Perfluorotoluene: Tracing the Chemical’s Story and Looking Ahead
The Roots and Pathway of Perfluorotoluene
The story of perfluorotoluene began with the larger family of perfluorocarbons. In the golden age of twentieth-century chemistry, teams pushed for alternatives to standard aromatic hydrocarbons, and researchers landed on fully fluorinated rings—sturdy, low-reactivity compounds with potential in specialty areas. Perfluorotoluene didn’t headline breakthroughs the way Teflon or Freon did, but it showed up reliably in scientific conversations, particularly in the labs studying fluorine’s effects on aromatic molecules. Chemists gained more control over fluorination techniques in the 1960s and 70s, making it possible to prepare materials like perfluorotoluene with decent purity and reasonable yields, despite fluorine’s appetite for chaos.
Physical and Chemical Personality
With a pale, clear appearance and a persistently sweet, ether-like odor, perfluorotoluene doesn’t look all that special on the surface. Get below the senses, though, and its physical footprint sets it apart from the toluene most people remember from paint thinners. The molecular structure replaces hydrogen atoms with fluorine—these C–F bonds make it stubbornly resistant to breaking down, holding up against acids, bases, oxidation, and even aggressive heat. Its density lands above water, its volatility raises caution about ventilation, and its vapor carries a slight weight that’s easy to underestimate in a closed space.
Technical Details, Labels, and the Real-World Bottle
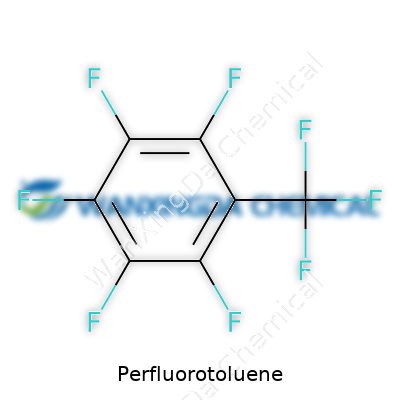
A label on a bottle in an academic lab reads with a cold certainty: chemical formula C7F8, molecular weight over 200, a boiling point hovering near 103°C. There’s no question about what’s inside; this material offers a sharply defined singularity—every corner of the molecule populated by the fluorine atom. With the world focused on material traceability, the correct technical specs reduce confusion, especially as new suppliers and international standards come into play. The tiniest contaminant can throw synthesis off course or confound analytical runs, so clarity in labels makes all the difference, and professionals stress traceability from the shipping manifest to the waste container.
Getting Perfluorotoluene: Synthesis and Chemical Tricks
Bringing perfluorotoluene into existence calls for strong nerves and smart equipment. Direct fluorination still terrifies even experienced chemists—run it too hot, and you get a blast of side products; too slow, and yield drops below practical use. Electrochemical fluorination sets the tone these days, where toluene dissolves into anhydrous hydrogen fluoride, and a silent jolt of electricity replaces fiery blasts in metal reactors. The result? Hydrogen swapped decisively for fluorine at every possible site. Still, there’s a dance: parameters such as temperature, current, electrode surface, and solvent ratios decide whether a flask yields pure stuff or a stubborn mess that sticks to glassware. No recipe fits every lab, but the community’s learned to walk the line between safety and productivity.
From Product Names to Synonyms—Talking About the Same Thing
The bigger the chemistry world gets, the longer the list of names for the same tool. A bottle’s label might read “octafluorotoluene,” “toluene, perfluoro,” or just “C7F8.” Scientists know what they’re seeing, but new students and cross-border collaborators stumble on names, so plain talk always wins: stick with perfluorotoluene, double-check the structure, and forget the naming debate. Language in chemistry is never static, but for practical purposes, clarity trumps creativity.
Safety Concerns and Operational Practice
With perfluorotoluene’s chemical stubbornness—those tight carbon-fluorine bonds won’t crack for ordinary reagents or mild heat—people often overlook its less obvious hazards. Poor ventilation can lead to vapor build-up, and high exposure might provoke headaches or throat scratchiness. Like other fluorinated aromatics, it doesn’t break down in the environment and can stay in air and water systems for a long time. Gloves and eye protection aren’t optional if any chance exists of touching or splashing the liquid. There’s another side to the safety issue rarely discussed outside technical circles—disposal taxes local systems, because ordinary incineration won’t fully destroy the molecule. Waste streams need high-temperature, specialized incinerators to force breakdown, which draws attention from regulators and communities watching water quality.
