubiquity Problem
The persistence of these chemicals offers manufacturers benefits — stain resistance, nonstick properties, and the kind of durability that makes products last. Living in a house with kids and pets, stain resistance sells peace of mind. At the same time, once these compounds hit soil or water, they don’t break down easily. That’s where the trouble deepens. PFPeA shows up in rivers, groundwater, and sometimes in household drinking water, based on studies out of Europe and the US. Some of those findings come from university labs, and government agencies in both regions have called out the need for deeper monitoring.
Underrated Health Concerns
Research keeps pointing to the idea that short-chain PFAS like PFPeA don’t get filtered out of our bodies as quickly as we once thought. Initial industry stories suggested less risk compared to older, long-chain PFAS, like PFOA and PFOS. But more recent findings link even these smaller cousins to effects on liver function, hormone disruption, and potential changes in immune response. Sitting down with neighbors who worry about water quality, it becomes clear why this conversation matters. No one wants to discover years later that routine exposure, even in tiny amounts, carried a hidden cost.
What the Science Shows
Toxicologists and environmental chemists studying PFPeA highlight its strong carbon-fluorine bonds, which help it resist breakdown. While agencies such as the US Environmental Protection Agency and the European Food Safety Authority regularly update lists of concerning PFAS chemicals, PFPeA often appears on watch lists. Animal studies point to possible kidney and liver effects, though scientists continue to fill in the gaps around human health data. A lot of the data comes from occupational exposures and environmental testing — not from day-to-day living — but early signals urge caution.
Moving Toward Solutions
Watching communities advocate for PFAS-free water brings a sense of hope. Municipalities invest in carbon filtration systems, and some push for cleaner alternatives during manufacturing. Government agencies encourage independent testing and keep tabs on levels in sources of drinking water. Families pick up home-grade filters, and public demand grows for safer consumer products.
Solutions come down to transparency, better science, and pressure from people who want answers. Regulators could require clearer labeling for products that contain PFPeA. Research labs can keep running long-term health studies, since short-term results rarely tell the full story. Companies that make products with these chemicals hold responsibility, too, for ensuring that alternatives don’t simply substitute one risk for another.
Accountability and Choice
Growing up near industrial sites underscored how slow change can be when health risks surface. As more communities test their water and demand greater corporate responsibility, those facing the challenge of PFPeA realize that clean living isn’t just about making safe choices at home — it comes from pushing for accountability, scientific clarity, and policies that put health before convenience.
Understanding the Risks Behind the Science
Perfluoropentanoic acid, or PFPeA, doesn’t make headlines every day, but it quietly raises important questions about safety, health, and our responsibilities around chemicals. I’ve seen plenty of labs and industrial spaces over the years, and certain basic rules keep people, equipment, and the environment out of harm’s way. PFPeA serves as a good reminder that chemical handling isn’t an abstract concern; it can have very real consequences.
Why Attention to Detail Matters
PFPeA comes from a group of chemicals known for their environmental persistence. Once it finds its way into water or soil, cleanup can get messy, expensive, and drawn out. This class has been linked to negative health effects, showing up in regulatory discussions and some contamination scandals. Even a single mishap with storage can be enough to spark a situation that lingers for years. That alone should push anyone handling PFPeA to treat storage as non-negotiable.
What Proper Storage Looks Like
A bottle of PFPeA doesn’t announce itself as hazardous just by sitting on a shelf, but trouble starts where laziness or short-cuts creep in. From experience, I’ve learned that safe chemical storage means taking every precaution possible, not just the bare minimum. Corrosive materials like this acid ask for containers that won’t react — often, this means high-density polyethylene or fluorinated plastics. Even then, regular inspection for leaks or breaks pays off.
Acid vapors and splashes present another challenge. I’ve seen good lab practice where folks keep these chemicals inside well-ventilated, dedicated storage cabinets, far away from anything that could burn, react, or dissolve. Spacing each container to prevent accidental mix-ups saves headaches down the line. Moisture only adds to the risks, so dry environments and tight seals work best. Every time a label wears out, that’s a chance for confusion — fresh, clear labeling should be routine.
