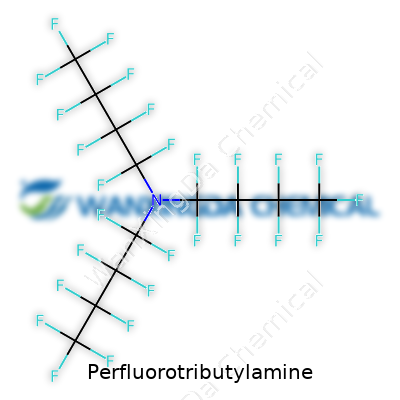
Perfluorotributylamine: History, Uses, and Where It's Going
A Glimpse Into History
Anyone who’s ever looked at the evolution of specialty chemicals recognizes Perfluorotributylamine (commonly called PFTBA or PFTBA-N) as something of an oddball. Developed back in the 20th century, PFTBA didn’t show up because someone woke up one day wanting a fancy coolant, but as a byproduct of tinkering with fluorinated organics. It was the chemical industry’s push for stability, heat resistance, and inertness that brought PFTBA into wider attention. Laboratories across North America and Europe explored it for electronics and high-vacuum technology, long before folks worried much about environmental legacy.
What Makes Perfluorotributylamine Stand Out
Call it a mouthful, PFTBA brings to the table a slick pedigree of resilience. It resists breaking down even under roasting temperatures, and water rolls right off it thanks to its perfluoro backbone. Unlike standard amines, swapping out hydrogens with fluorine atoms creates a non-polar compound, so PFTBA doesn’t like to mix with most things—neither acids nor bases have much luck with it. You rarely see that kind of chemical stubbornness. And in my own experience reading through scientific archives, every five years or so researchers start to see new uses just because it refuses to react like most other amines.
Breaking Down the Properties
PFTBA looks like a clear, oily liquid at room temperature. With a boiling point soaring past 170 degrees Celsius and a freezing point well below most living-room refrigerators, it hangs around as a liquid almost everywhere. Its density, noticeably higher than water, tells you right away it’s packed with fluorine. And if you ever sniff near a sample, you’ll notice almost no odor, which is rare for an amine. It holds on to its electrons, seldom gives up a proton, and shrugs off UV light. In electronic fields, engineers see this and recognize reliability, even if it takes special storage to avoid leaks and evaporation.
Technical Specifications and Labels
On any chemical shelf, you’d spot PFTBA under many guises: Perfluorotributylamine, PFTBA, or N(tributyl)trifluoroamine. There are standardized labeling requirements, though—hazard diamond, lot number, and purity rating. From what I’ve seen in labs, few manufacturers bother with lower-grade product because applications demand near-total purity. While regulations crack down on improper disposal and shipment, not every country syncs up rules for labeling or transportation, putting an informal onus on researchers and industry buyers to keep up.
How It’s Made: The Guts of Production
Creating PFTBA involves swapping in fluorine atoms into organic structures. Traditionally, chemical makers start with tributylamine and introduce elemental fluorine or specialty fluorination agents under strictly controlled settings. The process demands meticulous venting, scrubbing, and waste recovery systems because fluorination byproducts are nothing to mess with. Workers need special suits, too, not only from risk of chemical burns but because uncontrolled fluorination sometimes releases toxic gases. The push for greener methods surfaces from time to time, yet the heart of the process—direct, aggressive fluorination—stays unchanged due to limited options.
Chemical Reactions and Modifications
Most chemists at first glance think, “Maybe this will react with strong acids,” given its amine backbone. In practice, PFTBA’s tough exterior means it shrugs off almost every standard reaction. It doesn’t hydrolyze in water, doesn’t burn easily, and resists all but the harshest oxidizers. If you look into journals on compound modifications, researchers sometimes graft functional groups onto the molecule’s periphery to force reactivity, but they admit PFTBA resists transformation about as much as Teflon. Efforts to break it down for recycling lag, which stirs debate in environmental science circles.
Synonyms and Product Names
You might see this compound listed as FC-43—widely used as an electronics coolant. Sometimes academics call it “trihydroperfluoroamine” or use old trade names, yet the most common label in supply catalogs remains PFTBA. This leads to confusion for newcomers: reading a research article, getting lost between chemical abstracts, only to realize the same stuff is being discussed under five different monikers.