Where Perfluorotoluene Fits In
Chemists reach for perfluorotoluene in places where nothing else delivers. Analytical teams rely on it as a non-polar solvent for NMR and electron capture detection, sidestepping reactivity. Its full-fluorine armor makes it inert where ordinary toluene turns reactive. Industry leans on it as a benchmarking standard, reference for purity, or intermediate in making pharmaceutical and polymer precursors. Some electronic manufacturers look for ways to harness its chemical stability for specialty coatings, where device reliability rules over cost. As environmental regulation clamped down on other perfluorinated compounds, perfluorotoluene’s small total market kept it just under the radar—but it never dropped off the map.
Pushing the Chemistry—Reactions and Tweaks
Research groups keep poking at perfluorotoluene to uncover new reactions, but its very strength—resistance to breakdown—chokes off traditional organic reactions. With brute force, it can undergo nucleophilic aromatic substitution, where only the most determined reagents nudge a fluorine off the ring. Some scientists use it as a scaffold, building new materials that marry stability with subtle surface tweaks. Most of the world’s chemistry stops short of heavy modification, shifting research focus to using perfluorotoluene as a platform for finding tougher, smarter molecules for electronics or imaging.
What’s New—Ongoing Research and Open Doors
The investigative spirit around perfluorotoluene hasn’t faded. Academic and industrial labs look at it through the lens of sustainability and risk reduction. A push exists to understand how different bacteria and high-energy catalysts might degrade perfluorotoluene, eager to loosen fluorine’s grip and keep persistent materials from building up in landfills or groundwater. Another thread tackles environmental detection, with new sensors and analytical standards rolling out every few years, hoping to keep communities a step ahead if spills occur. There’s a shift in grant money, too: regulators and agencies now want results that connect lab practice with full-life-cycle environmental modeling.
Putting Toxicity Under the Microscope
Toxicity studies on perfluorotoluene trail behind those for more common perfluorinated compounds. What’s clear is that its resistance to environmental breakdown raises red flags, not only for what it does in living systems, but for how long it lingers. Some early studies hint at low acute toxicity for mammals at expected exposure levels, but data gaps persist, especially concerning chronic, low-dose scenarios and impacts on aquatic life. Communities near chemical plants keep up the pressure for stricter rules and clearer answers, demanding transparency in reporting and better baseline studies on bioaccumulation. Until scientists fill those blank spaces, the cautious route wins out: limit exposure, control emissions, and raise the bar for monitoring.
Looking Forward—Prospects and Pressures
Perfluorotoluene’s future sits at a crossroads shaped by technical innovation and public expectations. The field can’t ignore growing calls for sustainable chemicals and tighter regulation on persistent pollution. If new synthesis routes cut down emissions and breakthroughs in green chemistry deliver faster decomposition, perfluorotoluene could play a valuable, responsible role in analytical sciences and electronics. There’s room to expand applications if the community solves the waste problem and keeps toxicity in check with comprehensive studies. On the flip side, failure to adapt or provide rock-solid safety and environmental data could shrink the market. The next chapter will be written by how well chemists balance innovation with humility—trusting decades of experience and a few good, hard questions about what makes a chemical truly worth it in the long run.
The Understated Power of Chemical Innovation
Walk into any lab worth its salt and you’ll find rows of chemicals most of us have never heard of. Perfluorotoluene is one of those quiet powerhouses. You won’t see it on supermarket shelves, yet its influence stretches far past the lab doors. Think of it as a workhorse in the world of specialization—built for jobs where ordinary chemicals would fall flat.
More Than Just a Solvent
Perfluorotoluene is heavy with fluorine atoms, which means it’s one of those compounds that shrug off heat and don’t care much about mixing with water. That gives it a place in industries from electronics to pharmaceuticals. Its role as a solvent stands out, especially for chemists chasing high-purity results. It lets them separate, dissolve, or purify substances in ways that regular solvents just can’t handle.
Real-World Reach: Electronics and Engineering
Anyone dealing in electronics faces a nemesis: heat. Soldering circuit boards, making microchips, or building sensitive components demands more than patience. Perfluorotoluene steps up here, carrying heat away without breaking down. Its resistance to thermal decay gives engineers peace of mind. They can focus on innovation, knowing their cooling agents won’t add new problems.
Its chemical stubbornness also helps in cleaning. Standard solutions sometimes leave behind thin films or react with the surfaces they’re meant to clean. Perfluorotoluene, on the other hand, strips away grime without sticking around—no residue, no strange interactions. This comes in handy for cleaning optics, circuit boards, or even specialized glassware. Every manufacturer aiming for spotless clarity keeps one eye on their solvents, and this one better suits the job than most.