Protecting More Than Just Workers
Some chemicals only concern the people who handle them directly. PFPeA drags in the larger community when accidents happen. That’s what makes spill containment and secondary storage non-negotiable. I’ve seen people use trays to catch leaks before they can spread, and it never feels like an overreaction. There’s no undo button if PFPeA gets into groundwater or drains. In places with children or vulnerable adults nearby, extra barriers and warning signs speak volumes about a company’s real priorities.
Looking Forward: More Than Just Following Rules
Many guidelines come from hard lessons learned the tough way. Reading about contamination crises or workplace injuries where basics weren’t followed always leaves me shaking my head. Laws set the baseline, but culture determines if organizations go further. Training new staff, running through emergency drills, and keeping policy documents up to date actually make a difference. Companies that treat these routines as “just paperwork” court bigger problems than they realize.
In the end, PFPeA reminds us what’s at stake: workers deserve safe jobs, neighbors deserve clean water, and regulators look for proof that businesses take their roles seriously. The best storage approach comes down to planning, vigilance, and respect for chemicals whose effects stretch far beyond a warehouse door.
The Story Behind the Molecule
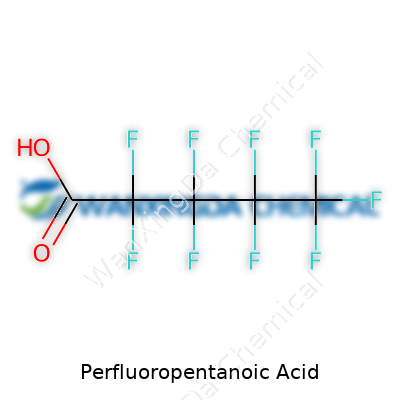
The chemical formula of Perfluoropentanoic Acid is C5HF9O2. At first glance, it might look like one more line in the endless catalog of industrial chemicals. But behind these letters and numbers lies a story about modern manufacturing, environmental ripple effects, and how society navigates the push and pull between progress and sustainability.
Real-World Use and Real-World Consequences
I remember chatting with a former chemist from a manufacturing plant in Louisiana. His badge was still dusty from factory floors lined with pipes and drums labeled with complicated names. He pointed out how these perfluorinated compounds keep popping up in day-to-day materials—nonstick pans, firefighting foams, waterproof fabrics. Perfluoropentanoic Acid, in particular, features a string of five carbons bonded to a flock of fluorines, an arrangement that forms a backbone hard for nature to break down.
This chemical sticks around in the environment far longer than most people expect. That's not just hearsay—scientists have measured these compounds in groundwater, rivers, and even in fish pulled from lakes a thousand miles from the nearest chemical plant. Research from environmental groups and agencies confirms these findings, showing that perfluorinated acids accumulate over time and spread well beyond their places of origin.
Why Structural Details Matter
The formula C5HF9O2 breaks down into five carbon atoms, one hydrogen, nine fluorines, and two oxygens. That isn't just trivia. The dense pack of fluorine atoms gives the molecule its famed resilience. For people working in chemical safety, that formula signals lasting power. For neighbors of a factory, it means any spill or runoff doesn't wash away easily. And for health officials, it raises tough questions about long-term exposure and cleanup.
Unlike biodegradable substances, perfluoropentanoic acid resists standard water treatment methods. Regular activated carbon filtration doesn’t pull it all out. That adds a new level of challenge for communities trying to keep drinking water safe. In the United States, state and federal agencies have begun tracking levels and funding research into new filtration or destruction techniques. These aren't abstract debates; real decisions about public health ride on understanding the guts of chemicals like this one.