Safety and Operational Do’s and Don’ts
While daily handling looks easy, PFTBA deserves real respect. It doesn’t burn your hands, but its vapor can displace oxygen in confined spaces. Lab veterans know you use closed systems, gloveboxes, and ventilation hoods, since even a mild leak can create a suffocation risk near the floor. Labels on shipping containers draw attention to fire and environment even though PFTBA itself won’t flash. The bigger risk often comes during waste collection or leaks near heat sources that could break down some residues into nasty perfluorinated byproducts. Waste streams require incineration in specialty facilities, adding cost and complexity for environmental managers.
Where Has PFTBA Found Its Niche?
Ask an engineer in microelectronics or a technician dealing with analytical instruments, and PFTBA rings a bell. It features prominently in gas chromatography as a calibration standard, especially for mass spectrometry. NASA and aerospace programs settle on it for thermal management in delicate circuits due to zero reactivity and non-conductivity. Older refrigeration systems sometimes used it as a heat-carrying fluid, although engineers now look elsewhere because of mounting environmental concerns. Some medical imaging techs tried it as a blood substitute for oxygen carriers—its oxygen solvency makes it an intriguing candidate, but toxicity and storage hurdles curbed wider trials.
Chasing New Applications: Where R&D Pushes Limits
Labs worldwide search for new perfluorinated compounds to handle heat, stress, or extreme environments. PFTBA keeps showing up in studies on inert media for electronics, specialty lubricants, and as a base for high-density coolants. Research isn’t limited to new uses; some groups look for additives or modifications that force it to break down under mild conditions to ease environmental pressure. I’ve read studies experimenting with catalytic systems that hope to transform these compounds into biodegradable products, but the chemical’s stubborn backbone keeps progress slow.
Toxicity: Unpacking the Concerns
Toxicologists see PFTBA as a warning flag: it doesn’t degrade quickly and persists in soil and water. Its inhalation risk comes not from chemical burns, but oxygen displacement and potential long-term effects if it seeps into the ecosystem. Research since the 2010s highlights its global warming potential, with atmospheric lifespans stretching centuries, and its presence in some urban air samples triggers concern. Regulators in Europe and Asia watch PFTBA alongside PFAS due to their fingerprint on environmental samples, and environmental scientists push hard for restricted use or alternatives. Health-wise, animal studies signal low acute toxicity, but chronic exposure paints a hazier picture, with worries about long-term accumulation outpacing any proven antidote.
Future Prospects and Solutions We Can Pursue
Over the next decade, the chemical world faces a crossroads with PFTBA and its relatives. Electronics industries crave stable, inert compounds, but society grows wary of “forever chemicals” that resist natural decay. One solution involves ramping up requirements for leak-free, fully closed systems to cut loss into the wider environment. Another involves heavier investment in biodegradation research, tracking down microbes or catalysts that can start the breakdown of perfluorinated amines without creating new hazards. Policy-makers toy with outright bans, yet the lack of suitable substitutes in some fields means pragmatic, phased retirement offers smoother transitions. Trade groups and public agencies have a role to play, nudging for transparency about sources, uses, and emissions data. By sharing results and funding cross-border research, we can hope to find that sweet spot where technology and environmental sense work together, not at loggerheads.
More Than Just a Chemical Name
Perfluorotributylamine doesn’t roll off the tongue, but this synthetic chemical carries surprising weight in technology and health. Many people think of chemicals like this as esoteric—stuff reserved for scientists in white coats. My experience, watching labs blend innovation with everyday technology, shows how something obscure shapes what we use and trust.
The Backbone of Mass Spectrometry
The main scene where Perfluorotributylamine really earns its stripes is mass spectrometry. Walk into any modern analytical lab and you’ll spot sophisticated instruments dissecting samples, hunting for concentrations of molecules you can’t see with the naked eye. Perfluorotributylamine acts as a calibration standard for these mass spectrometers. It’s reliable and stable, making it the go-to molecule for labs needing to check if their machines report results accurately. This step anchors vital work in drug testing, environmental science, and food safety. The accuracy of these devices traces back to how well a chemical like this calibrates the tool.
Teflon’s Chemical Cousin
What grabs my attention with perfluorinated chemicals is their ability to resist heat and chemical breakdown. Perfluorotributylamine is in the same class of chemicals as the ones found in nonstick cookware and firefighting foams, which means it won’t react or degrade easily. This stability isn’t just a marvel of chemistry—it poses challenges if the compound escapes the lab. The features that make it useful are the very same ones that allow it to linger in the environment.