Beyond Industrial Use: Tackling Environmental Concerns
With its strength comes a big question: What happens after perfluorotoluene leaves the factory? Its chemical makeup means it lingers. We’ve seen what persistence can do with similar “forever chemicals” like PFAS—showing up in groundwater and taking decades to degrade. The risk here is real. Companies have to think about how they dispose of it, not just how they use it. Countries including the US and EU are paying tighter attention to these compounds, drafting new regulations every year.
Better solutions often start at ground level—better waste processing, tighter controls over disposal, and more research into replacements that don’t last quite so long in nature. Some labs are now testing biodegradable alternatives for jobs once ruled by perfluorotoluene. While progress is slow, every experiment helps shave down future risks.
Straight Talk: Responsibility Comes Standard
Knowing what substances like perfluorotoluene can do is half the battle. The other half is choosing responsibility over convenience. Open discussion among chemists, regulators, and everyday consumers drives improvements. As more people call for safer handling and tighter oversight, real shifts follow. Cutting corners with powerful chemicals can seem easy, but cleaning up later costs far more—both in dollars and in trust.
Perfluorotoluene’s story is bigger than industry or chemistry. It’s about what happens when human curiosity creates tools both powerful and stubborn. The reality is simple: Use the best tools for the job, but never stop looking for ways to make them safer for everyone.
The Chemical in the Middle
Perfluorotoluene isn’t a household name. Still, it lives in some important corners of the science and industry world. This stuff belongs to a much bigger family of chemicals that goes by the name of perfluorinated compounds, which have been used for decades. They’re tough molecules, almost stubborn in how they refuse to break down in the environment or in our bodies.
Why We’re Worried
Plenty of chemicals show up in products and processes, but perfluorinated chemicals catch extra attention lately, mainly because of what science has learned about their cousins – the infamous PFAS (per- and polyfluoroalkyl substances). These related compounds have built up in water, soil, wildlife, and people, became a hot topic for environmental groups, and showed up in headlines around the world.
So, anyone working with Perfluorotoluene faces real questions: How dangerous is it? Will it stick around and cause problems the same way? Initial research points out that, like other perfluorinated chemicals, Perfluorotoluene resists breaking down. That’s its greatest strength for industry, but a real headache for health and safety folks. Once it gets out, it’s not easy to get rid of.
Health Risks and Toxicity
Looking into the science, researchers haven’t finished mapping out the risks of Perfluorotoluene for people or the environment. Still, decades of data on similar molecules make it clear: we shouldn’t assume it’s safe just because there isn’t a big file of incidents yet. PFAS exposure links to a range of problems—certain cancers, immune system changes, and developmental effects. Animal testing around the family of perfluorinated chemicals shows that some of these molecules hang out in bodies for years.
Even if Perfluorotoluene hasn’t been accused of those specific effects, its chemical stubbornness earns it extra scrutiny. I’ve worked in labs where gloves and fume hoods were standard when handling fluorinated solvents. These are not chemicals you want soaking into your skin or floating into your lungs. And once a chemical like this escapes outside, nobody’s got a good way to clean it up fully. That worries anyone who cares about public health and clean water.
Regulation and Next Steps
Some government agencies, including the EPA, track new perfluorinated substances closely. In recent years, companies and researchers have shed more light on the long-term persistence of these compounds. That’s led to bans, tough limits, and expensive cleanup projects for PFAS pollution, especially near factories and military sites.
Perfluorotoluene’s name isn’t in the law books as often as PFOA or PFOS, but if the past is any indication, regulators tend to catch up eventually. Trying to swap out problematic chemicals can lead to a cat-and-mouse game—one compound fades, only for a close cousin to take its place.
What’s the Solution?
We need a different mindset about chemicals that last forever. Scientists and watchdogs push for thorough testing before new chemicals leave factory doors. Transparency helps people in communities near plants understand what they’re facing, and regular testing of air and water stops surprises years down the line. Companies can use less persistent alternatives and close the loop on hazardous waste so it doesn't become someone else’s problem.
No one enjoys being told that a useful material can also be a long-term headache. Still, those are the trade-offs that real science and responsible industry have to face. It’s better to ask hard questions early than pay the price later.
Looking Beyond the Formula: Why Know Perfluorotoluene?