Looking for Smarter Solutions
No one expects manufacturers to toss out chemistry that works. Nonstick coatings and stain-proof gear make life easier, and emergency crews rely on the high-performance foams made with these acids. But the same stubborn resilience that makes perfluoropentanoic acid attractive in design turns into a thorn on the tail end. Some companies have started exploring safer alternatives, while researchers chase after methods like high-temperature incineration or electrochemical destruction to break down these bonds once and for all.
Public pressure keeps the spotlight on these compounds, not because the public craves a science lesson, but because families want clean air and water. Policy changes, like stricter limits and transparency on where these chemicals turn up, only go so far unless backed by solid science and innovative engineering.
Trust Through Clear Information
Knowing the chemical formula of perfluoropentanoic acid isn’t just for trivia buffs or lab techs. It cuts to the heart of urgent questions—how do we balance the benefits of advanced materials against the risks of chemicals that outstay their welcome? Reliable, factual data helps everyone, from regulators to workers to the families drawing water from the tap, ask sharper questions and push for meaningful answers.
Perfluoropentanoic acid doesn’t show up in everyday conversations, but it’s the kind of chemical that deserves a lot more attention. This “forever chemical,” a member of the PFAS family, doesn’t break down under ordinary conditions. News stories about contaminated drinking water and mounting waste streams have left many communities feeling uneasy—especially after learning that even at low levels, PFAS compounds have been tied to health risks. I’ve watched my own neighborhood face concerns over groundwater and felt the frustration that comes from a lack of clear answers. The real question becomes: how do we keep these stubborn substances from turning into yesterday’s environmental mistakes?
Recognizing Where the Problem Starts
Industrial sites, waste handling operations, and labs all contribute to perfluoropentanoic acid waste. While regulations now push businesses to track and report their emissions, improper handling still happens when guidance feels murky or disposal costs run high. Tossing residue into ordinary landfills or flushing down drains guarantees trouble. Rain, snowmelt, and time move those chemicals into water supplies and local ecosystems. Fish pick them up. People end up drinking them. Kids might carry those lifelong burdens without ever knowing what’s getting into their system.
Safe Disposal Isn’t Magic—It’s Science Paired with Responsibility
Traditional incineration often leaves behind dangerous byproducts unless the process runs at the right temperature and for long enough. Few facilities meet strict standards set for PFAS destruction. Landfilling in a lined cell might look secure, but leachate tests show these chemicals escape containment over the years. The science teaches us there is no “away”—if we bury it or burn it without care, everyone eventually pays the price.
To really solve it, communities and industry should tap into the best available technology. High-temperature incineration operated with real-time monitoring, plasma arc reactors, and advanced chemical treatments have proven safer for reducing these compounds to non-toxic forms. Research keeps pushing for methods that break the carbon-fluorine bond at the core of every perfluorinated molecule. Still, these solutions cost more and require more oversight. I’ve heard neighbors in my local water committee ask, “Who’s going to foot the bill?” That’s a debate every city and state faces today.
Emphasizing Transparency and Tracking
Any company generating this waste owes the public a clear log of where it goes and how disposal happens. State inspectors must get the resources to follow up, with real penalties for those who cut corners. I believe citizens should insist on seeing these records—the same way they expect access to water quality reports. Cleanups and containment plans should come with open meetings where families can ask questions and push for improvements. Secrecy creates distrust. Openness drives better practices.
Finding a Middle Ground with Prevention
While disposal matters, the best solution remains to quit using persistent PFAS chemicals unless there’s no safe substitute. Product reformulation isn’t easy, but the pressure to find safer alternatives means fewer gallons of hazardous waste waiting for expensive end-of-life treatment. The recent push by leading firms to phase out the worst compounds offers a bright spot in a tough debate.
The Bottom Line
Safe disposal of perfluoropentanoic acid stands out as an urgent challenge. High heat, new destruction technologies, tough reporting, and a focus on transparency will protect people far better than business-as-usual. Science offers answers, but community voices keep everyone honest. We all need the courage to demand more than the minimum, because cleaning up after PFAS costs much more than preventing its release in the first place.