Environmental Persistence and Human Safety
Perfluorotributylamine stands out because of its persistence. Studies highlight that many perfluorinated chemicals, sometimes called “forever chemicals,” accumulate in the environment. They don’t break down easily and can end up in water sources, soil, and even human tissues. Research continues to uncover how exposure affects health, with links to immune responses and developmental conditions showing up. Regulators and advocacy groups worldwide now scrutinize the use and disposal of perfluorinated compounds for good reason.
Balancing Progress With Responsibility
Innovation relies on tools that push the limits of measurement and safety. Perfluorotributylamine has given researchers the precision to analyze everything from drugs in a bloodstream to contaminants in park soil. My view: Every new invention comes with responsibility. Scientists and industries benefit from dependable chemicals, but can do more to prevent unnecessary release. Stronger collection methods, closed systems for handling, and investment in alternatives with shorter environmental footprints all look promising. In my own professional experience, the commitment to sustainability and transparency often comes from the ground up—scientists who see firsthand how their work touches public health.
Learning from the Forever Chemicals Story
Chemicals like perfluorotributylamine taught us that what happens in the lab doesn’t stay there. Continuous research and updated regulations offer hope that technology can strike a better balance—protecting both people and the planet without slowing down discovery. It starts with knowing more about what ends up hidden in our tools and water, and working together to find smarter paths forward.
People ask a lot about strange chemicals. Perfluorotributylamine (PFTBA) doesn’t ring a bell for most of us unless you’re knee-deep in labs, but questions about its danger seem fair. These days, trust in chemicals takes a hit—too many stories about toxic PFAS, polluted water, and sick communities. We get nervous, especially if long, unpronounceable names pop up.
Looking at PFTBA More Closely
PFTBA belongs to the family of perfluorinated compounds. That makes a lot of people nervous right away. Headlines about “forever chemicals” stick in our memories. PFTBA shows up in some electronics, testing fluids, and sometimes specialty industrial uses. Researchers know it persists, meaning it takes a very long time to break down in the environment.
Most of what’s public about PFTBA centers on its role as an insulating liquid or a heat-transfer medium. Some scientists tried to study how it interacts with the atmosphere; it turns out PFTBA is a potent greenhouse gas—thousands of times stronger than carbon dioxide at trapping heat. That raises some eyebrows, especially for anyone worried about climate change, but toxicity to humans or animals remains poorly documented. The Environmental Protection Agency and other agencies haven’t listed it as a high-priority chemical for human health risks—yet. Absence of proof doesn’t mean proof of absence.
Why It Matters
People have good reasons to pay attention. We’ve seen the fallout from other perfluorinated chemicals in water, food, and everyday items like nonstick pans and stain-resistant sprays. These compounds don’t leave the body quickly. Exposure to some PFAS links up with higher rates of cancers, developmental problems in kids, even higher cholesterol. That doesn’t mean every chemical in this group is automatically deadly, but the evidence drives a “better safe than sorry” feeling.
Workers handling PFTBA directly should use gloves and eye protection, since inhaling fumes or getting it on skin could cause irritation. It’s also pretty clear we don’t want this stuff leaking into streams, soil, or the air if it can be helped. Safety data from manufacturers skims the surface: avoid breathing the vapor, prevent spills, and use good ventilation.
What Can Be Done?
Chemicals like PFTBA demand careful management. It’s easy for those using it in tech labs, energy companies, or research centers to think a little spill doesn’t add up to much. But that thinking is how persistent chemicals end up scattered through the ecosystem. I’ve read about communities fighting for years over contaminated groundwater, so asking hard questions about PFTBA's lifecycle isn’t alarmist—it’s practical.
One path is clear labeling. Any product using PFTBA ought to name it directly so people can make informed choices. Facilities that handle it should set up containment systems and plan for emergencies. Regulators could keep a closer watch as new research emerges. Scientists could also run more toxicity tests—including independent studies—so health professionals and citizens get answers driven by evidence, not just industry claims.
Most people want honesty over polish. If the risks turn out low, say so. If surprises turn up later, don’t sweep them under the rug. We’ve seen too many slow burns with other synthetic chemicals. Good stewardship means respecting the long game—both for workers and the public down the road.