People rarely give much thought to chemical formulas unless faced with toxic spills or a lab experiment gone sideways. But knowing the basic makeup of perfluorotoluene opens a window into broader questions about industrial chemistry and our environment. The formula is C7F8. The whole idea hinges on swapping every hydrogen atom out of toluene and fitting in fluorine atoms instead. This twist gives the molecule its distinct character, since fluorine’s stubborn bonds resist breaking down. The structure follows the familiar ring of benzene, but each carbon on the ring, and the methyl group attached, hold fluorines instead of hydrogens—eight fluorine atoms, seven carbons.
Where Perfluorotoluene Shows Up
In chemical labs, folks have turned to perfluorotoluene as a specialty solvent or as a building block for advanced materials. Think of certain electronics or unique polymers built to survive harsh conditions. Industries needing high resistance to heat, acids, or bases find value in these fluorinated compounds. There’s also a footprint in specialty coatings. All of this comes with a price: fluorinated chemicals don’t break down easily.
The Persistent Perfluorinated Puzzle
One big reason to care is environmental persistence. The same strength that makes C7F8 attractive for industry makes it stubbornly hard for nature to deal with. These perfluorinated compounds can linger in soil and water far longer than most organic chemicals. Researchers have tracked their presence in rivers, lakes, and even remote ecosystems. Some even build up in wildlife. Regulatory scrutiny has grown intense, and “forever chemicals” is no exaggeration—cleanup feels endless and expensive.
Balancing the Books on Benefits and Risks
Chemists and engineers wrestle with a classic trade-off. On the one hand, molecules like perfluorotoluene deliver unmatched durability and chemical resistance. On the other hand, the downside takes generations. Properties like low reactivity and tough bonds make accidental releases a long-term concern. Just ask folks living near chemical plants—they know the cost of past carelessness when it comes to chemicals that outlast any manager’s career.
Potential Solutions: It Starts in the Lab
Prevention beats cleanup every time. Chemists now look for ways to design chemicals that get the job done but won’t stick around forever. There’s new work on less persistent alternatives, sometimes by shortening fluorinated chains or mixing in other atoms that let nature break a molecule down over time. Scientists also push for better methods to break apart these compounds, either through high-energy treatment or using catalysts developed specifically for tough fluorinated bonds.
The Call for Responsibility
Responsibility for chemicals like perfluorotoluene should rest both with those who manufacture and those who use these compounds. Stronger oversight and transparency build trust with communities. Public access to chemical release data and a clear plan for containment can limit surprises. People working in industry have the know-how to lead on this, not just follow regulations. Experience teaches that cleanup gets harder the longer it’s put off, and public faith in scientists can fade quickly unless harm is prevented up front.
More Than a Formula
Understanding perfluorotoluene goes way past memorizing “C7F8.” There’s a need for vigilance and creativity in chemistry. Environmental stewardship runs alongside scientific progress. As someone who’s spent time talking with both chemists and neighbors near manufacturing sites, I see the central lesson: decisions set in a lab can echo through nature and society for decades. That lesson deserves close attention with every new compound, from familiar old solvents to next-generation materials.
Practical Realities in the Workplace
I’ve seen too many labs try to cut corners with chemicals they think are “just another solvent.” Perfluorotoluene may sound like a mouthful, but pretending it’s harmless is more dangerous than folks realize. My own time in research hammered home that chemicals with fluorine deserve both respect and close attention. The reality is, a cavalier attitude in the storage room or at the bench can quickly turn into a health issue for you or your coworkers.
Trust But Verify Storage Conditions
I learned the hard way—never trust others' labeling or storage practices at face value. Perfluorotoluene thrives in tightly sealed containers, kept well away from heat, ignition sources, and direct sunlight. In one lab I worked in, someone left a volatile solvent on an open shelf near a sunny window. No one ever got hurt, but that bottle swelled up more than once.
Don’t assume ventilation systems will cover your mistakes. Air-tight containers aren’t negotiable for this compound, and corrosion-resistant shelving avoids nasty surprises. Some will store similar chemicals with acids or strong oxidizers, thinking it saves space, but that’s a great way to court disaster. Segregation by hazard class doesn’t take much effort, yet saves headaches that echo long after a spill.
Respect the Hazards
Even after years of lab work, I still check the safety sheets before handling perfluorotoluene. Its vapors can irritate the eyes, skin, or respiratory tract — a few minutes of exposure might not show immediate harm, but repeated incidents add up. I’ve seen coworkers brush off small spills, only to spend the afternoon rubbing irritated skin and cursing themselves. Gloves, goggles, and lab coats aren’t up for debate. Chemical-resistant gloves make a world of difference, especially after hours working in close quarters.
Don’t Wait for a Wake-Up Call
Hard lessons stick with you. One afternoon, a minor fume leak had half our lab coughing for hours. Since then, our team invested in better spill kits, regular HVAC checks, and a culture that never shames folks for asking safety questions. Training drills aren’t a chore—they’re insurance against acts of forgetfulness or bad luck.
Routine inspections don’t eat up as much time as some think. I’ve made it a habit to scan labels, check cap seals, and confirm chemical compatibility when shelving new bottles. Teams that talk openly about storage and handling build trust, avoid silent mistakes, and act quickly if something goes wrong.
Solutions Rooted in Real Life
Complacency creeps in fast in busy labs. A peer review of storage practices helps catch what individuals miss. Digital tools—simple spreadsheets or inventory trackers—keep tabs on expiration dates and quantities. Outdated stock should not gather dust. Prompt disposal means one less hazard during an emergency, like fire or flood.
People treat chemicals as safe as their leaders do. I’ve seen supervisors who set good examples shape new habits for everyone else. Every bottle of perfluorotoluene deserves the same respect you’d show a bottle of strong acid or a noxious gas. We don’t have to live in constant fear—just treat every step in the process with vigilance, routine, and a dash of humility.
The Physical Face of Perfluorotoluene
Perfluorotoluene strikes you as a bit of a chemical oddball the first time you come across it. Most liquid chemicals have a smell; some burn your nose, others waft quietly from the glass. Perfluorotoluene, though, keeps pretty quiet. Uses in labs taught me it’s a colorless liquid—clear as water—which sometimes tricks you into thinking it's harmless. At room temperature, it pours smoothly, almost like a thick spirit, and it doesn't evaporate fast, thanks to a high boiling point. That’s a key detail because it shows up in places where stability and staying power count.
This liquid doesn’t dissolve in water. Drop some into a beaker and it beads up, clinging to itself and pushing away anything not like it. Hydrophobic is the right word, but to me it always looked stubborn—unwilling to play with the ordinary crowd. Instead, Perfluorotoluene blends easily with other fluorinated and chlorinated solvents, which comes in handy for complicated chemical work. Its density tops water, so in a lab bottle, it settles underneath, creating pronounced layers that help with precise separations.
The Chemical Backbone: Stability with a Catch
Structurally, perfluorotoluene’s claim to fame comes from swapping all the hydrogen atoms on toluene for fluorine. This simple act makes it much less reactive than regular aromatic compounds. Fluorine atoms, packed around that ring, throw up shields; almost nothing rattles the molecule. Those shields make perfluorotoluene tough to break down, whether by heat, light, acids, or bases. In practice, chemists appreciate this because it doesn’t degrade or burn off during long syntheses.
That toughness sits at the center of why perfluorotoluene stirs concern outside the lab. Once it enters the environment, it tends to stick around. Unlike other solvents that microbes eventually chew up, perfluorotoluene holds out for years. Known as a “forever chemical,” this resistance means it accumulates in soils and water. If you’ve ever followed the debate about PFAS compounds—per- and polyfluoroalkyl substances—this is familiar ground. Many PFAS share these stubborn backbones, and their ability to carry on unchanged causes real-world harm, from water contamination to bioaccumulation in fish and animals.
Why This Matters—And What Needs Fixing
I've seen scientists lean on perfluorotoluene for tough tasks: separating tricky organics, crafting specialty polymers, testing materials for resilience. Industry benefits because the chemical keeps working, cycle after cycle. Yet, that very persistence puts us in a bind. Regulatory agencies now flag perfluorinated compounds as emerging contaminants. Research keeps piling up about their traces showing up in drinking water and wild places far from chemical plants.
Solutions start with using less and recycling what’s used. Facilities can switch to closed-loop processes, so the compound cycles from one application to the next without dumping leftovers. Chemists also need to design alternatives that break down after use. Green chemistry isn’t just a buzzword—it’s how we keep craft and industry safe for the next wave of workers and neighbors. We’ve already seen early versions of shorter-chain and partially fluorinated solvents that leave a lighter mark on the environment.
No single answer will solve it. Building cleaner chemistry takes discipline, openness, and, frankly, a willingness to rethink convenience. Perfluorotoluene’s properties give us plenty to admire, but they also demand respect. That's a lesson worth remembering, in labs and beyond.