Understanding Perfluorotributylamine
Perfluorotributylamine carries the chemical formula C12F27N. This formula might look intimidating to most people, but breaking it down opens a doorway into a bigger conversation about where science meets everyday life. Twelve carbon atoms, twenty-seven fluorine atoms, and one nitrogen atom, wrapped together in a way that’s all about stability and resilience.
Perfluorochemistry and Human Health
This compound belongs to the family of perfluorinated chemicals, commonly known as PFAS. Most have heard these called “forever chemicals” — not just a catchy nickname, but a sign of real world impact. PFAS don’t break down easily in nature, and that toughness comes straight from their chemical structure. Even a casual scan through the news makes it clear how persistent these molecules can be in water, soil, and eventually, in our own bodies.
There’s been plenty of talk about the risks tied to PFAS exposure — immune issues, hormonal changes, or impacts on fetal development. The U.S. Environmental Protection Agency and other organizations have warned about certain members of this family in drinking water. If you look at the formula for perfluorotributylamine, you see the building blocks that make these molecules so durable: carbon–fluorine bonds that outlast most natural processes.
Uses That Raise Questions
Labs around the world use perfluorotributylamine as a calibrant for mass spectrometry, mostly due to its stability. Its resistance to breakdown helps in giving precise, repeatable results. But after leaving the lab bench, leftover traces head somewhere. Waste streams from research and industry don’t always get captured. As someone who has worked in labs, I know about the tug-of-war between chasing accuracy and managing waste. Scientists take pride in their precision, but questions always bubble up about what happens to these chemical traces after the experiment ends.
The trouble with molecules like perfluorotributylamine is not that they’re bad in isolation. The formula gives you an unbreakable cage, but the real headache shows up when that same cage floats into rivers and lakes or rides the wind into food supplies. People downstream keep asking: how do you clean up something that resists breaking apart for centuries?
Rethinking Chemical Stewardship
Most would agree: science shouldn’t walk with blinders on. The breakthrough that lands a sharper mass spectrometer reading eventually bumps into bigger public health conversations. We have to start expecting more from chemical management, not just in labs but through the whole supply chain. Safer alternatives should get fast-tracked any time they can do the job with less environmental kickback. Industry and research should share data about toxicity and breakdown profiles without hiding behind trade secrets.
Some countries have set limits for PFAS levels in water. It’s a start, yet rules alone won’t catch every molecule. The chemical formula C12F27N serves as a reminder: chemical resilience in the product often becomes chemical persistence in the environment. Once that’s understood, it gets easier to push for better cleanup tech and to back policies that hold polluters responsible for every step from lab shelf to watershed.
Not Just Any Chemical: Understanding the Challenge
Perfluorotributylamine (PFTBA) stands out among chemicals. Most folks have never heard of it, but anyone working in a research lab or industrial setting understands its punch. This colorless liquid works as a calibration standard in mass spectrometry and has applications in electronics, refrigeration, and high-voltage systems. Its molecular stability makes it valuable, but that same property creates real storage and handling questions most chemicals don’t raise.
Lessons From the Storeroom
Over years of working in labs, storage rooms taught me their own brand of lessons. Some bottles leaked and others reacted far too easily. PFTBA requires extra attention—not because it bursts into flame if you look at it wrong, but because its stability and low reactivity bring quiet risks: environmental persistence and extreme slipperiness. Picture a spilled drop rolling across an otherwise clean floor: it resists cleanup, and without eye-catching odors, it slips the mind until someone slips on it. I’ve seen busy researchers curse at unseen patches that caused tools (and people) to skate across concrete floors.
Securing PFTBA takes a certain discipline. Keep it in tightly sealed bottles, away from direct sunlight and away from heat sources. Its vapor can be heavier than air, and over time, that can settle unnoticed in laboratory trenches or floor drains. Regular containers with tight-fitting lids do most of the job, but double containment (a chemical-resistant tray under every bottle) reduces risk. Labeling every bottle sharply reduces confusion on rushed shifts.
Why Ventilation Matters
Poor ventilation in storage spaces creates headache-inducing problems. Even though PFTBA won’t explode under room-temperature conditions, its vapors linger if rooms lack good airflow. Staff working near the storage area need clear knowledge about any leaks, since even low vapor concentrations become dangerous over the long term. Sufficient ventilation means more than propping open a door. Exhaust fans, vented cabinets, and annual checks on air handling systems mean fumes don’t gather where people breathe. Skimping here costs health and trust, both fiercely defended commodities in real labs.
Personal Safety: Not Just Gloves and Goggles
On hectic days, it’s tempting to skip some protective steps. I remember grabbing a bottle of PFTBA with bare hands just once because gloves seemed too much for a two-second job. My skin felt oily for a full day afterward. Even though it doesn’t burn or sting, long-term skin exposure brings risks no one wants to discover through trial and error. Nitrile gloves keep hands protected, and splash-proof goggles guard eyes from the rare but messy spill. Lab coats live up to their reputation during rough days, keeping surprises off regular clothes.
Waste and Environmental Responsibility
Perfluorinated compounds don’t break down quickly. They collect in water and soil, and over time, those environmental leftovers show up in places no one intended. Disposal needs to follow local hazardous waste rules. Pouring leftovers down the drain leads to real damage. Institutional waste collection turns a nuisance into a process, often involving high-temperature incineration designed to break apart the persistent chemical bonds. Better to handle the upfront inconvenience than to wait for downstream effects on communities and wildlife.
Handling PFTBA never fits the “out of sight, out of mind” approach. With tight procedures, informed staff, and a solid respect for the chemical’s quirks, real accidents become rare. Trust, once built through safe handling routines, benefits everyone who walks through those doors.
A Chemical That Lingers
Perfluorotributylamine, often abbreviated as PFTBA, has carved out a spot in advanced electronics and specialty applications due to its unusual chemical stability and resistance to breakdown. Just about anything that resists chemical change this strongly tends to draw attention from scientists who study what sticks around in our environment—and for good reason. My own background working with complex materials taught me that persistent chemicals can move along unintended paths. Neatly contained in a lab or a factory, PFTBA appears dull. Released into the wild, it can start to matter a lot more.
Unpacking the Risks
PFTBA does not mix well with water or break down in sunlight or soil like more familiar everyday compounds. Studies identify PFTBA as a long-lived greenhouse gas, thousands of times more potent than carbon dioxide in trapping heat, making its atmospheric presence more than just an interesting detail. Decades of environmental research confirm that such persistent compounds do not disappear on their own. Reports show that a single kilogram released can linger for centuries, adding to a cumulative burden in the air.
The struggle comes from the same characteristics that make PFTBA useful: it just does not break down. Its chemical bonds, loaded with fluorine, resist weathering, fire, and even heavy-duty treatment methods. Experience in industrial chemistry circles knows this kind of molecule means trouble if release controls go lax. The consequences are not immediate, but over years, persistent gases set the stage for gradual but real damage, whether in subtle atmospheric shifts or in hard-to-trace bioaccumulation.
Responsible Handling and Disposal
Talking with colleagues in environmental monitoring, most agree that the goal is to avoid introducing PFTBA to the environment at all. Lab-scale handling already calls for fume hoods and closed systems. For industries that use or produce this chemical, guarding against leaks has become a baseline practice, not just a suggestion. There’s no standard landfill or sewer capable of breaking it down, so dumping is out of the question.
Best practices tap into high-temperature incineration, though it takes very specialized equipment to destroy PFTBA’s fluorinated structure without making other toxic byproducts. Thermal processes operating above 1,200 degrees Celsius promise the most thorough destruction, something ordinary municipal waste facilities simply cannot offer. A few industrial-scale incinerators, mainly those licensed for hazardous wastes, can take on jobs like this with the right controls in place to catch escaping gases.
What’s Next for Regulation and Safer Chemistry?
Regulators, from the EPA to international panels, already track many perfluorinated chemicals closely given risks of persistence and bioaccumulation. While regulations may not yet single out PFTBA as strongly as PFOS or PFOA, growing scrutiny suggests that disposal rules will only tighten. Staying ahead of the curve means swapping PFTBA for alternatives wherever possible or investing in recovery and destruction technologies that stop releases at the source. Experience tells me that real safety comes from a blend of foresight and honest recognition of the risks—much more than a label or a promise on a datasheet.
This issue calls for sustained attention across labs, manufacturers, and government. Any practice that cuts down PFTBA’s spread, even before official rules demand it, protects air and climate for the long haul. People in the field know: the best time to start planning responsible disposal is far before the first drop hits the waste drum.